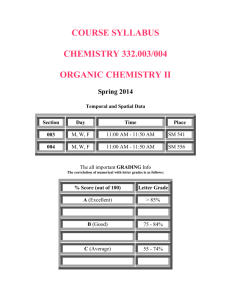
Chemistry 115 Spring 2007 Lec. 001, CRN# 11042; Lab 002, CRN# 11043 Instructor: Dr. M. Richards-Babb (Dr. Babb) Phone: 293-3435 Ext 6416 Office: 461 CRL (Chemical Research Lab) e-mail: mrichar2@wvu.edu website: www.as.wvu.edu/~mbabb Office Hours: T, Th (10:00-11:15 PM) and W (11:00 AM-12:00 PM). Other times by appointment. Lecture: Section 001 (T, Th - 8:30-9:45 AM) Clark Hall Room 101 Laboratory: Sec. 002 (W - 1:30-4:20 PM) Starting Wednesday, Jan. 17. Lab rooms to be assigned (see website). Course Objectives: The objectives of this course are to present the fundamental principles of chemistry upon which subsequent course work is built. Basic topics include, but are not limited to, atomic and molecular structure, the periodic table and periodicity, chemical stoichiometry, reactions (precipitation, acid-base, and redox), thermodynamics, and properties of gases, liquids, and solids. Laboratory experiments are designed to support the lecture topics. Textbook: “Chemistry”, by Brady and Senese, 4th Ed. Required of all students. Problem Book: “Chemical Problem Solving Using Dimensional Analysis”, by R. Nakon. Required of all students. Laboratory Text: “Laboratory Experiments for General Chemistry 115", by WVU Staff, 2nd Ed. Required of all students. Because of the use of fill-in blanks and graphs in the book, each student MUST have a new copy. Calculators: Only non-programmable calculators may be used during quizzes and exams. Scientific calculators ($8-$12) that perform all the needed mathematical calculations (including logarithms) are available at the University Book Store as well as at various commercial establishments. Programmable calculators will be confiscated at the examination site or the student will not be allowed to take the exam. Seating: Each student will be assigned a particular seat in the lecture room. Attendance will be checked each lecture. Seating assignments will be posted on the website by the end of the second week of classes. Attendance Policy: Attendance will be used in determining each student’s final grade as shown below. There are no “excused absences”, although minor adjustments may be made for personal emergencies. Good attendance (2 or fewer absences) will be rewarded by addition of 1% to the final numerical average. Student Absence 1-4 5-9 10-14 15-19 >19 Graded Online Homework: Penalty None Final grade dropped by one letter grade. Final grade dropped by two letter grades. Final grade dropped by three letter grades. Final grade dropped by four letter grades. In place of quizzes, there will be graded online homeworks (up to three assignments per chapter). It is your responsibility to complete the homeworks by the given deadlines. In general you will have three attempts at each question. The online homework can be found at the following website address: http://edugen.wiley.com/edugen/class/cls21918 for 115-001 Before you can access this homework, you must self-register (click the register button on the website). Be sure to register for the correct section of Chem 115. To register, you will need the WileyPLUS REGISTRATION CODE. You should have obtained this code when you purchased your Brady and Senese textbook package. A registration tutorial can be found at http://www.wiley.com/college/twomin/stu/register.html Hourly Exams Hourly exams will be given outside of lecture. The schedule for exams is as follows: Thur., Feb. 1 Exam #1 (7-8 PM) Thur., Feb. 22 Exam #2 (7-8 PM) Thur., March 15 Exam #3 (7-8 PM) Thur., April 19 Exam #4 (7-8 PM) Wed., April 25 Lab Final Exam (In Lab) Tue., May 1 Final Exam (7-9 PM) Students are expected not to register for a class that will conflict with these exam times. Cell phones are not permitted during exams/quizzes. Covers should be removed from nonprogrammable calculators before entering the exam/quiz site. Final Exam: A comprehensive final exam on the material covered in Chemistry 115 is scheduled for Tuesday, May 1 from 7:00 PM-9:00 PM. ANY STUDENT SCORING 40% OR BELOW ON THE FINAL EXAMINATION WILL EARN A GRADE OF “F”, REGARDLESS OF HIS/HER OTHER AVERAGES. SIMILARLY, OTHER VERY LOW SCORES COULD RESULT IN A REDUCTION OF ONE LETTER GRADE. This final exam will be comprehensive and cumulative covering the entire course. Due attention will be given to the cumulative nature of the learning process with emphasis being placed on major topics and concepts. Your study during the semester should be designed for comprehensive and long-term retention of the factual material, principles, and use of these. “Cramming” for individual exams largely defeats the purpose of a college education. Laboratory Final Exam: A comprehensive laboratory final examination will also be given during the last laboratory period on Wednesday, April 25. Additional details will be provided at a later date. Make-Ups: Make-up exams and labs will only be given for students with legitimate absences (university business, serious illness, medical emergencies and family emergencies). A documented excuse must accompany all requests for make-ups. The instructor should be informed of the absence as soon as possible. Prior knowledge especially in the case of absences due to university business is appreciated. Calculation of Grade: My object is to give you the best possible grade which can be justified by your achievement in the course. Final numerical averages will be calculated as shown below: Hourly Exams (4) 40% (10% each) Online Hmwk. Avg. 10% Lab Avg. 25% Final Exam 25% The Laboratory Average is calculated as follows: Unknown Average 65% Lab Final Exam 25% Lab Hmwk & TA Eval. 10% (Lowest unknown grade dropped.) NOTE: The lowest unknown grade from a completed lab will be dropped. A zero due to an absence from lab will not be dropped. There is only one scheduled make-up laboratory. While individual exams and quizzes may vary in relative difficulty, experience with my classes in the past has shown that final numerical averages in the sixty range will earn a student a grade of “D”, averages in the seventy range will earn a “C”, averages in the eighty range will earn a “B” and averages above ninety will earn a grade of “A”. In general, the hourly exam average is the best indicator of your performance. In order to determine how you are doing in the class at any given time, calculate your hourly exam average and compare it to the class average. If your hourly exam average is much higher than the class average, you are doing “A” or “B” work. If your average is close to the class average, you are doing “C” work. If your average is well below the class average, you are doing “D” or “F” work. Problems and At the end of each chapter in the text and in the problem book are numerous questions and Questions: problems. Specific problems related to the material covered in lecture will be assigned as other homework. Although these problem assignments will not be collected and graded, they should help you understand the various concepts in lecture and thereby prepare you for the examinations. Answers to many of these problems can be found in the back of your textbook and problem book. West Virginia University is committed to social justice. I concur with that commitment and expect to foster a nurturing learning environment based upon open communication, mutual respect, and non-discrimination. Our University does not discriminate on the basis of race, sex, age, disability, veteran status, religion, sexual orientation, color or national origin. Any suggestions as to how to further such a positive and open environment in this class will be appreciated and given serious consideration. If you are a person with a disability and anticipate needing any type of accommodation in order to participate in this class, please advise me and make appropriate arrangements with Disability Services (293-6700). West Virginia University recognizes the diversity of its students, many of whom must be absent from class to participate in religious observances. Students must notify their instructors by the end of the third class meeting regarding religious observances that will affect attendance. Further, students must abide by the attendance policy of their instructors as stated on their syllabi. Faculty will make reasonable accommodation for tests or field trips that a student misses as a result of religious observance. Chemistry 115 Approximate Lecture Syllabus Spring 2007 Instructor: Text: Prob. Bk: Dr. Richards-Babb - Office Room 461 CRL “Chemistry” by Brady and Senese, 4th Ed. “Chemical Problem Solving Using Dimensional Analysis”, by Nakon. -----------------------------------------------------------------------------------------------------------------Day T Date Jan 9 Chapter 1,3 Th T Jan 11 Jan 16 3 1 Th T Th T Th T Th T Th Jan 18 Jan 23 Jan 25 Jan 30 Feb 1* Feb 6 Feb 8 Feb 13 Feb 15 2 2 4 4 4 4 5 5 5, 6 T Th T Feb 20 Feb 22* Feb 27 6 6 6, 7 Th T Th Mar 1 Mar 6 Mar 8 T Text Pages 1-12; 79-90 Topics 90-105 13-34 Sig. Figs., Exp. Number Review, Factor-Label Method Chemical/Physical Properties, Classific. of Matter; Units, Measurement Atomic Theory, Subatomic Particles, Isotopes, Periodic Table Chemical Compounds, Equations 7 7 8 41-53 53-72 112-116 116-139 116-139 139-144 154-164 164-206 PB 155-157, 169-177 215-224 224-233 224-233 233-237, 1053 255-263 263-280 280-293 303-336 Mar 13 8 337-346 Hund’s Rule (PB 63-65), Paramag./Diamag., Periodic Properties Th T Mar 15* Mar 20 8 9 337-346 353-381 Periodic Prop. Cont, EXAM #3 (7-8 PM) Th T Th Mar 22 Apr 3 Apr 5 9 10 10, 11 381-394 401-417 417-437, 451-468 Formal Charges, Resonance T Apr 10 11 468-495 Ideal Gas Law, Gas Stoich., Dalton’s Law, Kinetic Molec. Theory Th Apr 12 12 504-532 Intermolecular Forces, Changes of State, Vapor Pressure, T Apr 17 12 534-540 Th T Th Apr 19* Apr 24 Apr 26 12 --------------- 534-540 ------------------- Ions and Naming, Compound Properties Atomic/Molecular/Formula Mass, Mole, NA Mass % , Empirical/Molec. Formulas, Stoichiometry Stoichiometry, EXAM #1 (7-8 PM) Limiting Reagent and % Yield Problems, Solutions Solutions, Electrolytes, Net Ionic Equations, Solubility Predict. Products, Acids/Bases, Anhydrides, Molarity Titrations, Normality and Equivalants, Redox Rxns. Balancing Redox Rxns. Redox Rxns. Cont, EXAM #2 (7-8 PM) Metal and Non-metal Activity Series, Intro. to Heat Calorimetry, Energy During Bond Breaking/Making Thermochemical Eqn., Hess’ Law, )Hf/ Electromagnetic Radn., Line Spectra, Orbitals, Quantum #’s, Electronic Configurations Water Hardness, Ionic/Covalent Bonds, Octet Rule, Electronegativity, Polar Bonds, Lewis Structures VSEPR Theory, Molecular Shapes, Polar Molecules Valence Bond Theory, Hybridization, Gas Intro., Specific Gas Laws (Boyle’s, Charles’...) Boiling Point Phase Diagrams, Heat Gained/Heat Lost Prob. (PB 184189) Heat Gained/Heat Lost Cont., EXAM #4 (7-8 PM) Special Topics (as yet to be determined) REVIEW (for Final Exam) FINAL EXAMINATION, TUESDAY, MAY 1 (7-9 PM) Chemistry 115 Laboratory Schedule SPRING 2007 If you want to leave lab at a reasonable time each day, read the experiment before coming to lab. For safety reasons, all students must leave the lab room at the specified time. This means that you should begin cleaning up at 12:10 PM (or 4:10 PM). If you have not completed the entire experiment, ask your TA or instructor about the best course of action. Week of: Experiment Jan. 8-12 No Lab Jan. 15-19 Check-In, Video Tape, Worksheets (Appendices A & B) Jan. 22-26 Exp. #1: Laboratory Techniques Jan. 29-Feb.2 *Exp #2: Measurement and the Metric System Feb. 5-9 *Exp #4: Determination of Percent KClO3 Feb. 12-16 *Exp #3: Chemicals in the Home Feb. 19-23 *Exp #5: Quantitative Analysis for Iron in Solution Feb. 26-Mar.2 *Exp #7: Titrations of Acids and Bases Mar. 5-9 *Exp #11: Halogens and Halides Mar. 12-16 *Exp #8: Enthalpy of Neutralization Mar. 19-23 Exp #9: Water Hardness Apr. 2-6 *Exp #10: Shapes of Molecules Apr. 9-13 *Exp #6: Molecular Weight Determination Apr. 16-20 Make-Up Lab (for students w/instructor excused absences only) Apr. 23-27 Lab Final Exam and Check Out * NOTE: A clean, dry, labeled test tube should be placed in the rack during the laboratory period where a * is shown. This is for the unknown sample to be handed out the following week. There is a laboratory fee of $60 payable with your tuition. The laboratory fee is not refundable after the first week of classes. A charge for excessive breakage may also be levied. IF YOU DROP THIS COURSE, YOU MUST CHECK OUT OF YOUR LABORATORY DESK. Chemistry 115/116 Laboratory Laboratory Fee: There is a laboratory fee of $60 payable with your tuition. The laboratory fee is not refundable after the first week of classes. A charge for excessive breakage may also be levied. IF YOU DROP THE COURSE, YOU MUST CHECK-OUT OF YOUR LABORATORY DESK. Laboratory Conduct: There is no smoking in the building. Each student is to do his/her own work in a quiet, efficient manner. The work area is to be kept clean and orderly. At the end of the laboratory period, the work area/sink should be clean and all common equipment should be returned to the common equipment container. The gas jets should be checked to ensure that they have been turned off. The student should inspect the desk area to ensure that no apparatus has been left out. Laboratory Safety: Read the safety regulations carefully and adhere to them rigorously at all times for your own well-being and that of your fellow students. YOU MUST WEAR SAFETY GOGGLES (NOT SUNGLASSES OR CONTACTS) WHENEVER YOU ARE IN THE LABORATORY. Covered shoes (not sandals, clogs, or open shoes) are to be worn at all times in the laboratory. Long pants or long skirts are to be worn instead of shorts. Old jeans, a Tshirt, and sneakers are the best laboratory attire. Portable radios, walkman, etc. are prohibited in the laboratory. NO FOOD OR DRINK IS PERMITTED IN THE LABORATORY. Laboratory Absences: Students should attend all laboratory sessions. Students absent because of severe illness or university business will be given a make up lab at the end of the semester. A documented excuse is required to make up a lab. This excuse should be given to the instructor in charge and not to the TA. Preliminary Work: Before coming to the laboratory, you should read through the experiment carefully. Make a list of all precautions to be observed and plan the best way to do the experiment beforehand. Desk Locks: You will be given a combination lock for your desk. You are responsible for the equipment in your desk. It is your responsibility to open and lock your own desk. Unknowns: Only one attempt will be allowed for each unknown. Check any calculations carefully before you hand in your result. These results should be handed in at the end of the period. If you have not completed the experiment, notify your teaching assistant.