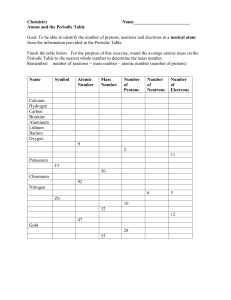
Element Practice 2 Atomic Number – Represents both the number of protons and electrons in an element. Atomic Symbol – a short hand way of writing each elements name. He Most of the mass comes from the neutron and proton. To find the number of neutrons, subtract the atomic weight from the atomic mass. 4 Atomic Mass – The weight of an element. . Fill in the necessary information. 2 26 13 He Fe Al 4 56 27 element: ________________ element: _______________ element: _______________ # protons: _______________ # protons: ______________ # protons: ______________ # neutrons: ______________ # neutrons: _____________ # neutrons: _____________ 20 10 6 Ca Ne C 40 20 12 element: ________________ element: _______________ element: _______________ # protons: _______________ # protons: ______________ # protons: ______________ # neutrons: ______________ # neutrons: _____________ # neutrons: _____________ 9 17 11 F Cl Na 19 35 23 element: ________________ element: _______________ element: _______________ # protons: _______________ # protons: ______________ # protons: ______________ # neutrons: ______________ # neutrons: _____________ # neutrons: _____________ Directions: Calculate the number of neutrons and complete the Bohr model for each element. 2nd shell can fit up to 8 more electrons 1st shell can fit up to 2 electrons Element: Helium Element: Neon Element: Nitrogen Neutrons: ________ Neutrons: ________ Neutrons: ________ Element: Beryllium Element: Oxygen Element: Chlorine Neutrons: ________ Neutrons: ________ Neutrons: ________ Element: Boron Element: Sodium Element: Fluorine Neutrons: ________ Neutrons: ________ Neutrons: ________ 3rd shell can fit up to 18 more electrons