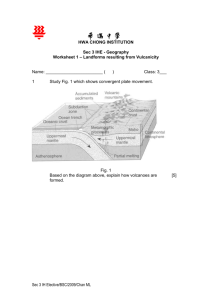
Name : Vinodaarshini Vigneswaran Year : 12 Subject : Earth and Environmental Science Yr. 12 Teacher’s name : Ms. Urbaniak Stratovolcano- pyroclastic flow investigation Contents Page number Introduction 3-5 Plan and designing 6-8 Conducting 9-18 Processing 19-29 Evaluation 30 Bibliography 31 2 Stratovolcano- pyroclastic flow investigation 1.0 Introduction Analyzing the impacts of geohazards, particularly the volcanic eruptions, by measuring the distance travelled by the pyroclastic flow are the basis of this experiment. Geohazards means the risk of damage caused by a geological process. Sometimes hazards are not obvious until pointed out. For example, we wash our hands to avoid biohazards such as viruses and bacteria even though they are too small to see. We are told that some foods are poisonous (a biohazard) so we don’t eat them. All geological hazards (geohazards) could be considered inactive until they are triggered. When the hazard occurs, it may then be called an event, accident, emergency, incident, or disaster. The study and monitoring of geohazards helps us to better prepare ourselves and respond to these geological events when they do occur. Geohazards can be small features that have an impact only on their local area such as a small landslide that partially blocks a road or track through to large earthquakes that affect entire cities. They can also be very large events that have a widespread impact such as large tsunami. Volcanoes are a type of geohazard. Volcanic eruptions may involve either the quiet or explosive ejection of lava, ash, and gases, as well as other associated phenomena, commonly pyroclastic flows and lahars (also called mudflows). All these events constitute hazards. The greatest hazard is associated with stratovolcanoes found near subduction zones (figure 1.1). These volcanoes may erupt violently at reasonable frequent intervals through geologic time. During eruption, large ash columns may be produced, and winds will distribute volcanic ash and larger fragments from these to great distances beyond the volcano, causing nearby towns and cities to be smothered in ash. Figure 1.1 The picture shows the Eruption of Mt Ruapehu in July 1997. Photo taken from Ohakune. Closer to the volcano, townships located near river valleys are in great danger from lahars or mudflows which are mixtures of ice, crater lake waters, ground water or rain water, and debris from the flanks of the volcano. Lahars travel down existing river valleys at high speeds pushing the river water ahead of them and causing extensive flooding and silting of river channels - which may lead to further flooding after the eruption. Lahars possibly account for the greatest loss of life. The most violent eruptions may produce hot pyroclastic flows which are turbulent mixtures of searing hot gases and tephra which move down the slopes of a volcano away from the vent. 3 Stratovolcano- pyroclastic flow investigation Pyroclastic flows are giant clouds of hot gases and ash particles formed by collapse of the buoyant ash column. They travel at tremendous speeds, too fast for anything or anyone to escape, and because they are so hot they incinerate everything in their path. These are the most fearsome events. Volcán de Fuego (Spanish for "Volcano of Fire") is one of the most active volcanoes in the world and is located 44 kilometers from Guatemala City. It is a stratovolcano (figure 1.2) that has had more than 60 eruptions since 1524, including a major eruption in 1974 which produced pyroclastic flows that destroyed the region's winter harvest and ashfall that covered nearby cities. The 3 June eruption is one of the deadliest in the country, including the Santa María eruption of 1902 and the Santiaguito dome collapse of 1929, which killed hundreds. The most eruptive phase began in 2002 and produced an explosive eruption in 2012 that forced 33,000 people to evacuate but had no reported deaths. The population around the volcano is estimated to be 54,000 within 10 kilometers and more than one million within 30 kilometers . Many of these volcanoes are characterized by magmas of intermediate (or andesitic) bulk composition (55–70 wt % SiO2) with H2O contents ranging from 3 to 6 wt % and typically display both dome‐building and explosive activity during the same eruption. Some examples include the 2010 eruption of Gunung Merapi and the 2014 eruption of Gunung Kelud, both located in Java, Indonesia, and both having a bulk magma composition of basaltic andesite (55–60 wt % SiO2). Both of these eruptions were characterized by the collapse of viscous lava domes, along with explosive events, all generating pyroclastic density currents (PDCs). Figure 1.2 The picture shows the Eruption of Volcan De Fuego in January 25, 2017. Photo taken from Climate in Guatemala. 4 Stratovolcano- pyroclastic flow investigation 1.1 .1 Purpose To analyze the impacts of geohazards, particularly the volcanic eruptions, by measuring the distance travelled by the pyroclastic flow. 1.2 .2 Aim To investigate the effects of pyroclastic flow from the stratovolcano eruptions on humans, infrastructure and the environment 5 Stratovolcano- pyroclastic flow investigation Plan and designing- Stratovolcano 2.1 Investigation set up Figure 2.1.1: Diagram of Stratovolcano Investigation Set-up PH probe, used to measure the PH level inside and outside of the bag. Bread, represents the crust nearby the stratovolcano. Popsicle sticks, representing the houses (2cm apart from each other). Glass rectangular tray, used to hold the set-up. Plants, representing the environment near the stratovolcano. Bi-carb, representing the SiO2 content in magma chamber. Plastic bottle, representing the stratovolcano. Zip-lock bag, representing the magma chamber and holds the 200ml of vinegar and bi-carb. Figure 2.1.1: The above diagram shows the set-up of the pyroclastic flow- stratovolcano investigation. It is a representation of a stratovolcano. The bowl which is 13.5cm tall and 21cm wide is used to hold the set-up of the stratovolcano. There are three different experiment with different amount of Bi-carb, representing the SiO2 level in the magma. The various amount of Bi-carb in the 200ml of vinegar had a reaction which represents the pyroclastic flow from the stratovolcano. However, the popsicle stick represents the houses and plants represent the environment. The popsicle sticks are arranged 2cm apart from each other to measure the pyroclastic flow accurately. The food coloring is used to show the direction of the lava flowed. The angle (30) represents the angle of elevation and the bread around it acts as the slope of a stratovolcano. The Ph and the time taken for the vinegar to travel was observed throughout the experiment. 6 Stratovolcano- pyroclastic flow investigation 2.1.2 Table of Variables Figure 2.1.2: Table of Variables from the observations taken during the stratovolcano experiment Constant (The same) • • Ratio (Volcanic Eruption Index) of Bi-carb – vinegar The amount of vinegar Independent (changed) • The amount of bi-carb Dependent (measured) • • • • • • The effects (displacement and distance of lava) The time (seconds) The Ph of the lava inside and outside of the bag The temperature of the lava inside and outside of the bag The pulse rate of the pyroclastic flow The pressure inside of the bag. Figure 2.1.2: The above table is providing the variables (constant, independent and dependent) of the effects on infrastructure and humans by pyroclastic flow from stratovolcano investigation. The ratio (Volcanic Eruption Index) of Bi-carb - vinegar was kept the same throughout the investigation. However, The amount of bi-carb is the prominent change in this investigation. The effects (displacement and distance of lava), the time (seconds), the Ph of the lava inside and outside of the bag, the temperature of the lava inside and outside of the bag, the pulse rate of the pyroclastic flow and the pressure inside of the bag were measured throughout this investigation. 2.1.3 Materials and Equipment Figure 2.1.3: Materials and Equipment for the effects on infrastructure and humans by pyroclastic flow from the stratovolcano experiment • • • • • • • • 200ml of Vinegar Bi-carb Food coloring Popsicle stick Zip lock bag Plastic water bottle A measuring cup A temperature probe • • • • • • • A Ph probe Timer Ruler Phone Playdough Plastic plants Rectangular glass tray 7 Stratovolcano- pyroclastic flow investigation 2.1.4 Method and Procedure Figure 2.1.4: Method and procedure from the Stratovolcano- pyroclastic flow experiment 1. Prepare the materials and equipment required for the experiment. 2. Set-up the investigation according to the set-ups in figure 2.1.1. Conduct the investigation in groups of three/four. 3. Spread butter on the bread which is representing the subduction plate. Butter is used to hold the bi-carb on the bread. 4. After this, put 1 tsp (8g of Bi-carb) on the bread and ensure it is accurate before subducting it into the zip lock bag containing vinegar. 5. Once this is prepared, the testing takes place. While the testing takes place, the pH and temperature should be monitored at all time. 6. Altogether there will be three experiments with three tests each. 7. Ensure there’s enough time to conduct three investigations in the time provided. 8. As the investigation is conducted, team members are to observe and take pictures of the progress. If there’s no change in observation, add pressure to the zip lock bag. 9. At this point of time, the change in temperature and PH is observed. 10. Observe the pulse rate of the lava from the stratovolcano as the reaction of the vinegar and bi-carb takes place in the experiment. 11. Repeat the above steps for each testing and experiment. 12. Jot down the observations in a table of observation and measurements. 13. Clean up your place and return the materials and equipment to the place it was retrieved from. 2.1.5 Hypothesis As the amount of Bi-carb increases, the distance travelled by the eruption increases, as the time progresses. 8 Stratovolcano- pyroclastic flow investigation Conducting (Stratovolcano- pyroclastic flow Investigation) 3.1 Table of Observations and Measurements Figure 3.1.1: Table of Observations and Measurements of the Ph and the time taken for experiment 1 (1tsp Bi-carb: 200ml vinegar). Test 1 Test 2 Test 3 9 Stratovolcano- pyroclastic flow investigation Figure 3.1.1: The above figure shows the measurements of pH inside and outside of the zip lock bag as the time progressed. The pH inside the bag is observed to increase gradually and the pH outside the bag is observed to stay constant throughout the experiment. This could be due to the lesser reaction rate between the vinegar and bi-carb ratio. From these three testing of the 1 st experiment, test 1.3 was the most successful one. As shown in figure 3.1.1, the right amount of pressure was applied, and the time taken for the eruption to occur was faster than the other tests. This could be due to the increase in amount of Bicarb reacting with the vinegar at a faster rate. Figure 3.1.1 This picture shows the set-up of experiment 3 after eruption. Lava Bomb Due to the fast eruption in test 3, the solution of vinegar and bi-carb reached 12cm away from the impact site which means it travelled approximately 12km from the stratovolcano. As shown in figure 3.1.2, a lava bomb is observed 16 cm from the impact site. Lava bombs are formed when a volcano ejects viscous fragments of lava during an eruption. They cool into solid fragments before they reach the ground. 12 cm from impact site Figure 3.1.2 This picture shows the patch of blue coloured solution (lava) soaked into breads (crust). 10 Stratovolcano- pyroclastic flow investigation Figure 3.1.2: Table of Observations and Measurements of the Ph and the time taken for experiment 2 (2 tsp Bi-carb: 200ml vinegar). Test 2 Test 1 Test 3 Figure 3.1.2: The above table of measurements show the pH level inside and outside of the bag in the 2nd experiment. The pH level inside the bag is observed to increase gradually in three of the tests whereas the pH level outside the bag is observed to stay constant. These results show how the pH level inside the bag is increasing as the reaction of vinegar and bi-carb takes place. There were a few observations jotted down for each of the tests. 11 Stratovolcano- pyroclastic flow investigation Test 2.1 -Displacement of pyroclastic flow : 14cm from impact site Test 2.2 -Displacement of pyroclastic flow : 4cm from impact site Point of eruption for all three tests Test 2.3- Displacement of pyroclastic flow : 4cm from impact site Figure 3.1.3 The above pictures shows the point of eruption for three different tests. The eruption rate is observed. Effect on houses (infrastructure) Figure 3.1.4 This picture shows the effect the pyroclastic flow has on the infrastructure. Figure 3.1.4 shows the effect of the pyroclastic flow on the houses which were arranged 2cm apart from each other from the stratovolcano. Volcanoes are a geohazard and it does have an effect on the infrastructures, houses and environment. . Figure 3.1.1 shows the points of eruption of three of the tests in the 2nd experiment. As per observation, the displacement of the lava from the impact site varied for all three of them. This could be due to the different amount of pressure applied to the zip-lock bag (magma chamber of stratovolcano). The more pressure applied, the more the distance of the pyroclastic flow travelled over time. Another factor which might have effected the displacement is the leveling of bread surrounding the stratovolcano (plastic bottle). The higher the level of bread near the stratovolcano, the lesser the pyroclastic flow travels from the impact site. However, the displacement of the pyroclastic flow in test 2.1 is 14cm from impact site, test 2.2 and 2.3 is 4cm from the impact site. 12 Stratovolcano- pyroclastic flow investigation Figure 3.1.3: Table of Observations and Measurements of the Ph and the time taken for experiment 3 (3 tsp Bi-carb: 200ml vinegar). Test 2 Test 1 Test 3 Figure 3.1.3: The above table of measurements show the pH level inside and outside of the bag in the 3rd experiment. The pH level inside the bag is observed to increase and decrease gradually in three of the tests whereas the pH level outside the bag is observed to stay constant. These results show how the pH level inside the bag is increasing as the reaction of vinegar and bi-carb takes place. There were a few observations jotted down for each of the tests. 13 Stratovolcano- pyroclastic flow investigation Test 3.2- Displacement of pyroclastic flow : 4cm from impact site Figure 3.1.5 This picture shows the pyroclastic flow soaking to the bottom of the crust. Figure 3.1.5. shows the pyroclastic flow soaking into the bread crust as soon as the eruption occurs. As from previous observations from different experiments, the main reason of the inaccurate result is that the pressure applied to the zip lock bag (magma chamber) is inconsistent. However, the pyroclastic flow is observed to not have much impact after 4cm from impact site because bread wasn’t arranged uniformly, its in an angular instead. Most of the bottom of the breads absorbed most of the liquid. The solution in the bag was observed to be close to none as all of the solution erupted outside. Test 3.3- Displacement of pyroclastic flow : 6cm from impact site Figure 3.1.5 This picture shows the pyroclastic flow backfiring from the eruption. As for figure 3.1.5, the eruption was observed to go up to a height of 8cm and then flowed to the crust. The vertical eruption could be due to the massive amount of pressure being released from the magma chamber as the bi-carb soda bread slab was subducted into the vinegar solution. The solution was observed to backfire and soak through the bread where the stratovolcano was located. However, 6 cm of the houses were destroyed as volcanoes are a geohazard. The bread in the zip lock bag stayed intact whereas the vinegar became saturated as there weren’t any reaction occurring at this point of time. The dissection of the experiment took place as the bread was observed to soak 6cm in depth near the stratovolcano. 14 Stratovolcano- pyroclastic flow investigation Figure 3.1.4: Table of Observations and Measurements of the Temperature and the time taken for 3 experiments. Experiment 1 ent 1 Experiment 2 Experiment 3 Figure 3.1.4: The above table of measurements show the average temperature for each of the experiments as the time progressed. For all these three experiments, the temperature is observed to decrease gradually as the time increased. This is due to the endothermic reaction of the vinegar and bi-carb soda. The release of temperature from the reaction allows the temperature inside the bag to decrease. However, the temperature remained constant when the reaction stopped occurring. 15 Stratovolcano- pyroclastic flow investigation 3.2 A Flowchart Figure 3.2.1: Diagram of the process through the stratovolcano- pyroclastic flow investigation To begin, we need to make sure we have all the materials for the investigation. The vinegar will be constant (200ml), but the Bi-carb quantity varies from 8g to 16g and 24g. Then arrange the popsicle 2cm apart from each other. The vinegar is contained in a zip lock bag. Finally, observe the pH of the lava before and after the investigation is conducted. Observe if there’s any differences in the pH level inside and the outside the zip lock bag. Record all the observations and measurements in a table, ready to be finalized in a report. Arrange the materials and equipment according to figure 2.1.1. make sure the plastic bottle makes an angle of 30 to the horizontal surface. The amount of Bi-carb varies from 1tsp to 3tsp. Follow the steps given in figure 2.1.4. Repeat it 3 times. Then, observe the experiment while conducting, the time is measured as the vinegar and bi-carb solution travels across the distance. From this 16 can be information, the displacement of the lava determined. Make sure to take pictures of the experiment. Stratovolcano- pyroclastic flow investigation 3.3 A Diagram of Changes and Alterations Figure 3.3.1: Diagram of Changes and Alterations in the Stratovolcanopyroclastic flow Investigation Part A: Adding playdough as a barrier to decrease the area of the rectangular glass tray. Part B: Shortened the height of the stratovolcano. 17 Stratovolcano- pyroclastic flow investigation Part C: Added a magma chamber using zip lock bag. Figure 3.3.1: The above diagrams show the changes and alterations made over the duration of testing. Part A shows the adding of playdough to the experiment to decrease the surface area of the rectangular glass tray. Part b shows the decrease of height of the stratovolcano for the experiments. Finally, part c shows the adding of magma chamber underneath the stratovolcano. The diagram in part A shows the playdough barrier added to the experiment. The group was thinking how to decrease the surface area of the glass rectangular tray and figure out a solution. The playdough barrier was kept only for the testing before the actual experiment took place as the group figured out new solutions to the problem. The playdough acting as a barrier was holding up the stratovolcano in place and not allowing the solution to leak through the crust where the houses and the plants were placed. However, part b showing the shortening of the stratovolcano’s height as it was a disruption and the group weren’t getting any results. The slope of the stratovolcano was kept the same, but the height of the stratovolcano was decreased to get better results. After the height of the plastic bottle (representing the stratovolcano) was shortened, the eruption of the pyroclastic flow was observed more clearly, and the results were close to accuracy. The slope of the plastic bottle allowed the pyroclastic flow to go into the bread (representing the crust). Finally, the diagram in part c shows the zip lock bag (representing the magma chamber) being added to the experiment as the group thought it was a good idea to have a subducting bi- carb soda slab into the magma chamber containing vinegar. This made the whole experiment to go smoothly and accurate results of the eruption of the pyroclastic flow was obtained. 18 Stratovolcano- pyroclastic flow investigation Processing (Stratovolcano Investigation) 4.1 Calculations Diagram 4.1.1 Calculations to find the velocity of the pyroclastic eruption from the stratovolcano Displacement Formula: 𝑽𝒆𝒍𝒐𝒄𝒊𝒕𝒚 = 𝑫𝒊𝒔𝒑𝒍𝒂𝒄𝒆𝒎𝒆𝒏𝒕 𝑻𝒊𝒎𝒆 Bi-Carb Pyroclastic flow vv Vinegar Experiments conducted Time taken for the pyroclastic flow travelled (s) Test 1.1 Displacement from the Stratovolcano (cm) 4cm Test 1.2 4cm 7.7s 𝑉𝑒𝑙𝑜𝑐𝑖𝑡𝑦 = Test 1.3 12cm 11.47s 𝑉𝑒𝑙𝑜𝑐𝑖𝑡𝑦 = 12𝑐𝑚 =1.046cm/s 11.47𝑠 Test 2.1 14cm 12.52s 𝑉𝑒𝑙𝑜𝑐𝑖𝑡𝑦 = 14𝑐𝑚 =1.118cm/s 12.52𝑠 Test 2.2 14cm 13.96s 𝑉𝑒𝑙𝑜𝑐𝑖𝑡𝑦 = Test 2.3 4cm 9.16s 𝑉𝑒𝑙𝑜𝑐𝑖𝑡𝑦 = 4𝑐𝑚 =0.437cm/s 9.16𝑠 Test 3.1 6cm 7.92s 𝑉𝑒𝑙𝑜𝑐𝑖𝑡𝑦 = 6𝑐𝑚 =0.758cm/s 7.92𝑠 Test 3.2 4cm 7.93s 𝑉𝑒𝑙𝑜𝑐𝑖𝑡𝑦 = 4𝑐𝑚 =0.504cm/s 7.93𝑠 Test 3.3 6cm 6.91s 𝑉𝑒𝑙𝑜𝑐𝑖𝑡𝑦 = 9.2s Velocity of the pyroclastic flow (cm/s) 𝑉𝑒𝑙𝑜𝑐𝑖𝑡𝑦 = 4𝑐𝑚 9.2𝑠 4𝑐𝑚 7.7𝑠 = 0.435cm/s = 0.519cm/s 14𝑐𝑚 13.96𝑠 6𝑐𝑚 6.91𝑠 =0.429cm/s =0.868cm/s 19 Stratovolcano- pyroclastic flow investigation Diagram 4.1.1: The above diagram shows the calculation of the velocity of pyroclastic eruption from the stratovolcano. As per observation, the displacement from the stratovolcano (cm) increases and then decreases. The houses are placed 2cm apart from each other to ensure an approximate result. The higher the displacement from the stratovolcano the longer the time taken for the pyroclastic lava to travel. The velocity is calculated by using the above formula. Velocity is directly proportional to the displacement of pyroclastic flow from stratovolcano and inversely proportional to the time taken for the pyroclastic flow to travel to the houses, infrastructure and environment. From the above results, the velocity increases, decreases and increases, over the period of time. Diagram 4.1.2 Calculations to find the pressure of the vinegar inside the bag during the eruption Formula: 𝑵𝒖𝒎𝒃𝒆𝒓 𝒐𝒇 𝑴𝒐𝒍𝒆𝒔 × 𝑮𝒂𝒔 𝒄𝒐𝒏𝒔𝒕𝒂𝒏𝒕 × 𝑻𝒆𝒎𝒑𝒆𝒓𝒂𝒕𝒖𝒓𝒆 𝑷𝒓𝒆𝒔𝒔𝒖𝒓𝒆 = 𝑽𝒐𝒍𝒖𝒎𝒆 Constant values: Number of Moles: 4.165 mole in 200ml of Vinegar Gas Constant: 8.314 J/Mol 20 Stratovolcano- pyroclastic flow investigation Experiments conducted Time when the eruption happened (seconds) Temperature when the eruption happened (C) Pressure inside the zip lock bag when eruption happened (kPa) 0s 1.1 1.2 1.3 2.1 2.2 2.3 3.1 3.2 3.3 5s 3s 3s 2.2s 8s 7s 3s 2.3s 2.3s 18.5C 18.5C 18.5C 19C 18C 17C 17C 17.5C 16.5C 5s 10s 30s 20.18 20.18 20.05 20.15 20.08 20.11 20.08 20.04 20.18 20.22 20.15 20.08 20.08 20.118 20.15 20.15 20.15 20.08 19.98 20.04 20.15 20.08 20.08 19.94 19.94 19.98 20.04 19.87 19.94 19.87 19.87 19.87 20.049 20.09 19.98 19.87 Diagram 4.1.2: The above diagram shows the different experiments conducted, the time when the eruption happened, temperature when the eruption happened and the pressure inside the zip lock bag when eruption happened. The time for eruption in experiment 2.2 is 8s. This could be due to the less pressure applied to the zip lock bag- therefore slowing down the vinegar-Bicarb reaction. However, the time taken for the eruptions to occur in all the tests were close to each other. The temperature increases and decreases when the eruptions happened. As per observation, the pressure inside the bag is consistent throughout the experiment. The time is selected according to the change in temperature. The above results only shows pressure inside bag as when the volcano erupts, the pressure is released. 21 Stratovolcano- pyroclastic flow investigation 4.2 Table of averages Diagram 4.2.1 Table of averages for the displacement of the lava from the stratovolcano. Tests Time (seconds) 1.1 1.2 1.3 2.1 2.2 2.3 3.1 3.2 3.3 9.2s 7.7s 11.47s 12.52s 13.96s 9.16s 7.92s 7.93s 6.91s Displacement of the pyroclastic flow from the stratovolcano (cm) 4cm 4cm 12cm 14cm 14cm 4cm 6cm 4cm 6cm Average Displacement of the pyroclastic flow from the stratovolcano (cm) = 7.56 cm Diagram 4.2.1: The above table shows the average displacement measurements from each of the testing in each experiment. The displacement of the pyroclastic flow is observed to increase and decrease gradually, and the time progressed. The time of the eruption for each the test is jotted on the table above. The average displacement of the pyroclastic flow was 7.56cm. Diagram 4.2.2 Table of averages for the pressure inside the zip lock bag. Tests 1.1 1.2 1.3 2.1 2.2 2.3 3.1 3.2 3.3 Time (seconds) 5s 3s 3s 2.2s 8s 7s 3s 2.3s 2.3s Average pressure inside the bag (kPa) 20.10 kPa 20.12 kPa 20.13 kPa 20.08 kPa 20.08 kPa 19.99 kPa 19.97 kPa 20.00 kPa 19.997 kPa 22 Stratovolcano- pyroclastic flow investigation Diagram 4.2.2: The above diagram shows the average pressure inside the bag during the eruption as the time progressed. The time of the eruption is jotted on the table above. However, the pressure inside the bag during the eruption time, for each test is observed to be consistent. The highest amount of pressure was observed in test 1.3 of experiment 1 (20.13) and the lowest amount of pressure inside the bag was observed in test 3.1 of experiment 3 (19.97kPa). Diagram 4.2.3 Table of averages of pH inside the bag for all the tests Time 2 4 6 8 10 12 14 16 18 20 22 24 26 28 30 pH 1- Test 1 (average) 3.6 3.6 3.6 3.6 3.6 3.7 3.7 3.7 3.8 3.8 3.8 3.8 3.8 3.8 3.8 pH 1- Test 2 (average) 3.13 3.27 3.4 3.84 4.1 4.23 4.33 4.5 4.77 4.93 4.97 5.03 5.07 5.1 5.1 pH 1- Test 3 (average) 3.33 3.77 4.33 4.47 4.9 5.07 5.27 5.37 5.5 5.63 5.67 5.7 5.73 5.73 5.77 Diagram 4.2.3: The above diagram shows the table of averages of the pH level inside the bag for all the test as the time progressed. For all the three tests, the pH level is observed to increase tremendously. This is due to the reaction of the vinegar and bi-carb inside the bag which is causing the solution to be more neutral. The most highest pH level (5.77) observed was in experiment 3, at the 30-minute point. However, experiment 3 is observed to have a higher rate of increase in pH level rather than experiment 1 and 2. 23 Stratovolcano- pyroclastic flow investigation Diagram 4.2.4 Table of averages of pH outside the bag for all the tests Time 2 4 6 8 10 12 14 16 18 20 22 24 26 28 30 pH 1- Test 1 (average) 4 4 4 4.1 4.1 4.1 4.1 4.1 4.1 4.1 4.1 4.1 4.1 4.1 4.1 pH 1- Test 2 (average) 5.73 5.73 5.73 5.73 5.73 5.73 5.73 5.73 5.73 5.73 5.73 5.73 5.73 5.73 5.73 pH 1- Test 3 (average) 5.17 5.17 5.1 5.1 5.1 5.1 5.1 5.1 5.1 5.1 5.1 5.1 5.1 5.1 5.1 Diagram 4.2.4: The above diagram shows the averages for the pH level outside the bag for all the tests as the time progressed. The pH level for all these tests are observed to be almost constant as there wasn’t much solution reaching the pH probe and when the pyroclastic flow erupts the pH level stays the same. However, the highest average pH level observed was in experiment 2 where the level of 5.73 stayed constant throughout the experiment. 24 Stratovolcano- pyroclastic flow investigation 4.3 Graphs (Stratovolcano Investigation) Figure 4.3.1: Graph of the pH inside bag for all tests vs the time taken for the lava to travel in the investigation. Figure 4.3.1: The above graph shows the graph of the average pH level inside the bag for all the test against the time taken for the experiment (seconds). The most prominent trend observed is the gradual increase of the pH level of test 3 over the period of time. Test 1 is observed to stay almost constant whereas test 2 is observed to increase after 4 seconds into the experiment. However, the steepest increase of pH level is observed in test 2 between 14 and 16 seconds into the experiment. This could be due to the increase of reaction rate when the bi-carb slab was subducted into the magma chamber containing 200ml of vinegar. The measurements for the pH level was observed only for 30 seconds as the endothermic reaction took place very fast. The observation of the bread and the effect took place throughout the experiments. 25 Stratovolcano- pyroclastic flow investigation Figure 4.3.2: Graph of the pH outside bag for all tests vs the time taken for the lava to travel in the investigation. Figure 4.3.2: The above graph shows the graph of the average pH level outside the bag for all the test against the time taken for the experiment (seconds). The graph above shows a common trend as the results of the pH level outside the bag was almost constant. However, test 2 is observed to remain constant throughout the experiment. This is due to the pyroclastic flow not reaching the pH probe throughout this experiment. The graph proves that the pH level outside the bag didn’t get effected by the pyroclastic flow from the stratovolcano. The measurements for the pH level was observed only for 30 seconds as the endothermic reaction took place very fast. The observation of the bread and the effect took place throughout the experiments. 26 Stratovolcano- pyroclastic flow investigation Figure 4.3.3: Graph of the average temperature for all tests vs the time taken for the lava to travel in the investigation. Figure 4.3.3: The above graph shows the graph of the average temperature of the pyroclastic flow (C) against the time taken for the experiment (seconds). The most prominent trend observed is how the temperature is decreasing for all the experiments in various time period. The steepest slope is observed in test 2 between 16 and 18 seconds into the experiment. This could be due to the reaction rate increasing as the vinegar and bi-carb is combined. However, in test 3, between 7 and 24 seconds, the temperature is observed to stay constant before it decreased from 15C to 14C. The temperature of the pyroclastic flow was only observed for 30 seconds as the endothermic reaction doesn’t take much time. The observation on the set-up was taken, as the time progressed. 27 Stratovolcano- pyroclastic flow investigation 4.4 Diagrams Figure 4.4: Diagram showing how the pyroclastic flow from the stratovolcano effected the houses and environment Pyroclastic flow Plan view Bi-Carb Elevation view Figure 4.4: The above diagram shows the before and after picture of the pyroclastic flow from the stratovolcano. The main purpose of this experiment was to investigate the effect of the pyroclastic flow to the infrastructure, house and the environment. In the plan view, it is clearly showing how the pyroclastic flow from the stratovolcano has destroyed some of the houses and the plants as the eruption occurs over time. This proves that volcanoes are a geohazard to humans and the environment. The houses are arranged 2cm apart from each other to have a better understanding and observation of the displacement of the pyroclastic flow. The diagrams from the elevation point of view shows how the pyroclastic flow is erupted and the effect it has on the houses, plants and the crust itself. For this experiment, vinegar represented as the mantle and the bi-carb represented as the SiO2 concentration. The release of pressure and energy is due to the endothermic reaction of the vinegar and the 28 bi-carb. However, the effects of the pyroclastic flow was well-observed by the group and the results were a good proof. Stratovolcano- pyroclastic flow investigation 4.5 Discussion of Results of the stratovolcano- pyroclastic flow It was initially predicted that, the more the amount of Bi-carb was subducted into the vinegar (magma chamber), the more the distance travelled by the pyroclastic flow as the time progressed. From the results obtained and observed, this hypothesis wasn’t accurate as there were a few problems arising throughout the experiment. Although the predictions were inaccurate, there were many observations taken for all the experiments. As per observation, the stratovolcano is a geohazard and it definitely has an effect towards infrastructure, humans and the environment. The most farthest distance reached by the pyroclastic flow was 14 cm in both test 2.1 and 2.2. This means that if this was a real-life stratovolcano, it would’ve effected anything which is 14km from the impact site. However, this could be due to the increase in the amount of bi-carb which represents the SiO2 content in the magma. The eruption of the pyroclastic flow of the stratovolcano observed in this experiment can be related to the 2018 Volcán de Fuego eruption. It was a series of volcanic explosions and pyroclastic flows from the Volcán de Fuego (Spanish for Volcano of Fire) in Guatemala on Sunday 3 June 2018. The eruption included lahars, pyroclastic flows, and clouds of volcanic ash, which left almost no evacuation time and caused over one hundred people confirmed killed. It was the deadliest eruption in Guatemala since 1929. However, the pressure inside the bag was observed to be consistent throughout the experiment but the pressure applied to the magma chamber was big. This caused a bigger eruption due to the solution going from high pressure to low pressure. The pyroclastic flow is the hot gas which travels at a very fast speed and this was observed throughout the experiment. The stratovolcano has a pyroclastic flow because the volcano layers consist of lava and ash. The SiO2 content was also used as a slab to subduct into the vinegar solution because Stratovolcanoes are common at subduction zones, forming chains and clusters along plate tectonic boundaries where oceanic crust is drawn under continental crust (continental arc volcanism, e.g. Cascade Range, central Andes, Campania) or another oceanic plate (island arc volcanism, e.g. Japan, Philippines, Aleutian Islands). The pH level inside the bag was observed to become more neutral because the vinegar and the bi-carb solution was concentrated by the end of the experiment. The pH level outside the bag was almost constant as the pyroclastic flow doesn’t have much of an effect outside (atmosphere). The temperature inside the bag was observed and the group made a conclusion on why the temperature was decreasing over the period of time. It is because when sodium bicarbonate and vinegar mix, they react with each other and one of the things that is made out of is carbon dioxide gas. The reaction needs heat to make it happen, so it takes heat from its surroundings, leaving the bottle feeling cold. 29 Stratovolcano- pyroclastic flow investigation Evaluation Before I say anything, this year has been a speed track, and everything is moving too fast. But knowing that I’ll be continuing what I love studying in university next year makes me happy. Year 10 Core, year 11 and 12 EES is always revolved around the concept of volcanism and earth processes. I enjoy studying the geological aspect of the Earth as it gives me the opportunity to be aware of why volcanoes erupt and how is an earthquake triggered. As this is my last report, I was excited to choose a topic relating to the concepts learned and start an investigation with a group of peers who are as curious as me. Investigations and writing a report has always been my favorite since the very beginning. Not only that, choosing what I want to experiment gives me an opportunity to think about what to do relating to the concepts learned during theory lessons. The lessons on how volcanoes are formed, the plate boundaries, the plate tectonic settings and the processes gave me and my peers an idea of what can be tested and observed over the certain period of time. Without any hesitation, planning for our investigation was made. Instead of complicating the investigation, we thought it through thoroughly and did a few test runs before making the final decision. Throughout this investigation, I have succeeded in learning many things, that I can apply not only to the classroom environment, but also the working environment, and of course in my life in general. In relation to the EES year 12 course, I learned all about the volcanoes and how they are functioning. The integration of the rock cycle, earth processes, Bowen’s series and geohazards help me understand everything in more depth. This investigation has given me a platform to learn many new skills, and to get into the right mindset in anticipation of how I will need to perform in the working world, in the near future. I learnt many things about time management and teamwork. Without teamwork and communication skills, I believe this investigation wouldn’t be a success. Through the process of writing up the investigation report, I have developed so many skills which I believe will benefit me in future. I sure have developed my technology literacy. Now, I would be confident to use a google spreadsheet and drawing for a simple table or even a graph and beautiful diagrams. In the upcoming years, I would love to improve my time management skills and I’m looking forward to being more organized. However, I will also need to plan the investigation beforehand in order to be more prepared for the actual investigation. This investigation tested my creativity skills, determination, teamwork, confidence, efficiency and time management skills. I was able to achieve all this, and in doing so, learnt much about the way I work among my peers; and what areas I must improve or develop which would be helpful in a professional working environment, in future. I’m now aware that I’m ready for a university learning environment. 30 Stratovolcano- pyroclastic flow investigation Bibliography • Impacts of pyroclastic flow, (2016), by Advancing Earth and Space Science [ONLINE]. Available at: https://agupubs.onlinelibrary.wiley.com/doi/full/10.1002/2015JG003316 • Volcán de Fuego - Wikipedia. 2018 [ONLINE] Available at: https://en.wikipedia.org/wiki/Volc%C3%A1n_de_Fuego • Volcán de Fuego 2018 eruption 2018. [ONLINE] Available at: https://en.wikipedia.org/wiki/2018_Volc%C3%A1n_de_Fuego_eruption • What are geohazards? [ONLINE] Available at: http://www.learnz.org.nz/geohazards152/what-are-geohazards 31