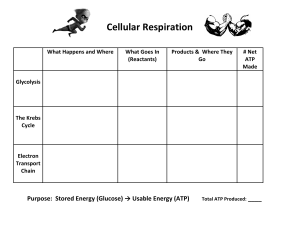
Cellular Respiration Investigation 6 AP Biology and AP STEM Biology Essential Question: What factors affect the rate of cellular respiration in multicellular organisms? Objectives: To learn how a respirometer system can be used to measure respiration rates in plant seeds or small invertebrates, such as insects or earthworms. To design and conduct an experiment to explore the effect of certain factors, including environmental variables on the rate of cellular respiration. To connect and apply concepts, including the relationship between cell structure and function (mitochondria); strategies for capture, storage, and use of free energy; diffusion of gases across cell membranes; and the physical laws pertaining to the properties and behaviors of gases. Background: In order for our bodies to function, we need fuel. This fuel comes from the foods we eat in the form of proteins, carbohydrates, and fats. Of these organic compounds, it is the carbohydrate glucose that is the most preferred energy source for our bodies. However, in order for this energy to be useful to us, it must first be converted to ATP (adenosine triphosphate), the common carrier of chemical energy in the cell. All body cells split glucose molecules to transfer the energy to ATP through a process called cellular respiration. This energy transfer occurs in two stages: glycolysis and aerobic respiration. In the first stage, a small amount of ATP is produced when glucose is broken down to pyruvate during glycolysis. In the second stage, the pyruvate either passes through aerobic respiration (producing a large amount of ATP) or (anaerobic) fermentation (producing a small amount of ATP). In the absence of oxygen, pyruvate is converted to either lactate or ethanol and carbon dioxide. The difference between aerobic respiration, and anaerobic respiration, is that in aerobic, ATP is produced by cells breaking down organic compounds with oxygen as the final electron acceptor. With anaerobic respiration, energy is liberated that does not require the presence of oxygen. 1 ATP provides the energy used for synthetic reactions, active transport, and all cell processes. Nearly all organisms’ cells respire and in eukaryotes, this occurs in the mitochondria. However, in bacteria and other prokaryotes, which do not have mitochondria, the conversion of NADH and pyruvate to ATP occurs in the cell membrane. The overall equation for the breakdown of glucose can be represented in the following way: Glucose + Oxygen Carbon Dioxide + Water + Energy (ATP) The energy-rich carbohydrates that undergo cellular respiration in plants and animals are produced in the chloroplasts of plants and algal cells. Photosynthesis uses carbon dioxide, water, and light energy to produce oxygen and carbohydrates. Cellular respiration uses carbohydrates and oxygen to produce carbon dioxide, water, and energy for the activities of the cell. Cellular respiration, then, is essentially the reverse of photosynthesis. It is the link between the energy captured by plant cells and the energy utilized by both plant and animal cells and is the means by which energy flows from the sun through living things. Glucose Metabolism Most cells use glucose as an energy source for producing ATP. The first stage of glucose metabolism is referred to as glycolysis, a process that occurs outside the mitochondria in the cytosol. Glycolysis is anaerobic, meaning that it does not require oxygen. Glucose is converted to two molecules of pyruvate with little breakdown; however, oxidation by removal of hydrogen atoms (e-+ H+) does occur, and the energy of oxidation is used to generate two molecules of ATP. A small amount of NADH is also produced. The fate of pyruvate, the end product of glycolysis, depends on the availability of oxygen. It can either be involved in fermentation or incorporated into the Krebs Cycle. 2 Under anaerobic conditions, fermentation occurs within the cytosol and pyruvate accepts the two molecules of hydrogen that were removed from glucose during glycolysis. Because only two molecules of hydrogen are available, fermentation yields only two ATP molecules. Pyruvate can be reduced in one of two ways. It can become lactate in fermentation, which is used by many bacteria and some small animal cells, or it can be reduced to alcohol with the release of carbon dioxide, which is known as alcoholic fermentation and is utilized by yeast and plant cells. Cellular Respiration If oxygen is available to the cell, cellular respiration occurs. This is an aerobic process that produces much more energy than fermentation. In this case, pyruvate is not reduced, but instead enters the mitochondria and is completely oxidized to carbon dioxide and water. The removal of hydrogen atoms (e-+ H+) is accompanied by the release of carbon dioxide, which diffuses out of the mitochondria and the cell; the hydrogen atoms are sent to an electron transport system located in the mitochondrial cristae, which are folds of the inner membrane. As electrons are passed from one carrier to the next in the cristae, energy is made available that is eventually used to generate ATP molecules. Oxygen, the final acceptor for these electrons, is reduced to water. Most of the ATP production in cells comes from this electron transport system. If glycolysis is followed by cellular respiration, then the overall reaction for glucose breakdown to carbon dioxide and water can be represented as: C6H12O6 + 6O2 36 ATP + 6CO2 + 6H2O The complete oxidation of glucose by cellular respiration yields a total of 36 ATP molecules, most of which are produced by the electron transport system. It is possible to calculate the ATP yield for the complete breakdown of glucose to carbon dioxide and water. Oxidative Respiration In the presence of oxygen, the pyruvate from glycolysis enters a mitochondrion, where a series of reactions called oxidative respiration occurs. This is the transition reaction between glycolysis and the Krebs Cycle. The electrons made available are passed to the electron transport system by either NAD+ or FAD. The reactions of oxidative respiration drive the production of large amounts of ATP. In order to prepare for the ATP-producing part of oxidative respiration, the pyruvate molecules from glycolysis are first converted from three- to two-carbon fragments. As this occurs, a carbon dioxide molecule and a hydride ion with high-energy electrons are removed from each pyruvate molecule. The carbon byproduct leaves the 3 mitochondrion and then the cell. The donation of hydride ions to an NAD + molecule forms NADH, which is later used in oxidative respiration. The remaining two-carbon fragment, an acetyl group, is attached to coenzyme A to form a compound acetyl-CoA. Acetyl-CoA is funneled into fat synthesis if the cell’s supply of ATP is plentiful, allowing high-energy electrons to be stored for later needs. If the cell needs ATP immediately, however, acetylCoA is directed to the next phase of oxidative respiration, which is the Krebs Cycle. The Krebs Cycle The Krebs Cycle is a repeating series of reactions that produces ATP, electron carriers, and carbon dioxide. The cycle begins when the two-carbon fragment of acetyl-CoA is attached to a four-carbon molecule found in mitochondria. This forms a six-carbon compound called citrate; it is for this reason the Krebs Cycle is also known as the citric acid cycle. During the cycle, oxidation occurs, and most of the hydrogen atoms are donated to NAD+, except for one instance where the hydrogen atoms are donated to FAD, another coenzyme of oxidation and reduction in cells. Some of the energy of oxidation is used immediately to form ATP by substrate-level phosphorylation, as in glycolysis. Since the Krebs Cycle occurs once for acetyl-CoA, it occurs twice for each glucose molecule. Electron Transport System Due to the transition reaction and the Krebs Cycle, the carbons of glucose are completely oxidized to six molecules of CO2. The hydrogen atoms (e- + H+) that were removed from the substrates of these metabolic pathways are donated by NADH and NADH2 to the electron transport system. This system is a series of carriers and enzymes located on the cristae of mitochondria. It is also known as the cytochrome system, since some of the electron carriers are cytochromes. Each electron transport chain passes highenergy electrons to proton-pumping membrane channels. As electrons are passed from one carrier to the next, oxidation occurs, and the energy released is used to form ATP molecules. (ATP leaves a mitochondrion through other protein channels, entering the cytosol where it can be used by other reactions). This process is sometimes known as oxidative phosphorylation because oxygen receives the energy-spent electrons from the chain. The enzyme cytochrome oxidase splits and reduces molecular oxygen (O2) to water: ½ O2 + 2e- + 2H+ H 2O The final acceptor for electrons during cellular respiration is oxygen. Substrate-Level Phosphorylation For every glucose molecule, four ATP are formed directly by substrate-level phosphorylation—two during glycolysis and one during each of the two turns of the Krebs Cycle. 4 Oxidative Phosphorylation For every glucose molecule, ten molecules of NADH and two molecules of FADH 2 take hydrogen atoms (e- + H+) to the electron transport system. For each molecule of NADH produced inside the mitochondria by the Krebs Cycle, three ATP are produced by the electron transport system, but for each FADH2 there are only two ATP. This is because FADH2 delivers its hydrogen to the transport system at a lower level, pumping only 4H+ instead of 6H+ into the intermembrane space per molecule of FADH 2. NADH generated outside the mitochondria during glycolysis requires a shuttle mechanism to transport the electrons that yield ATP, because NADH cannot cross the mitochondrial membrane. This shuttle, which consists of an organic molecule that can cross the outer membrane, allows electrons to be delivered to the electron transport system inside the mitochondria. The shuttle molecule crosses the outer membrane, accepts the electrons, and delivers them to the FAD molecule in the inner membrane. At this point, the FAD can produce two ATP molecules; therefore, the NADH produced in the cytosol results in the production of only two ATP rather than three. Respiration applies to two distinct but interrelated processes: the active acquisition of oxygen by an organism and the release of energy through the breakdown of organic compounds by metabolic chemical oxidation. Various factors, such as temperature change, will affect respiration, as well as the chemical oxidation of glucose. Oxygen consumption during respiration can then be measured as a change in gas volume by using a respirometer. In cold-blooded organisms, (poikilotherms) there is a link between the temperature of the environment and the organism’s metabolic rate. Reptiles are a common example of a cold-blooded organism with which most people are familiar. If you have ever seen a lizard or snake in the early morning when the air and ground are cool, you may have noticed how slowly they move. They move slowly when the environment is cold because they require heat from their surrounding to increase their internal temperature and metabolism. Once their internal body temperature has warmed, they can metabolize foods more quickly and produce the energy they need. Oxidative respiration is the process of metabolism where sugars are broken down. Under aerobic conditions, respiration yields ATP, carbon dioxide, and water. The Investigation: In this lab, we are interested in measuring the rate of respiration. In theory, we could measure: - the consumption - the production of carbon dioxide or water - the temperature rise (since this reaction releases 686 cals) As it turns out, we will measure the volume of oxygen consumed to study respiration. Organisms we will use It is difficult to find humans that are small enough to fit into little glass vials and submerge in pans of water. Fish and baby hamsters might be small enough, but they are too cute. Insects work well, but who wants to work with giant cockroaches? The solution: use PLANTS. PLANTS RESPIRE AND MAKE ATP – especially GERMINATING SEEDS. GERMINATING SEEDS RESPIRE. GERMINATING SEEDS MUST MAKE ATP, SINCE 5 THEY ARE TRYING TO GROW AND START OUT LIFE!! Germinating seeds do not photosynthesize; they respire stored organic compounds (just like you and me). Think of a germinating seed as a cooperative, tiny round respiring human being. How Do Our Respirometers Work? The carbon dioxide produced by the respiring seeds will be removed by KOH (potassium hydroxide) on a cotton ball and turned into solid potassium carbonate. As the seeds respire and remove oxygen from the inside of the vial, the air pressure inside the respirometer will fall. This pulls water down the little calibrated pipette toward the vial where the seeds are. We will read the water level in the pipette at 5-minute intervals, as the seeds inside consume oxygen. The reason we can do this is because of the general gas law, which you all remember fondly from CHEMISTRY: PV = nRT P = pressure of a gas V = volume of gas n = number of moles of gas R = the gas constant T = temperature in (oK) This law implies that if temperature and volume are held constant, pressure of a gas is directly proportional to the number of moles of the gas. The air pressure in the respirometer falls (sucking water in) because oxygen molecules are being consumed. Of course, conditions may change during the course of the experiment (from 8 to 9 am, temperature will rise and air pressure may change, too, depending on the weather) so we set up control respirometers containing glass beads instead of seeds. In Part 1 of the lab, you will be comparing respiration of germinating seeds (which have been soaked in water) vs. dormant (nongerminating) seeds (dry – out of the package). Which will show a faster respiration rate? You will also be measuring respiration rate (mls oxygen consumed/min). Guess at the respiration rate of germinating seeds. For comparison, a resting person respires at the rate of about 7.5 liters of air each minute; a mouse, .02 liters per minute; How fast does a seed respire? Make a guess – do you think it is one-tenth as fast as a person? one-one thousandth? or do seeds respire fast than we do? In Part 2 of the lab, you will have the opportunity to design and conduct your own experiment. 6 General Safety Precautions: You must wear safety goggles or glasses, aprons, and gloves during the investigation (s) because KOH is caustic. Follow your teacher’s instructions when using the hot glue gun to seal microrespirometers (if you are asked to make them). Do not work in the laboratory without your teacher’s supervision. Vocabulary: cellular respiration aerobic respiration anaerobic respiration glycolysis Kreb’s Cycle substrate-level phosphorylation oxidative phosphorylation Adenosine triphosphate glucose lactate pyruvate NADH FADH2 electron transport chain electron carrier final electron acceptor Ethanol carbon dioxide metabolism fermentation Pre-lab: Complete the lab bench activity (including the quiz). Print the quiz and put the results in your pre-lab questions section. http://www.phschool.com/science/biology_place/labbench/lab5/intro.html Pre-lab Questions: 1. Why is it necessary to correct the readings of the respirometers containing seeds with the readings taken from respirometers containing only glass beads? Your answer should ref to the concepts derived from the general gas law (PV = nRT). 2. What happens to the volume of the gas being measured (O2 consumption or CO2 production) when the temperature or pressure changes during the experiment? 3. If pressure and temperature remain constant, will the volume of gas in the respirometers increase or decrease? Explain. 4. Imagine you are given 25 germinating pea seeds that have been placed in boiling water for 5 minutes. You place these seeds in a respirometer and collect data. Predict the rate of oxygen consumption (i.e. cellular respiration) for tehse seeds and explain your reasons. 5. Imagine that you are asked to measure the rate of respiration for a 25 g reptile and a 25 g mammal at 10 degrees celcius. Predict how the results would compare, and justify your prediction. 6. Imagine that you are asked to repeat the reptile/mammal comparison of oxygen consumption, but at a temperature of 22 degrees celcius. Predict how these results would differ from the results at 10 degrees celcius, and explain your prediction in terms of the metabolism of the animals. 7. What difficulties would there be if you used a living green plant in this investigation instead of germinating seeds? 7 Materials: water bath tape (yellow masking) to act as a sling- sticky side up please 3 respirometers dye thermometer 3 weights ice stopwatch/timer (you may use your own) parafilm or vasiline (for sealing purposes) For repirometer assembly: absorbent cotton 15% KOH Eyedropper nonabsorbent cotton glass vial stopper with sealed 1-ml serological pipette. 1 L beaker Paper towels Procedure for respirometer assembly: Respirometers 1,2,3 are one set – they will be placed at 25oC. GOAL: Compare respiration in 20 germinating peas with 20 non-germinating dry peas and 20 glass beads. BUT, their volumes are different! So first part of procedure is all about getting their volumes equalized so they take up the same space in the vial – that means the amount of space in the vial for OXYGEN will be the same. VOLUME EQUALIZATION BETWEEN EXPERIMENTAL AND CONTROL SETUPS: 1. Estimating the density of non-germinating (dry) peas: Weigh 100 non-germinating peas (class set) using an electronic scale. Weight of 100 non-germinating peas = g 2. Take a 100 ml glass measuring cylinder. Add 50 mL water to it. Place the 100 nongerminating peas in the cylinder. Record the volume in the cylinder after adding peas = mL. Calculate the volume of the peas = ________________ (Final vol of cylinder – initial vol) Calculate the density of the peas = _____________ (mass/volume of peas) 8 3. Take 20 germinating peas and place them on a paper towel. Fill a smaller graduated cylinder to the 20 mL mark with tap water ACCURATELY. Add the germinating peas to the tube and record the FINAL volume below: Volume of germinating peas = (FINAL VOL mL – 20 mL ) = mL Place the germinating seeds on a paper towel and label them. 4. Re-fill the graduated cylinder to the 20 mL mark. Add glass beads (one of your controls) to the tube until the volume in this tube is equal to the FINAL volume in the germinating pea tube in step 3: Number of glass beads needed = Volume of glass beads = (FINAL VOL mL – 20 mL) = be the same as in step 3! If not, you are NOT thinking/reading). mL (this should Place the glass beads separately on a paper towel and label them. 5. Use the calculated density of the non-germinating seeds (step 2) and figure out the needed mass of (dry peas and glass beads) that you need to amount to the same volume as germinating peas. Known density = (from step 2) Desired volume = (as calculated in step 3) Therefore, the mass of the (dry peas) needs to be = gm (volume x density). 6. Count out 20 of your wet, non-germinating pea seeds and place them on a weighing boat and record weight. Add glass beads until you record the total mass as in step 5 above (this way the volume in the dry peas vial and germinating peas vial will be the same). a. Tips: i. Look back at your calculated mass in step 1 for 100 peas. ii. Estimate what the approximate mass would be for 20 peas based on this number. iii. You can search for dry peas that will give you your desired weight, but you can still only use 20 peas. 20 dry non-germinating peas weight = Did you have to add any glass beads? How many? (Some groups may not end up added glass beads): _____________ 9 SETTING UP RESPIROMETERS: 7. To assemble respirometers, obtain 3 glass vials for EACH run at a particular temperature, each with an attached stopper and pipette. Number the vials 1 through 3. Place a small wad of absorbent cotton (read label) in the bottom of each vial and, using a dropper, saturate the cotton with 15% KOH (potassium hydroxide; corrosive – wear goggles, gloves). This means the cotton ball needs to just appear wet. It is important that the same amount of KOH and cotton be used for each respirometer. DO NOT GET KOH on the sides of the glass vial – your respirometer. 8. Place a small wad of dry, nonabsorbent cotton on top of the saturated cotton. 9. Place the contents on the paper towels labeled from 1 – 3 in the correct Respirometers over the cotton. Check Table 1 if you have questions about what to place in them. Insert the stopper with the calibrated pipette. Seal the set-up with parafilm or vasiline – very, very important to get a good seal. Place a weighted collar on each end of the vial. Several washers around the pipette make good weights. 10. Prepare the room-temperature bath (approx. 25 degrees Celsius) 11. Make a sling of masking tape attached to each side of the water baths. This will hold the ends of the pipettes out of the water during an equilibration period of 7 minutes. Place the respirometers on the sling and time this equilibrium period. 12. Add a small drop of colored dye to the end tip of the respirometer. This will help you to see the oxygen as it moves up the respirometer. Place respirometer glass vials 1,2,3 in room temp. bath very, very gently. A little water should enter the pipettes and then stop. If the water continues to enter the pipette, check for leaks in the respirometer. Look for a small bubble that should be on the top end of your pipette. Finding the bubble is the most crucial part of this setup! You have 3 min. to find the bubbles. RECORDING DATA AND MAINTAINING TEMP: 13. After you have allowed the respirometers to equilibrate for these 3 minutes, record the initial position of the water in each pipette to the nearest 0.01mL (time 0). Check the temperature in the bath and record. Record the water level in the pipettes every 2 minutes for 14 minutes. Student monitoring temperature should keep the water baths at the desired temp throughout the 14 min. of recording ‘bubble position’. Bubble position indicates volume of oxygen consumed by peas. 14. After completing SETUP 1 data recording, graph your data. 10 Table 1: Measurement of O2 Consumption by Soaked and Dry Pea Seeds at Room Temperature (250C) Using Volumetric Methods. Temp (oC) Room Time (min) Glass Beads Alone Reading Diff* at time X Germinating Peas Reading at time X Diff* Corrected Diff. ^ Dry Peas (and glass beads) Reading at time X Diff* Corrected Diff ^ Initial 0 0-2 2- 4 4 -6 6-8 8-10 10-12 12-14 * Difference = (initial reading at time 0) - (reading at time X) ^ Corrected Difference = (Reading for the pea seed at time X) - (Reading for the beads at time X) Table 2: Rate of Oxygen Consumption per minute Condition Germinating peas (Room Temp.) Show Calculations Here Rate in ml.O2 / min Dry Peas (Room Temp.) 11 Analysis Questions: 1. At what temperature was the rate of carbon dioxide production highest in part A? How does this relate to the internal body temperature of warm-blooded organisms? 2. During aerobic respiration, glucose is broken down to form several end products. Which end products contain the carbon atoms from glucose? The hydrogen atoms from glucose? The oxygen atoms from glucose? The energy stored in the glucose molecules? 3. What is fermentation? What are the two types of fermentation? What organisms use fermentation? 4. Matching: Write the letter of the pathway that best fits each of the following processes. Pathway a. Glycolysis b. Krebs Cycle c. Electron Transport System Process ________1. Carbon dioxide is given off ________2. Water is formed ________3. PGAL ________4. NADH becomes NAD+ ________5. Oxidative phosphorylation ________6. Cytochrome carriers ________7. Pyruvate ________8. FAD becomes FADH2 5. Name some other biological processes that are affected by temperature. (Remember, not all biological processes are about humans, or even animals) 6. According to your graph, what happens to the rate of carbon dioxide production by germinating peas over time? What does this indicate to you? 7. Which of the two pea types, germinating or non-germinating, consumes the most oxygen? Why? 8. What was the effect of temperature on pea respiration? 9. Was this what you expected? Explain. 10. Predict what might happen if you were to carry out the above procedure on germinating, and non-germinating peas in a 50° C environment. 12 Designing and Conducting Your Own Investigation: Now that you have learned how to measure the rate of cellular respiration in germinating seeds, you have a tool for exploring questions on your own. 1. Think about the process of cellular respiration. When does it occur? Are there any situations when living cells are not respiring? Why might some living cells respire more than others? Are there differences between major groups of organisms and how they respire? Does the temperature of germinating seeds affect the rate of cellular respiration? Do germinating seeds just starting to germinate consume oxygen at a greater rate than seeds that have been germinating for several days (age dependence)? 2. Design an experiment to investigate one of your own questions about cellular respiration or of the questions above. When identifying your design, be sure to address the following: What is the essential question being addressed? What assumptions are made about the questions being addressed? Can those assumptions be verified? Will the measurements you choose to make provide the necessary data to answer the question under study? What are possible sources of error in the experiment? 3. Make a hypothesis, which should include a prediction about the effect of the factor you chose to investigate on the rate of cellular respiration. 4. Conduct your experiment and record data and any answers to your questions. 5. Graph the results to show the effect of the factors you investigated on the rate of cellular respiration. 6. Calculate the rate of cellular respiration. 7. Perform statistical analysis of your data, comparing results of the experimental variable to the control. You should at least express the uncertainty of your measurements with error bars. Conclusion: Summarize the parts of the lab (what you did and what the findings were). Remember to address your hypotheses and always use your data to support your statements. 13