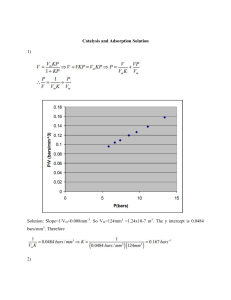
Chapter 2 GENERAL RADIOCHEMISTRY I- ISOTOPIC EXCHANGE I- ISOTOPIC EXCHANGE 1- Basic concepts Isotopic exchange is the redistribution of atoms of isotopes of a given element within the molecule, between different molecules or phases, which does not lead to other changes in the qualitative and quantitative molecular composition of the system. XR + X*R’ === X*R + XR’ XR X*R’ Isotopic exchange involves the substitution of one isotope of an element by another isotope of the element in the molecules of a given substance without a change of their element composition. For example, if hydrogen chloride HCl, enriched by the heavy isotope of chlorine 37Cl, is mixed with chlorine Cl2 with the normal isotope composition (75.53 percent 35Cl and 24.47 percent 37Cl), then as a result of the isotopic exchange reactions H37Cl + 35Cl2 = H35Cl + 35Cl37Cl (H-37Cl + 35Cl- 35Cl H-35Cl + 35Cl-37Cl) H37Cl + 35Cl37Cl = H35Cl + (H-37Cl + 35Cl-37Cl H-35Cl + 37Cl 2 37Cl-37Cl) Isotopic exchange reactions may take place under various conditions: • Under homogeneous conditions For example, between solute and solvent, isotopic exchange reactions is interchange of iodine atom between alkyl iodide and potassium iodide in alcohol solution. R127I + K131I == R131I + K127I • Under heterogeneous conditions For example, between solids or liquids, the interchange of silver atoms between metallic silver and its ions in solution 110Ag + 107,109Ag (solid) == 107,109Ag + 110Ag (solid) Isotopic exchange reactions play an important role in, • The synthesis of labeled compounds. H2O R-MgI + *CO2 R-*COOH • The investigation of redox process of C = C CH3 – CH = CH2 KMnO4/H+ CH3 – COOH + *CO2 KMnO4/OH- COOH – *COOH + CO2 2- Mechanism of isotopic exchange – Isotopic exchange by dissociation – Isotopic exchange by association – Isotopic exchange through other reversible chemical processes. – Electron exchange reactions Mechanism of isotopic exchange • Isotopic exchange by dissociation The scheme of an exchange reaction through dissociation is as follows: AX == A + X BX* == X* + B AX* BX For example • In the heterogeneous of halide ions between solid silver halide and metal halides in solution AgBr + NaBr* == Ag+ + Br - + Na+ + Br* == AgBr* + NaBr • In the homogeneous of halide ions in solution between metal halides and compounds containing halogen atoms as ligands: K2[PtBr6] + KBr* == 2K+ + [PtBr5]- + Br - + K+ + *Br == K2[PtBr5Br*] + KBr • The thermal dissociation mechanism operates in the exchange of sulfur atoms between sulfur monochloride and elemental sulfur in vapors: S2Cl2 + *S8 == S2 + Cl2 + *S2 + S6 == *S2Cl2 + S8 • Isotopic exchange by association The exchange reaction may be represented schematically as follow: AX + BX* == ABXX* == AX* + BX Example: The inter-exchange of bromine atoms between hydrogen bromide and molecular bromine. HBr + *BrBr == HBr2Br* == HBr* + Br2 • Isotopic exchange through other reversible chemical processes. Example The intramolecular exchange of carbon atoms in methylcyclohexane between the side chain and the ring. C6H11 - *CH3 == CH3 – C5H8 – *CH3 == CH3 - C5H11C* - *CH3 CH3 - – *CH3 CH3 - * • Electron exchange reactions. The transfer of electrons from the isotopic atoms which are contained in compounds of a given element in different oxidation states leads to a redistribution of the isotopes, the atoms being not actually transferred from one compound into another. FeCl2 + Fe*Cl3 === Fe*Cl2 + FeCl3 Fe2+ + *Fe3+ === Fe3+ + *Fe2+ e- II- ADSORPTION OF RADIOACTIVE ISOTOPES 1- DEFINITION OF ADSORPTION Adsorption is the transfer of a material from one liquid or gaseous state to a surface. • The substance that is transferred to the surafce is the adsorbate. • The material on which the adsorbate deposits is the adsorbent. Example: Silica gel, Activated carbon, Alumina, Zeolites and molecular sieves, Polymers. adsorbate: material being adsorbed adsorbent: material doing the adsorbing. (examples are activated carbon or ion exchange resin). A (gas, liquid) adsorbent adsorbate ADSORPTION. Adsorption is the concentration of a substance at a phase boundary Adsorption Absorption M Diffusion > 99% of surface for removal is internal ABSORPTION ADSORPTION Adsorption PHASE I ‘PHASE’ 2 Absorption (“partitioning”) PHASE I PHASE 2 Causes of Adsorption • Attraction to the Sorbent Surface – Van der Waals forces: physical attraction – Electrostatic forces (surface charge interaction) – Chemical forces (e.g., - and hydrogen bonding) Van der Waals Forces A polar molecule that has two poles. Van der Waals Forces Johannes Diderik van der Waals Van der Waals Forces Two electrically neutral, closed-shell atoms dTemporary dipole resulting from quantum fluctuation d+ Gives net attraction d- d+ Induced dipole, due to presence of other dipole • Although Van der Waals forces are weak, they are often the only attractive force between molecules. Electrostatic Interactions Interaction of charged side chain with the opposide charged side chain. O + NH3 - O H2C C O C CH2 O + NH3 (CH2)4 Charges Two charges of the same type repel one another + + Two charges of the opposite type attract one another - + The two charges will experience a FORCE pushing them apart or pulling them together Adsorbents Characteristics and general requirement Most adsorbents fall into one of three classes: - Oxygen-containing compounds – Are typically hydrophilic and polar, including materials such as silica gel and zeolites. - Carbon-based compounds – Are typically hydrophobic and non-polar, including materials such as activated carbon and graphite. - Polymer-based compounds - Are polar or non-polar functional groups in a porous polymer matrix. Zeolite Graphite is a crystalline form of carbon. Graphite is the most stable form of carbon under standard conditions Adsorbents Characteristics and general requirement: * Adsorbents are used usually in the form of spherical pellets, rods, moldings, or monoliths with hydrodynamic diameters between 0.5 and 10 mm. * The adsorbents must also have a distinct pore structure which enables fast transport of the gaseous vapors. Different physical forms of activated carbon Increasing magnification Adsorbent • Activated Carbon • Activated Alumina • Silica Gel • Molecular Sieves (Zeolites) • Polar and Non-polar adsorbents activated carbons Activated alumina is manufactured from aluminium hydroxide by dehydroxylating it in a way that produces a highly porous material; this material can have a surface area significantly over 200 m²/g. It is made of aluminium oxide (alumina; Al2O3) silica gel Silica gel synthetic polymers variety of functional groups Adsorption Process Classified as: – Physical adsorption – Chemical adsorption • Physical adsorption occurs when the bonding forces are dispersion and coulombic type. • The amount of heat released during this process is equal to the heat of condensation. Physical adsorption: Van der Waals attraction between adsorbate and adsorbent. The attraction is not fixed to a specific site and the adsorbate is relatively free to move on the surface. This is relatively weak, reversible, adsorption capable of multilayer adsorption. A (gas, liquid) adsorbent adsorbate Chemical adsorption: Some degree of chemical bonding between adsorbate and adsorbent characterized by strong attractiveness. Adsorbed molecules are not free to move on the surface. There is a high degree of specificity and typically a monolayer is formed. The process is seldom reversible. Chemical adsorption • Chemical adsorption occurs when there is sharing of electrons between adsorbent and adsorbate. • The amount of heat released during this process is equal to the heat of reaction. • Heat liberated during chemisorption is in the range of 20-400 kj/g mole • Results from a chemical interaction between the adsorbate and adsorbent. Therefore formed bond is much stronger than that for physical adsorption Adsorptive Equilibration in a Porous Adsorbent Pore Early Later Laminar Boundary Layer GAC Particle Adsorbed Molecule Diffusing Molecule Equilibrium ADSORPTION EQUILIBRIA If the adsorbent and adsorbate are contacted long enough an equilibrium will be established between the amount of adsorbate adsorbed and the amount of adsorbate in solution. The equilibrium relationship is described by isotherms. Isotherm models: • Langmuir Isotherm • Freundlich Isotherm Langmuir isotherm model In Langmuir isotherm assuming a unimolecular layer can be obtained by a kinetic approach: At equilibium knowing that the rate of adsorption is equal to the rate of desorption: ra=CaP(1-f) rd=Cdf Ca P f Ca P + C d ra: rate of adsorption Ca, Cd: constant P: partial pressure of the adsorbate f: is the occupied fraction of the total solid surface Freundlich isotherm model: For the special case of heterogeneous surface energies (particularly good for mixed wastes) in which the energy term, “KF”, varies as a function of surface coverage we use the Freundlich model. 1 q e K F Ce n n and KF are system specific constants. Application of Adsorption In ordinary chemistry, adsorption is important when a gas or solution is in contact with a material of high surface area, usually either a porous or a very finely powered solid. • In clarification of sugar • In gas masks • In catalysis • In adsorption indicators • In chromatographic analysis • In softening of hard water • In paint industry • In removing moisture from air in the storage of delicate instruments 2- ADSORPTION OF RADIOACTIVE ISOTOPES The adsorption of radioactive isotopes from solution plays an important role in radiochemistry. Radioactive isotopes present in solution in micro-concentrations may be lost during the work as a result of: • their adsorption on the walls of the vessel, • contamination of solution, • precipitation process, • or due to adsorption on the previously formed precipitates, • on filters, • etc. • Ionic adsorption Ionic adsorption occurs on finely crystalline precipitates, bulky precipitates of such types as hydroxides, silica gel, alumosilic gel, etc., on particles of suspensions, colloidal particles, carbon, ionexchange materials, filter paper. In a number of cases ionic adsorption also obeys the equations of molecular adsorption. For examples: • The adsorption of Ra2+ on glass over a wide range of concentration • The adsorption of Ba2+ and Sr2+ at concentration of 5.10-3 to 10-5 M takes place according to the Freundlich equation III- DISTRIBUTION OF MICROCONCENTRATIONS OF RADIOACTIVE ISOTOPES BETWEEN TWO PHASE • Specific and most important to radiochemistry are the distribution of micro-concentrations of radioactive isotopes between a solution and a solid phase. • The distribution of micro-concentrations of radioactive isotopes between a gaseous and a liquid phase is governed by the solubility of radioactive gases in liquids and obeys Henry’s law. • If a radioactive isotope is in solution in the form of ions in an ultrasmall concentration so that when there are added substances capable of forming with a given element slightly soluble compounds it cannot form an independent solid phase, then it can be separated from solution by coprecipitation with a so-call carrier, whose concentration in the solution is sufficient for precipitation. 1- General Properties of Aqueous Solutions General Properties of Aqueous Solutions • Solution - a homogeneous mixture – Solute: the component that is dissolved – Solvent: the component that does the dissolving Generally, the component present in the greatest quantity is considered to be the solvent. Aqueous solutions are those in which water is the solvent. Concentration of Solutions • Concentration is the amount of solute dissolved in a given amount of solvent. • Qualitative expressions of concentration – Concentrated – higher ratio of solute to solvent – Dilute - smaller ratio of solute to solvent Solubility is the maximum amount of a solid that can dissolve in a given amount of solvent at a specified temperature Comparison of a Concentrated and Dilute Solution • Quantitative concentration term – Molarity is the ratio of moles solute per liter of solution – Symbols: M or [ ] – Different forms of molarity equation mol M L mol L M mol M L Calculate the molarity of a solution prepared by dissolving 45.00 grams of KI into a total volume of 500.0 mL. Calculate the molarity of a solution prepared by dissolving 45.00 grams of KI into a total volume of 500.0 mL. 45.00 g KI 1 mol KI 1000 mL 0.5422 M 500.0 mL 166.0 g KI 1L How many milliliters of 3.50 M NaOH can be prepared from 75.00 grams of the solid? How many milliliters of 3.50 M NaOH can be prepared from 75.00 grams of the solid? 1 mol NaOH 1L 1000 mL 75.00 g NaOH 536 mL 40.00 g NaOH 3.50 mol NaOH 1L • Dilution – Process of preparing a less concentrated solution from a more concentrated one. 2- Precipitation – precipitation reaction Precipitation – precipitation reaction Some things (chemists use term compounds) dissolve in water – these are said to be soluble. Some things (compounds) don’t dissolve in water – these are said to be insoluble. For example • Hydroxide Precipitation pH is Raised by Addition of Hydroxide Cu(NO3)2(aq) + 2 NaOH(aq) Cu(OH)2(s) + 2 NaNO3(aq) Hydroxide Precipitation of soluble metals Cu(NO3)2(aq) + 2 NaOH(aq) --> Cu(OH)2(s) + 2 NaNO3(aq) Ni(NO3)2(aq) + 2 NaOH(aq) --> Ni(OH)2(s) + 2 NaNO3(aq) 2 AgNO3(aq) + 2 NaOH(aq) --> Ag2O(s) + 2 NaNO3(aq) + H2O(l) • Sulfide Precipitation of Soluble Metals Cu(NO3)2(aq) + Na2S(aq) --> CuS(s) + 2 NaNO3(aq) CdCl2(aq) + Na2S(aq) --> CdS(s) + 2 NaCl(aq) Ni(NO3)2(aq) + Na2S(aq) --> NiS(s) + 2 NaNO3(aq) Agents to precipitate: Ammonia/Hidroxide, NH3/OHSulfide, S2Phosphate, PO43Carbonate, CO32Cyanide, CN…. A precipitation reaction is a reaction in which soluble ions in separate solutions are mixed together to form an insoluble compound that settles out of solution as a solid. That insoluble compound is called a precipitate. Precipitate Formation soluble insoluble Precipitation (formation of a solid from two aqueous solutions) occurs when product is insoluble Identify the Precipitate A lead iodide precipitate. (?) + PbI2 (?) (s) Pb(NO3)2(aq) + 2NaI(aq) 2NaNO3 (aq) Mixing Solutions of Pb(NO3)2 and NaCl Isolating the precipitate The precipitate from a precipitation reaction can be separated from the reaction mixture by filtration. Buchner funnel vacuum pump A Buchner funnel and flask can be used to accelerate the process. filter paper This apparatus uses a vacuum pump to draw the mixture through the filter. Buchner flask The filtrate is finally washed and dried. The solubility product constant, Ksp Precipitation is accomplished by combining a selected ion(s) in solution with a suitable counter-ion in sufficient concentrations to exceed the solubility of the resulting compound and produce a supersaturated solution Sr2+ (soln) + CO32- (soln) SrCO3 (s) The solubility product constant, Ksp, is the equilibrium constant for the former process, a solid dissolving and forming ions in solution. Ksp is defined in terms of the concentrations of Sr2+ and CO32Ksp = [Sr2+][CO32-] = 1.610-9 In order for the carbonate to precipitate, the product of the concentration of the ions in solution representing the ions in the equilibrium expression, the common ions, must exceed the value of the Ksp. For example, if [Sr+2] is 1.10-6 molar, then the carbonate ion concentration must be greater than 0.0016 molar for precipitation to occur because (1.10-6) x (0.0016) = 1.6x10-9. Uses of precipitation reactions Most precipitation reactions are very fast reactions that occur between ions. This makes them very useful for identifying specific ions based on the type of precipitate formed. Precipitation reactions have a number of other uses: production of coloured pigments for paints and dyes removal of toxic chemicals from water separation of reaction products. A lead iodide precipitate. Usually Used For Larger Flows Addition of OH Controlled by pH Meter / Controller NaOH Storage Clarifier React Flocculation pH Process Water Chemical Treatment .. Equalization Tank Treatment Tank Treated Water Back To Equalization Tank Clarifier Filter Press Sludge Tank Sludge Bin 3- Coprecipitation Radiochemical Precipitation Coprecipitation • Processes in which the microcomponent is carried away from the solution by a precipitating macrocomponent are called coprecipitation. • The term coprecipitation refers to the carrying down of normally soluble impurities during precipitation of an insoluble compound. Coprecipitation The simultaneous precipitation of a normally soluble component with a macro-component from the same solution by the formation of mixed crystals, by adsorption, occlusion or mechanical entrapment. For example, Radium in most environmental samples, for example, is not present in sufficient concentration to cause its very insoluble sulfate (RaSO4) to precipitate. The radionuclide can often be brought down selectively and quantitatively from solution during precipitation of an alternate insoluble compound by a process called coprecipitation. The insoluble compound commonly used to coprecipitate radium isotopes is another insoluble sulfate, BaSO4. The co-precipitate (concentrated) ion –Ra2+: Ra2+ (trace) + SO42- (soln) --/-> RaSO4 (s) (Concentration of Ra is very low) Ra2+ - the co-precipitate (concentrated) ion – the impurity ion that comes down together with main precipitate. BaSO4 - main precipitation Ba2+, Ra2+(soln) + SO42- (soln) (Ba,Ra)SO4 (s) Ba2+: the ion doing precipitate (by adding Na2SO4 solution - precipitant) Co-precipitation by mean of inorganic reagents Main precipitate Zn(OH)2 LaPO4 BaSO4 Fe(OH)3 Precipitant Zn(OH)2 Na2HPO4 Na2SO4 NH4OH Element being coprecipitated Fe, Tl, Al Cu, Mg Pb Be, Zn, Cu, Pb, Co, Cd Co-precipitation with crystalline precipitates , in which the micro-component is distributed throughout the entire volume of the solid phase and participates in the building-up of the crystal lattice of the macro-component, is known as cocrystallization. The processes of precipitation of the microcomponent on the surface of the solid phase of the macro-component are referred to as adsorption. Coprecipitation may be: - Internal coprecipitation - External coprecipitation. Internal coprecipitation may involve processe: “The coprecipitated ions replace the ions in the crystal lattice of the main precipitate. This may occur if the main precipitate and the impurity ion together with the coprecipitating agent crystallize in the same system”. For example, There takes place the coprecipitation of lead (Pb2+) with barium sulphate since both BaSO4 and PbSO4 crystallize in the orthorhombic crystal system. Animated figure of orthorhombic crystal system External co-precipitation External co-precipitation takes place due to adsorption processes. In this case, the ions that are co-precipitate are absorbed on the surface of the main precipitate. Such a co-precipitation is especially effective with precipitates that have a highly developed surface, as is the case when the precipitate is a colloid . 4- Carrier • Radiochemical analysis frequently requires the radiochemist to separate and determine radionuclides that are present at extremely small quantities. The amount can be in the picomole range or less, at concentrations in the order of 10-15 to 10-11 molar. micro 10-6 mega (M) 106 nano (n) 10-9 giga (G) 109 pico (p) 10-12 tetra (T) 1012 femto (f) 10-15 peta (P) 1015 atto (a) 10-18 exa (E) 1018 If a radioactive isotope is in solution in the form of ions in an ultrasmall concentration so that when there are added substances capable of forming with a given element slightly soluble compounds it cannot form an independent solid phase, then it can be separated from solution by coprecipitation with a so-call carrier,. For example, To determine 90Sr in environmental samples, stable strontium (containing no radioisotopes of strontium) is added to increase the concentration of total strontium to the point that the common ion effect causes precipitation. The added ion that is present in sufficient concentration to cause a precipitate to form is called a carrier. 90Sr2+ (ultratrace) + SO42- (soln) --/-> 90SrSO4 (s) (Concentration of 90Sr is very low 90SrSO don’t precipitate) 4 Addition of stable strontium (Sr2+) - to increase the concentration of total strontium Sr2+(soln) + SO42- (soln) (90Sr, Sr)SO4 (s) Sr2+ - carrier • The use of a material that is different in isotopic make-up to the analyte and that raises the effective concentration of the material to the macro level is referred to as a carrier. Separations are most easily accomplished when performed on a macro scale • In many cases, the carrier is a nonradioactive isotope of the analyte (Isotopic Carriers). • Some carriers are stable isotopes of chemically similar elements (Nonisotopic Carriers). Isotopic Carriers An isotopic carrier is usually a stable isotope of the analyte. Stable strontium (consisting of naturally occurring 84Sr, 86Sr, 87Sr, and 88Sr) is frequently used as the carrier in the analysis of 89Sr and 90Sr. Nonisotopic Carriers Nonisotopic carriers are materials that are similar in chemical properties to the analyte being separated, but do not have the same number of protons in their nucleus. • For several elements, nonisotopic carriers are chosen from a different family of elements, but they have the same ionic charge or similar crystalline morphology as the analyte. For example, Lanthanum and neodymium as +3 ions are frequently used as nonisotopic carriers for U4+ and Pu4+ in their final separation as insoluble fluorides (LaF3) by the process of coprecipitation Holdback Carriers • Coprecipitation of a radionuclide with ferric hydroxide carries other ions in addition to the analyte, because of its tendency to adsorb other ions and occlude them in its crystal matrix. • The addition of a holdback carrier, a highly charged ion, such as Co3+, represses counter-ion exchange and adsorption to minimize the attraction of foreign ions. • to add “holdback carriers” to analytical mixtures to prevent unwanted radionuclides from being carried in a chemical process. • Hold-back carriers: The role of hold-back carriers is to prevent the unwanted coprecipitation of radionuclides or to prevents the coprecipitation of impurities • Highly charged ions, chemical homologs, and ions isotopic with the radionuclide are among the most efficient holdback carriers. • Therefore, ions capable of displacing the radionuclide ions (the hold-back carrier) are added to prohibit the coprecipitation of the radionuclide. scavengers • Another group of nonisotopic carriers can be described as general scavengers. Substances with high surface areas, or the ability to occlude contaminants in their floc, can be used to effect gross separation of all radionuclides from macro quantities of interfering ions. • Ferric hydroxide, manganese dioxide (MnO2) and sulfides (MnS), and hydrated oxides [Mn(OH)x] are examples of these nonspecific carriers that have been used in many radiochemical separations to eliminate gross quantities of interfering substances. • Scavengers The role of scavengers is to move impurities with strongly adsorbing precipitation. The scavengers help to remove miscellaneous traces of radioactivity, leaving behind one or two chose radioelements for which hold-back carriers have been added. Ferric hydroxide and aluminium hydroxide have been widely used as scavengers. Yield of Isotopic Carriers The use of an isotopic carrier to determine the chemical yield (recovery) of the analyte is a critical step in the plan of a radiochemical analysis. The analytical method being used to determine the final amount of carrier will govern the method of separation. If a gravimetric method is to be used for the final yield determination, the precipitate must have all the characteristics that would be used for macro gravimetric analysis.easily dried, definite stoichiometry, nonhygroscopic, etc. Similarly, the reagent used as source of carrier at the beginning of the analysis must be of primary-standard quality to ensure that the initial mass of carrier added can be determined very accurately. For a gravimetric yield determination, the aquation would be the following: mass of carrier in final separation step Percent Yield = ------------------------------------------------- x 100 mass of carrier added THANK YOU FOR YOUR ATTENTION