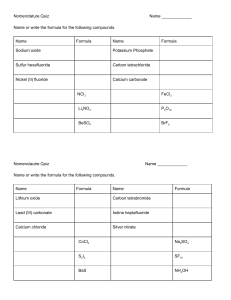
Inorganic chemistry (e) Acids, alkalis and titrations What colour will these indicators turn in acidic and alkaline solution? ACIDIC SOLUTION ALKALINE SOLUTION Litmus Phenolphthalein Methyl Orange What pH would the following solutions be and what colour with Universal Indicator? Solution pH Colour with Universal Indicator Strongly acidic Weakly acidic Neutral Weakly alkaline Strongly alkaline Acids in aqueous solution provide ______________________ ions and alkalis in a aqueous solution are a source of ____________________ ions. Alkalis and acids together make a _______________________________ reaction. f) Acids, bases and salt preparations Solubility rules to learn! common sodium, potassium and ammonium compounds are soluble all nitrates are soluble common chlorides are soluble, except those of silver and lead(II) common sulfates are soluble, except for those of barium, calcium and lead(II) common carbonates are insoluble, except for those of sodium, potassium and ammonium common hydroxides are insoluble except for those of sodium, potassium and calcium (calcium hydroxide is slightly soluble) Are the following compounds soluble (S) or insoluble (I) Compound Calcium nitrate Iron hydroxide Sodium chloride Aluminium carbonate S or I? Compound Silver chloride Gold sulfate Potassium carbonate Calcium sulfate S or I? Do acids donate or accept protons? Do bases donate or accept protons? What salts do you make when you add these together? Name of salt made Hydrochloric acid + iron Hydrochloric acid + sodium hydroxide Hydrochloric acid + potassium carbonate Sulfuric acid + calcium Sulfuric acid + potassium hydroxide Sulfuric acid + lead carbonate Nitric acid + barium hydroxide Nitric acid + beryllium carbonate Alkalis are _________________ that are soluble in water. How could you prepare a pure, dry sample of a soluble salt, starting from an insoluble reactant. (g) Chemical tests Describe tests for these gases. Gas Test Hydrogen Oxygen Carbon dioxide Ammonia Chlorine How would you carry out a flame test? What colours are formed in flame tests for these cations: Cation Lithium Li+ Sodium Na+ Potassium K+ Calcium Ca2+ Copper Cu2+ Colour Describe tests for these cations: Cation Ammonium NH4+ Copper Cu2+ Iron (II) Fe2+ Iron (III) Fe3+ Test Describe tests for these anions: Cation Chlorine + acidified silver nitrate Bromine + acidified silver nitrate Iodine + acidified silver nitrate Sulfate + acidified barium chloride Carbonate + hydrochloric acid Test How could you use anhydrous copper(II) sulfate to test for water? How could you tell whether a sample of water is pure? Organic chemistry (a) Introduction Hydrocarbons are compounds with only ______________________ and _______________________ These are multiple ways to represent compounds What is meant by the following terms: homologous series functional group isomerism Write the possible structural and displayed formulae of this organic molecule. What is meant by these types of reactions Substitution Addition Combustion (b) Crude oil How does the industrial process of fractional distillation separate crude oil into fractions? What are some uses of these main fractions obtained from crude oil: USES refinery gases gasoline kerosene diesel fuel oil bitumen What are the trends of the main fractions. colour boiling point viscosity What are the possible products of complete and incomplete combustion of hydrocarbons with oxygen in the air. Why is carbon monoxide poisonous to humans? In car engines, nitrogen and oxygen from air to react and form oxides of nitrogen. Why does this happen in car engines? What do the impurities in hydrocarbon fuels result in? Sulfur dioxide and oxides of nitrogen both contribute to which environmental problem? When long-chain alkanes are added to silica or alumina and heated to 600–700 oC, what are the results? Why is cracking necessary? (c) Alkanes What is the general formula for alkanes ? Why are alkanes classified as saturated hydrocarbons? When there is more than one carbon a functional group can be located it is important to distinguish which carbon the functional group is on. Each carbon is numbered and these numbers are used to describe where the functional group is When 2 functional groups are present di- is used as a prefix to the second part of the name Branching also needs to be considered, the carbon atoms with the branches are described by their number When the location of functional groups and branches needs to be described the functional group takes precedence so the functional group has the lowest number What are the structural and displayed formulae for these alkanes. Alkane Structural formula Methane Ethane Displayed formula Propane Butane Pentane What happens when alkanes react with halogens in the presence of ultraviolet radiation? (d) Alkenes Which functional group do alkenes have? What is the general formula for alkenes? Why are alkenes classified as unsaturated hydrocarbons ? What are the structural and displayed formulae for these alkenes. Alkene Ethene Structural formula Displayed formula Propene Butene What is produced when alkenes react with bromine? How can bromine water be used to distinguish between an alkane and an alkene? (e) Synthetic polymers What is formed by joining up many small molecules called monomers? Repeat units are used to make drawing polymers easier Change the double bond in the monomer to a single bond in the repeat unit. Add a bond to each end of the repeat unit. Try drawing the repeat units of the following polymers. Name poly(ethene) Repeat unit poly(propene) poly(chloroethene) (poly)tetrafluoroethene How can you work out the structure of a monomer from the repeat unit of a polymer? What are the problems in the disposal of addition polymers?