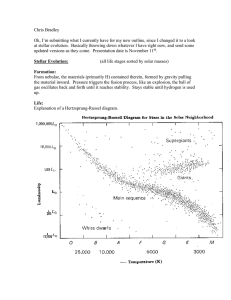
PHYSICAL SCIENCE 1 – Formation of the Heavy Elements – Dapul TOPIC / LESSON NAME CONTENT STANDARDS PERFORMANCE STANDARDS LEARNING COMPETENCIES SPECIFIC LEARNING OUTCOMES TIME ALLOTMENT We Are All Made of Star Stuff (Formation of the Heavy Elements) The learners demonstrate an understanding of the formation of the elements during the Big Bang and during stellar evolution. The learners demonstrate an understanding of the distribution of the chemical elements and the isotopes in the universe. The learners can make a creative representation of the historical development of the atom or the chemical element in a timeline. 1. Give evidence for and describe the formation of heavier elements during star formation and evolution (S11/12PS-IIIa-2) 2. Write the nuclear fusion reactions that take place in stars, which lead to the formation of new elements (S11/12PS-IIIa-3) 3. Describe how elements heavier than iron are formed (S11/12PS-IIIa-b-4) At the end of the lesson, the learners will be able to: 1. Briefly discuss stellar nucleosynthesis or fusion 2. Discuss other processes that led to other elements 3. Write out fusion reactions involved 120 minutes ~ 2 hours If the allotted subject time is 1 hour, it is recommended to end the first hour with #9. LESSON OUTLINE: 1. Introduction/Review: Overview of the objectives and key terms, Review of the Big Bang and Big Bang nucleosynthesis 2. Motivation: Discussion of text The Cosmic Connection 3. Instruction/Delivery: Continuation of Big Bang into star formation, Discussion of star fusion processes, Discussion of other fusion processes 4. Practice: Concept map, Alpha process practice 5. Enrichment: Brief discussion of man-made nuclei (presentation of performance task) 6. Evaluation: Question bank, Performance task to present one of the man-made elements MATERIALS RESOURCES Projector, computer Dhaliwal, J. K. (2012a). Nucleosynthesis: Alpha Fusion in Stars. [Powerpoint slides]. Retrieved from http://earthref.org/SCC/lessons/2012/nucleosynthesis/ Dhaliwal, J. K. (2012b). Nucleosynthesis: Heavier Elements. [Powerpoint slides]. Retrieved from http://earthref.org/SCC/lessons/2012/nucleosynthesis/ Nave, C. R. (2012). Nuclear fusion in stars. Retrieved http://hyperphysics.phy-astr.gsu.edu/hbase/astro/astfus.html September 23, 2015 from PHYSICAL SCIENCE 1 – Formation of the Heavy Elements – Dapul Penovich, K. (n.d.). Formation of the High Mass Elements. Retrieved September 23, 2015, from http://aether.lbl.gov/www/tour/elements/stellar/stellar_a.html Sagan, C. (2000). Chapter 26: The Cosmic Connection. In J. Agel (Ed.), Carl Sagan's Cosmic Connection: An Extraterrestrial Perspective. Cambridge: Cambridge University Press. Images: Figure 1. Equilibrium of the Sun [illustration]. (August 2007). Retrieved September 24, 2015 from http://lasp.colorado.edu/education/outerplanets/solsys_star.php#nuclear Figure 2. Elert, G. (2015a). Proton-proton chain (main branch) [diagram]. Retrieved September 23, 2015, from http://physics.info/nucleosynthesis/ Figure 3. Commonwealth Scientific and Industrial Research Organisation (CSIRO). (2015). Hydrogen Shell Burning on the Red Giant Branch [illustration]. Retrieved September 24, 2015 from http://www.atnf.csiro.au/outreach//education/senior/astrophysics/stellarevolution_postmain.html Figure 4. Elert, G. (2015b). Triple alpha process [diagram]. Retrieved September 23, 2015, from http://physics.info/nucleosynthesis/ Figure 5. Elert, G. (2015c). Carbon nitrogen oxygen cycle [diagram]. Retrieved September 23, 2015, from http://physics.info/nucleosynthesis/ PHYSICAL SCIENCE 1 – Formation of the Heavy Elements – Dapul PROCEDURE INTRODUCTION (5 MINUTES) 1. Introduce the following learning objectives using any of the suggested protocols (Verbatim, Own Words, Read-aloud) a. I can give evidence for and describe the formation of heavier elements during star formation and evolution. b. I can write the nuclear fusion reactions that take place in stars, which lead to the formation of new elements c. I can describe how elements heavier than iron are formed MEETING THE LEARNERS’ NEEDS Teacher Tip: 1. Display the objectives and terms prominently on one side of the classroom and refer to them frequently during discussion. 2. 2. To serve as an outline, you may map out the lesson using the diagram in the Practice portion of the lesson. Introduce the list of important terms that learners will encounter: a. fusion b. stellar nucleosynthesis c. proton-proton chain reaction d. triple alpha process e. alpha ladder f. CNO cycle g. main-sequence star h. red giant i. supernova explosion j. supernova nucleosynthesis k. r-process l. s-process REVIEW (15 MINUTES) 3. Review the stages of the Big Bang model, giving particular focus to nucleosynthesis and the formation of light elements such as H and He. Discuss briefly that more significant amounts of Li, Be and B formed through other processes, such as cosmic ray spallation. Remind them that once matter had recombined, gravity and other forces acted to bring matter together, eventually forming stars 200 B years after the Big Bang occurred. 4. Remind students how !"## !"#$%& !!!"#$ !"#$%& !"#$%& 𝑒𝑙𝑒𝑚𝑒𝑛𝑡 𝑠𝑦𝑚𝑏𝑜𝑙 symbols for an atom are written. Have (!!!) , or in terms of particle count, (!)𝑋 (!!!) them recall 3. Cosmic ray spallation is outside of the lesson’s scope, but simply explain that when particles in cosmic rays collide with heavier elements they generate Li, Be and B – among other elements – through nuclear fission. that MOTIVATION (15 MINUTES) 5. Briefly discuss the selection given as an assignment, Carl Sagan’s The Cosmic Connection. Sagan found it remarkable that the elements we find on Earth are also those we find amongst the stars, and that we find that most of what we know as matter was made by processes inside stars themselves. Enrichment: Using the Think, Pair, Share protocol, ask students to reflect on the selection. From their sharing, draw out one of the theses of the text: how being made of stardust makes us both cosmic – we are as much a part of the universe as the stars – and yet helps us realize that we are not the center of the universe. Teacher Tip: PHYSICAL SCIENCE 1 – Formation of the Heavy Elements – Dapul INSTRUCTION/DELIVERY (55 MINUTES) 6. Introduce that once hydrogen-helium stars had formed from the action of gravity, the hydrogen and helium atoms in stars began combining in nuclear fusion reactions that release a tremendous amount of light, heat, and radioactive energy. Fusion resulted in the formation of nuclei of new elements, so these reactions inside stars are known as stellar nucleosynthesis. Emphasize that the first fusion process occurs in the hydrogen core of stars with a temperature of less than 15 million K, such as the Sun. These kinds of stars are called main-sequence stars. Discuss the three steps of the process known as Figure 1. Equilibrium of the Sun and other mainthe main-branch proton-proton chain. sequence stars. (Equilibrium of the Sun, 2007). Deuterium (D or 2H) forms from proton fusion, with one proton turning into a neutron via beta-plus decay, giving off a neutrino and a positron: 1 H + 1H → 2H + ν + e + 3 He forms from deuterium and proton fusion, also known as deuterium burning. This immediately consumes all deuterium produced. 2 H + 1H → 3He + γ 4 He formation from 3He fusion. 3 He + 3He → 4He + 2 1H The entire three-step process releases about 26.7 MeV (megaelectronvolts) of energy. Emphasize that the energy released is responsible for the thermal pressure that pushes against gravity, and for the light, heat and radiation emitted by the star. Add that a different process facilitates hydrogen fusion in main-sequence stars with temp. greater than 15 million K. Figure 2. The main branch of the proton-proton chain reaction (p-p chain) resulting in the formation of 4He. (Elert, 2015a). Teacher Tip: 6. You may emphasize the rates of reaction in the proton-proton (p-p) chain, and point out that it will likely take a billion years before a specific proton is involved in a successful p-p fusion. 2He often immediately decays back into two protons, and rarely is a proton converted into a neutron to form deuterium (betaplus decay). However, with billions of protons reacting, enough make it to the next step. After discussing each of the steps, ask students to give the balanced equation for the formation of one 4He atom in the proton-proton chain. The answer is as follows: 4 1H → 4He + 2 ν + 2 e+ PHYSICAL SCIENCE 1 – Formation of the Heavy Elements – Dapul 7. Discuss how, as H is depleted, the core of a star becomes comprised of He instead, while H fusion only occurs in a shell around it. Due to this process, the temperature and density of the core of the star increases, up to 100 million K, and the star’s thermal pressure causes it to push out H gas. The star balloons into a red giant. Figure 3. A star with a very dense helium core and a hydrogen shell expands into a red giant due to increased radiation pressure. (CSIRO, 2015) Figure 4. The triple alpha process resulting in the formation of 12C. (Elert, 2015b) Several nuclear fusion processes occur in a red giant aside from hydrogen fusion, the first of which is the triple alpha process. Alpha particles refer to 4He, so this reaction involves the fusion of three 4 He atoms in the following steps: 4 He + 4He → 8Be 8 Be + 4He → 12C + γ Note that the 8Be intermediate is unstable, so either it decays or forms 12C. As it accumulates mass, the star can keep growing into a supergiant, where alpha fusion processes continue in the core via the alpha ladder. More and more alpha particles are fused to create heavier elements all the way until iron, making the core and star itself more massive. !" ! !" !𝐶 + !𝐻𝑒 → !𝑂 !" ! !" !𝑂 + !𝐻𝑒 → !"𝑁𝑒 !" ! !" !"𝑁𝑒 + !𝐻𝑒 → !"𝑀𝑔 !" ! !" !"𝑀𝑔 + !𝐻𝑒 → !"𝑆𝑖 !" ! !" !"𝑆𝑖 + !𝐻𝑒 → !"𝑆 !" ! !" !"𝑆 + !𝐻𝑒 → !"𝐴𝑟 !" ! !" !"𝐴𝑟 + !𝐻𝑒 → !"𝐶𝑎 !" ! !! !"𝐶𝑎 + !𝐻𝑒 → !!𝑇𝑖 !! ! !" !!𝑇𝑖 + !𝐻𝑒 → !"𝐶𝑟 !" ! !" !"𝐶𝑟 + !𝐻𝑒 → !"𝐹𝑒 7. Don’t give the complete alpha ladder. Show only the ladder until neon. Later on in Practice, ask students to write out the complete the alpha ladder. PHYSICAL SCIENCE 1 – Formation of the Heavy Elements – Dapul 8. Mention that once carbon was present from alpha processes, mainsequence stars hotter than 15 million K could facilitate the production of helium through a process where 12C is used as a catalyst: the carbon fusion cycle or the CNO cycle. Go through the cycle briefly, demonstrating that this process involves repeated proton capture and beta-plus decay. Figure 5. The CNO cycle, which uses 12C as a catalyst to form more 4He in larger or hotter main-sequence stars. (Elert, 2015c) 9. Finally, share how due to the formation of heavier elements, a star will eventually be unable to generate energy to push against gravity, causing it to collapse on itself. It then undergoes a supernova explosion. Discuss that this releases a tremendous amount of energy, enough to synthesize elements heavier than iron, including some of the heaviest elements known (uranium, thorium). This is done through the r-process, which involves rapid capture of neutrons by the atom. Mention that other heavy elements are also synthesized through s-process, which involves slow neutron capture in red giants. PRACTICE (30 MINUTES) 10. Review the lesson using the following concept map. Give the map with blanks in place of most of the terms, and ask students to fill the diagram in. Figure 6. Concept map for the current lesson. 8. It is good to teach this in a way that accounts for the number of protons and neutrons in each step: p+ n Next step: 12 C 6 6 Add a proton 13 N 7 6 Convert a proton 13 C 6 7 Add a proton 14 N 7 7 Add a proton 15 O 8 7 Convert a proton 15 N 7 8 Add a proton 12 C 6 6 4 He 2 2 9. r-process and s-process need not be discussed at length, but it will help to mention that these processes change the atom’s atomic weight, after which the atom undergoes various decay processes to change its identity. Teacher Tip: 10. Alternately, this concept map may be used at the start of the lesson to guide students with all the terms. PHYSICAL SCIENCE 1 – Formation of the Heavy Elements – Dapul 11. Using a provided periodic table, allow students to write out all the equations involved in the alpha ladder. What do they notice about the atomic number patterns of the elements found in the ladder? Explain that this feature of the alpha ladder, as well as other rules of stability, results in the oddnumbered elements being generally less abundant than the even-numbered elements beside them on the periodic table. Emphasize that many other processes allowed for the odd-numbered elements, including supernova nucleosynthesis, radioactive decay, electron and neutron capture, nuclear fission, and cosmic ray spallation. 11. Students should notice that mostly even-numbered elements emerge through the alpha ladder, and that other elements between carbon and iron need to be accounted for in other ways. ENRICHMENT (10 MINUTES & OUTSIDE OF CLASS) 12. Mention that quite a few elements were first discovered as man-made elements, as many of them were not found to emerge from the major nucleosynthesis reactions (or their minor processes). These include elements Americium through Lawrencium, and also include some of the newer, recently discovered elements (eg. Flerovium, Livermorium). Inform students that they will have to research on one of these elements for the Performance Task. Teacher Tip: EVALUATION (20 MINUTES) A. Question Bank (sample questions for Written Evaluation) Teacher Tip: A. Correct answers: 1. b. r-process 1. 2. Which of the following processes is likely to generate the heaviest element? a. CNO cycle c. triple-alpha process b. r-process d. Big Bang nucleosynthesis Which of the following reactions is not a part of the alpha ladder? ! !" ! !" a. !" c. !" !"𝑀𝑔 + !𝐻𝑒 → !"𝑆𝑖 !"𝐴𝑟 + !𝐻𝑒 → !"𝐶𝑎 ! !" ! !" b. !" d. !! !!𝑇𝑖 + !𝐻𝑒 → !"𝐶𝑟 !"𝑃 + !𝐻𝑒 → !"𝐶𝑙 3. If an element is used up by a star in fusion, it is sometimes called “burning”, even though no actual combustion occurs. Which of the following processes is likely to involve “carbon burning”? a. alpha ladder c. triple-alpha process b. CNO cycle d. s-process 4. Modified True or False: If the statement is true, write True. Else, replace the underlined portion with the correct word or phrase. a. A star gets lighter as time goes on. b. Most of the heaviest elements were formed in main-sequence stars. c. The heavy elements in a star are found in its core. 2. !" !"𝑃 + !!𝐻𝑒 → !" !"𝐶𝑙 3. a. alpha ladder (CNO cycle does not consume any carbon; it simply uses carbon as a catalyst) 4. a. heavier b. supernova c. True d. True B. Emphasize the importance of proper research skills and citation throughout the task, even with creative outputs. If there is no time to present in class, they may present outside of class directly to you, or submit in a form that can be viewed separately. PHYSICAL SCIENCE 1 – Formation of the Heavy Elements – Dapul d. In stellar nucleosynthesis, heavier elements are formed from combining lighter ones. B. Performance Task Students will create an output that discusses the origin of one of the man-made elements. In their output, they must: • discuss the element’s basic characteristics • give a brief timeline leading up to the element’s discovery Students may present their research in the form of a poster, powerpoint, infographic. A sample rubric may be given as follows: 1 pt. 3 pts. Presentation of the Most basic Basic aspects of the element’s characteristics of the element that can be characteristics element are missing or found on the periodic absent table are present Timeline of the There is no clear chain element’s discovery of events or key moments presented (add creative metric) (add other metrics) The scientific history of the element’s discovery was made clear, including notable people and groups involved a report or essay, video, or 5 pts. Unique aspects of the element, for example the element’s potential significance or uses, were presented The element’s history was presented clearly and the process of creating the element was discussed