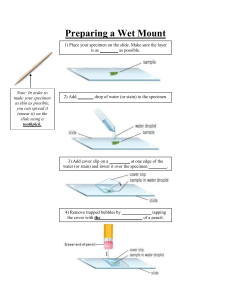
SPECIMEN COLLECTION SPECIMEN Specimen may be defined as a small quantity of a substance or object, which shows the kind and quality of the whole sample PURPOSE 1. 2. 3. 4. 5. To To To To To know the normal function of the body make diagnosis and prescribe treatment know the progress or regress of the diseases know the effect of special treatment and drugs assess the general health of the patient PRINCIPLES OF SPECIMEN COLLECTION 1. Contaminated and improperly collected specimen will produce false results which will adversely affect in diagnosis and treatment 2. Provide proper instruction and explanation to patients such as when to collect, what to collect, how to collect and quantity 3. Equipments used for collection should be neat and dry 4. Collect all kind of specimen at morning if possible 5. For culture specimen, use sterile container 6. Label the container with name, age, sex, MRD no, date, time and specimen name Types of specimen collection 1. 2. 3. 4. Urine- routine and culture Stool- routine and culture Sputum- routine and culture Blood- routine and culture COLLECTION OF URINE SPECIMEN FOR ROUTINE TEST DEFINITION Collection of a small quantity of urine sample in a clean container for testing it in the lab setting PURPOSE To identify and measure the presence of abnormalities in urine such as white blood cells, red blood cells, casts, puss cells, pH, sugar, albumin and specific gravity CHARACTERISTICS OF URINE Volume An amount of 1000 to 2000 ml of urine is excreted in 24hours. It may vary according to the water intake in the season. In winter output is more and summer it is less Variation in urine volume 1. Abnormal increase in volume is called polyuria. 2. Decreased quantity of urine or below 500ml in 24 hours is known as oliguria 3. Total absence or marked decrease in urine is known as anuria Color and the normal urine: Normal colour is pale yellow or umber or straw colored depends up on the concentration. If quantity increases the color will be pale yellow and decreases it becomes deep yellow Variations or abnormal colours: 1. 2. 3. 4. Bright red- indicates presence of blood in urine (hematuria) Pink- indicates small amount of blood in urine Smoky brown- blood pigments Milky white- chyluria due to filariasis, pus in blood Turbidity and appearance: The normal urine is clear with no deposit Odor: normal urine has aromatic odour Specific gravity: it varies from 0.010 to 1.025 Alkalinity or acidity pH value: it varies from 4.6 to 8 ARTICLES A clean tray containing, 1. 2. 3. 4. Clean, wide mouthed plastic container Bed pan or urinal Soap and water Lab investigation forms 5. Clean gloves 6. Appropriate labels marked with full name, age, sex, MRD number, date, time, name of the ward and name of the test to be done PROCEDURE S.NO 1 STEPS Identify the patient Check physician’s order and provide privacy if needed 2 Explain the procedure to the patient with special instructions about clean and wash the genital areas with soap and water thoroughly Female: Wash the urethral meatus and surrounding area. Male: Hold the penis with one hand and cleanse the end of penis moving the centre to outside using soap and water. For helpless patient: The nurses should do the perineal care. Give labelled container and instruct the patient not to wet the label on the container. Ask the patient to void the first and last part of the urine stream into the urinal or toilet and collect the middle part of the stream into the specimen container and do not touch inside of the container. Ask the patient to place the filled container in allotted place. With gloved hand place the specimen container in polythene bag. Send specimen to the lab with completed, signed lab form within 15minutes or refrigerate it. Remove gloves and wash hands. Record the procedure in the nurse’s note and lab register. 3 4 5 6 7 8 RATIONALE Obtain specific instruction and confirming the necessity of doing this procedure Privacy allows patient to relax and reduce embarrassment Washing genital area prevents contamination of urine specimen. Labelling helps in identification of samples. Collecting midstream urine avoids contamination of the specimen with organisms normally present on the skin. Four ounce (120ml) is required for the test. Unnecessary placement causes contamination. Protects the health workers from contamination of urine. To prevent decomposition of urine. It act as a proof. COLLECTION OF URINE FOR URINE CULTURE DEFINITION Collection of a small amount of urine, i.e.30 to 60ml for detecting the presence and growth of micro organisms in the sample PURPOSES 1. To culture pathogenic micro organisms present in the urine 2. To determine antibiotic sensitivity of the pathogens in the urine ARTICLES A clean tray containing 1. 2. 3. 4. 5. 6. Sterile urine container in a plastic cover Scissors (optional) Lab forms Soap and water Bed pan or urinal Appropriate labels marked with full name, age, sex, MRD number, date, time, name of the ward and name of the test to be done PROCEDURE S.NO 1 ACTION Identify the patient Check physician’s order and provide privacy if needed 2 Explain the procedure to the patient with special instructions about clean and wash the genital areas with soap and water thoroughly Female: Wash the urethral meatus and surrounding area Male: Hold the penis with one hand and cleanse the end of penis moving the centre to outside using soap and water For helpless patient: The nurses should do the perineal care and assist in giving bedpan or urinal Give labelled container and instruct the patient not to wet the label on the container Instruct to open specimen container and place cap with sterile inside surface 3 RATIONALE Obtain specific instruction and confirming the necessity of doing this procedure Privacy allows patient to relax and reduce embarrassment Washing genital area prevents contamination of urine specimen. Labelling helps in identification of samples Contaminated specimen will lead to inaccurate reporting of culture and 4 5 6 7 8 9 10 up and not to touch inside of container and lid Ask the patient to void the first and last part of the urine stream into the urinal or toilet and collect the middle part of the stream into the specimen container and do not touch inside of the container (midstream sample) Replace cap securely on specimen container, cleanse any urine form external surface of the container Ask the patient to place the filled container in allotted place With gloved hand place the specimen container in polythene bag Send specimen to the lab with completed, signed lab form within 15minutes Remove gloves and wash hands Record the procedure in the nurse’s note and lab register sensitivity Collecting midstream urine avoids contamination of the specimen with organisms normally present on the skin. Prevents transfer of micro organism Unnecessary placement causes contamination Protects the health workers from contamination of urine To prevent decomposition of urine It act as a proof Special consideration 1. Patients who are catheterized should have the specimen withdrawn using a sterile needle and syringe from the catheter’s sample port. Clamp the collection tube for about 30 minutes before taking samples 2. Urine sample should be sent to lab immediately within 30 minutes if not refrigerate it and culture done within 24hours 3. About 30 minutes prior to collecting the specimen, patient may be advised to drink fluids unless contraindicated 24 hours urine specimen For a 24hours urine specimen, all urine voided in a 24 hour period is collected. The collection is initiated at a specific time, which is noted, and the client is asked to empty his bladder at that time. The urine is discarded. After this all the urine is collected in a common receptacle for the next 24hours. Usually, it is done from 6AM to 6AM of next day STOOL/FAECES ROUTINE TEST AND CULTURE Collection of stool specimen for specific or routine test PURPOSE 1. 2. 3. 4. To identify specific pathogens To determine the presence of blood, ova and parasites To determine the presence of fat To do gross examination of stool characteristic such as colour, consistency and odour NORMAL CHARACTERISTICS OF FAECES 1. 2. 3. 4. 5. Colour: light to dark brown Odour: pungent smell Frequency; 1 to 2 times per day Quantity: 4 to 5 ounces per day Composition: 30percent of water, shed epithelium from intestine, a considerable quantity of bacteria and a small quantity of nitrogenous waste matter ABNORMAL CHARACTERISTICS OF FAECES Color 1. 2. 3. 4. Tarry, black stools: bleeding in the upper gastro intestinal tract Black color stools: melena, administration of iron or charcoal Clay color stool: obstruction to the flow of bile White color stool: presence of barium salts after barium tests Odor 1. Melena and dysentery: foul smell Frequency 1. Diarrhea: increased frequency 2. Constipation: decreased frequency Consistency and form 1. Watery stools: diarrhea 2. Rice water stools: cholera 3. Pea soup stools: typhoid fever Appearance 1. Fresh blood in large amounts: bleeding piles 2. Blood and mucus stool: amoebic or bacillary dysentery 3. Worms or worm segments in stools: parasitic cysts, ova or larvae GENERAL INSTRUCTIONS 1. Faecal specimens should be collected in the early stages of disease, preferably before the antibiotic treatment is given 2. Stool specimen should collect in a sterile container (using a scoop which is attached to the lid itself) 3. After collection, the lid should be immediately replaced tightly 4. After proper labelling, the collected stool should be handed over to the laboratory without delay, i.e. within 15 minutes ARTICLES A clean tray containing, 1. 2. 3. 4. 5. Appropriate specimen container (sterile) Spatula (clean for routine and sterile for culture) Bedpan or portable commode Gloves Waste paper PROCEDURE S NO 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 STEPS Explain the procedure to the patient Obtain lab request and container Provide privacy Arrange the articles at the bedside Provide bed pan if necessary and instruct the patient to defecate into clean dry bedpan or commode Instruct to not to contaminate specimen with stool Wear gloves Collect the stool specimen with clean spatula for routine stool examination and with sterile spatula for culture test Close it with lid tightly Remove gloves Wrap spatula in a waste paper and discard appropriately Label specimen container with patient name, hospital number ward name and date and time of collection Send to lab immediately, within 15 minutes Replace equipments Record procedure in nurse’s record with time and date RATIONALE To obtain co-operation For proper documentation and sending the specimen to lab It provides comfort Saves time and energy To avoid infections Fresh specimen accurate results provides more SPUTUM CULTURE Collection of coughed out sputum for culture studies to identify respiratory pathogens PURPOSES 1. To detect the micro organism, that causes respiratory tract infections 2. To treat with specific antibiotics CHARACTERISTICS OF SPUTUM Quantity: normally no sputum is expectorated but the amount of sputum coughed up in 24 hours varies with the disease Consistency: the sputum may be classified into various types according to the consistency and appearance. E.g. serous, frothy, mucoid, purulent, seropurulent and hemorrhagic Odor: normally the sputum is odorless, in case of respiratory tract infections the sputum will be foul smelling. Color: sputum consists of mucus may be colourless and translucent 1. 2. 3. 4. 5. 6. 7. Yellowish color: presence of pus Blackish sputum: excessive smoking Blood: hemoptysis Red and frothy sputum: fresh bleeding from lungs Greenish colour: bronchiectasis Brown colour: gangrenous condition of the lungs Rusty colour: pneumonia GENERAL INSTRUCTION 1. 2. 3. 4. Collect the sputum in the early morning. Do not use any antiseptic mouth washes prior to sputum collection. If sterile specimens are required- sterile bottle with cover is given to the patient. Collect at least 10ml of sputum for sputum culture test. ARTICLES A clean tray containing 1. 2. 3. 4. 5. Sterile Specimen container Sputum cup Tissue paper/ gauze pieces Clean gloves Kidney basin PROCEDURE S.NO 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 ACTION Check physician’s order Arrange all the articles Explain the client that the specimen, must be taken at morning before brushing teeth Ask the patient to sit erect in bed if possible Wash hands, put on gloves and wear mask if necessary Keep sterile specimen container ready for the sample and take a tissue paper in hand Remove lid of container and place with inner side facing upwards Instruct the patient to take deep breaths and then cough out deeply Explain the patient to spit the sputum into the sterile container without touching the side of it. Close the container without touching inside of it Provide client with tissue paper and a comfortable position Replace articles Wash hands Provide mouth care if patient needs it or encourage patient to carry out oral hygiene Document obtained specimen with name, age, sex, MRD number, date and time of collection and characteristics of the specimen and send specimen to lab RATIONALE To obtain secretions overnight accumulated Provide easy access for collection of specimen To prevent transmission of infection Prevents contamination It helps to loosens the secretions and obtain adequate specimen Prevents contamination of the specimen To clean the lips Documentation serves as an evidence SPUTUM CULTURE FOR AFB The reason AFB or TB culture is recommended is to confirm the diagnosis of TB. Secondly, the culture test can be followed by susceptibility testing, which is very useful, since many cases are now multi-drug resistant, and the drugs susceptibility results can be used to modify the treatment and make it more specific for the patient. PURPOSE OF TEST Positive AFB cultures identify the particular mycobacterium causing symptoms, and susceptibility testing on the identified organism gives the doctor information about how resistant it may be to treatment. GENERAL INSTRUCTION 1. It is preferably collect an early morning sputum specimen before brushing or rinsing the mouth. 2. Deeply coughed specimen, preferred. 3. Recommended screening procedure: 3 first morning specimens collected on 3 successive days. However, 3 samples collected at 8 hour intervals will be accepted. Have patient rinse mouth well with water to avoid contamination with food particles and mouth flora 4. Minimum volume 2ml TEST RESULT INTERPRETATION: Growth or No Growth with sensitivity pattern PROCEDURE: see the procedure of sputum culture COLLECTION OF BLOOD FOR ROUTINE TEST DEFINITION Obtaining blood sample by venipuncture for routine lab investigation PURPOSE 1. To detect the micro organisms 2. To treat the disease condition with correct antibiotics 3. To detect the right antibiotic to kill the particular micro organism GENERAL INSTRUCTIONS 1. Special kind of tubes with coloured top are using for blood sample collection 2. Colour coding of the top depends upon the kind of blood test. E.g. red indicates biochemistry, lavender or violet indicates pathology, blue indicates clotting studies ARTICLES A clean tray containing, 1. Mackintosh and towel 2. Clean gloves (optional) 3. Surgical spirit or betadine solution 4. Disposable syringe 5ml or 10ml with needles 5. Cotton swabs 6. Specimen container- test tube or bottle 7. Paper bag 8. Kidney tray 9. Tourniquet 10. Lab requisition form 11. Adhesive tapes PROCEDURE S.NO 1 2 3 4 5 STEPS Check the physician’s order Identify the patient Explain the procedure to patient Wash hands and put on clean gloves Select and examine the veins, visualize the vein, including the antecubital area, wrist, dorsum (back) of the hand and top of foot (if necessary). Choose the appropriate wide vien to be drawn by touching the skin over the vien in a circular motion. RATIONALE Ensures co-operation from patient To prevent infection and avoid getting blood in hands Select a vein that is visible, palpable and fixed to the surrounding tissues so that it does not roll away 6 7 8 9 10 11 12 13 14 15 16 17 Instruct the patient to extend his arm. Hold the arm straight at the elbow with fist clenched. Apply the tourniquet 5 to 15cms above the selected site with just sufficient pressure to obstruct venous flow Cleanse the skin with alcohol swabs (or betadine if alcohol is not available) in a circular motion; centre to periphery. Allow to dry it. Fix chosen vein with thumb and draw the skin taut immediately below the site before inserting the needle to stabilize the vein. Hold the syringe between the thumb and last three fingers with the bevel up and directly in line with the course of the vein. Insert the needle quickly and smoothly under the skin into the vein Obtain blood sample by gently pulling back on the plunger Release the tourniquet as soon as the specimen is obtained and ask the patient to open the fist Apply sterile gauze piece to puncture site without applying pressure and withdraw needle slowly along the line of vein Request patient to apply gentle but firm pressure to site for 2-4 minutes Remove the needle from the syringe as soon as possible after withdrawing blood, gently eject the blood sample into the appropriate container without forming bubbles in the test tube or bottle Invert the tube gently several times to mix blood with anticoagulant where applicable. For some tests blood is allowed to coagulate in the test tube Label specimen correctly and send to laboratory immediately with completed requisition forms Proper positioning visualization of veins increases the A tourniquet when applied increases venous pressure and makes the vein more prominent and easier to enter Cleansing the skin reduces number of micro organism the The vein may slip and move under the skin when the needle approaches its outer surface, especially in elderly, extreme thin patient and diabetic patient. Use minimal suction to prevent haemolysis of blood and collapse of vein Slow withdrawal of the needle is less painful and reduces trauma Firm pressure over puncture site prevents leakage of blood into surrounding tissues with subsequent hematoma development Gentle ejection of blood prevents haemolysis Gentle handling of specimen prevents risk of haemolysis Specimen should reach the laboratory with the minimum of delay for optimum reliability 18 19 20 21 Dispose the needle and syringe in appropriate containers If there is any spill, Clean all spills with sodium hypochlorite solution Record in the patient’s chart the procedure and the tests for which the sample was sent to the laboratory Replace the tray with the reusable articles in proper place Avoids possible spread of blood borne disease Avoids possible spread of blood borne diseases COLLECTION OF BLOOD FOR CULTURE TEST DEFINITION Collection of blood for culture to determine presence of micro organisms in the blood GENERAL INSTRUCTIONS 3. Blood culture should be taken before antibiotic treatment 4. For children, 2-5ml and neonate 1-2ml of blood is required for culture investigation 5. Blood should never taken from IV line 6. Special kind of culture bottles are using for blood sample collection 7. All blood culture bottles should be carefully examined for clarity of media, any medium showing turbidity or cloudiness should not be used. 8. Only disposable syringes and needles should be used for collection of blood 9. The top of the bottle must be carefully disinfected just before the bottle is inoculated 10. Blood should never be taken from an IV line or above the IV line 11. If blood culture bottles are available, blood should be immediately added to the culture medium 12. If blood culture bottles are not available, blood may be transported in a sterile tube containing a sterile anticoagulant solution 13. The amount of blood collected is 10ml for adult, 2 to 5 ml for children and 1 to 2 ml for infants and neonates ARTICLES A clean tray containing, 1. 2. 3. 4. 5. 6. 7. 8. Blood culture bottles Cotton swabs Spirit Syringe 10-20ml and needle Povidone iodine or betadine solution Sterile gloves Tourniquet Laboratory requisition forms PROCEDURE S.NO 1 STEPS Assess the physician’s order for blood culture investigation RATIONALE Obtains knowledge of samples to be collected and the reason for doing 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 Explain procedure to the patient and provide a comfortable position Wash hands. Don sterile gloves Apply tourniquet above the puncture site and palpate the venipuncture site Wipe the site with 70% alcohol in a circular manner from centre to periphery for approximately 5cm in diameter and allow to dry Cleanse the site again with betadine starting from centre in even widening circles. Allow the betadine to remain on the skin for at least one minute. Clean the site with 70% alcohol Puncture the site and draw 10ml of blood Remove the tourniquet once the blood is collected Remove the needle and apply pressure to the puncture site with dry cotton simultaneously Change the needle with a fresh needle before injecting the blood into the bottles Remove the metal cover on the cap of culture bottles and push 10ml of blood into the bottles be careful not to touch the sides of the bottle Mix the blood and culture media by shaking the bottle gently Discard the contaminated articles. Remove gloves Wash hands Fill the lab requisition form appropriately and label the bottles with patient’s name, identification number, date and time of collection Transfer the specimen to the lab immediately Record the procedure in the patient’s chart with date and time of collection Repeat the procedure within an interval of 30minutes to one hour as per the number of samples required for different puncture sites. culture Gains co-operation of the patient during the procedure Reduces transmission of micro organisms and maintains aseptic technique Restricts blood flow and promotes easy visibility of veins Avoids contamination and maintains a sterile field Restore circulation Stops bleeding from puncture site Maintains strict aseptic technique Prevents transmission of infection Communicates adequate information to members of health care professionals