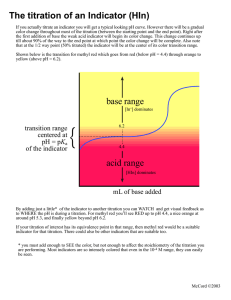
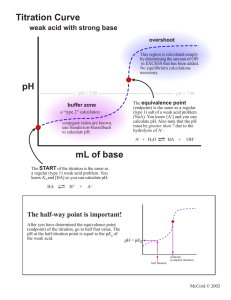
CHEMISTRY LAB ASSIGNMENT BY SAKTHIVEL SIR Why other acids rather than dil H2SO4 can not be used in mohr’s salt titration ? HCl and HNO3 are not used in the titration of Mohr's salt in place of sulfuric acid because: HCl is a mild reducing agent and it reacts with some of the indicator that is KMnO4 solution so the final value of the indicator used come to be more than the actual value disturbing the experiment. HNO3 is also a powerful oxidizing agent along with KMnO4 so it also plays its role in oxidation which causes a decline in the value of KMNo4 used. As dilute sulfuric acid is ideal for redox titration because it is neither an oxidizing agent and nor a reducing agent. • Potassium Permanganate is a flexible and potent oxidant which can be used by overt or indirect titration to classify many compounds. A special value of Potassium Permanganate is that it acts as an indicator of its own. Permanganate titration must be carried out in a heavy acid solution. • It is reduced to brown coloured Mn2+ ion (in acidic media) at the end point when used in redox titration, and the colour transition at the end point can be quickly observed. Since the function of the indicator is to detect endpoints, a separate indicator for permanganate titration is not needed. What property of the liquid causes viscosity? • Cohesive forces between like molecules are responsible for a liquid’s viscosity (resistance to flow) and surface tension (elasticity of a liquid surface).