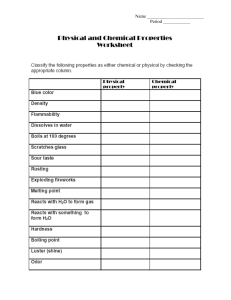
Chapter 6: Chemical Reactions Identify and predict single displacement reactions. The reactivity series is a series of metals, in order of reactivity from highest to lowest. It is used to determine the products of single displacement reactions, whereby metal A will replace another metal B in a solution if A is higher in the series. Activity series of some of the more common metals, listed in descending order of reactivity. The reactivity of metals is due to the difference in stability of their electron configurations as atoms and as ions. As they are all metals they will form positive ions when they react. Potassium has a single outer shell electron to lose to obtain a stable "Noble gas" electron configuration; the precious metals which exist in the d-block cannot form structures which are much more stable than their elemental state with the loss of just a few electrons. Metals that require the loss of only one electron to form stable ions are more reactive than similar metals which require the loss of more than one electron. Group 1 metals are the most reactive for that reason. Metals with a greater total number of electrons tend to be more reactive as their outermost electrons (the ones which will be lost) exist further from the positive nucleus and therefore they are held less strongly. In a single-displacement reaction, one element displaces another in a compound. Single displacement reactions follow this general equation: A + BC AC + B In single-displacement reactions, one metal can replace hydrogen or another metal. A metal will not always replace a metal in a compound dissolved in water because of differing reactivities. An activity series can be used to predict if reactions will occur. Halogens frequently replace other halogens in replacement reactions. Halogens also have different reactivities and do not always replace each other. An activity series can be used to predict if reactions will occur. Identify which two elements would be likely to swap. Consult the activity series to see which element is more reactive. If the free element is more reactive, it will trade places with the element in the compound. If the free element is less reactive, no reaction will take place. Predict whether the following single displacement reactions will occur. If a reaction occurs, write a balanced equation for the reaction. K(s) + ZnCl2(aq) HF(aq) Cl2(g) + Fe(s) + Na3PO4(aq) Al(s) + Pb(NO3)2(aq) Metals Metal Ion Reactivity K Na Li Ba Sr Ca Mg Al Mn Zn Cr Fe Cd Co Ni Sn Pb H2 Sb Bi Cu Hg Ag Au Pt K Na+ Li+ Ba2+ Sr2+ Ca2+ Mg2+ Al3+ Mn2+ Zn2+ Cr2+ Fe2+ Cd2+ Co2+ Ni2+ Sn2+ Pb2+ H+ Sb2+ Bi2+ Cu2+ Hg2+ Ag+ Au3+ Pt+ reacts with water reacts with water reacts with water reacts with water reacts with water reacts with water reacts with acids reacts with acids reacts with acids reacts with acids reacts with acids reacts with acids reacts with acids reacts with acids reacts with acids reacts with acids reacts with acids included for comparison highly unreactive highly unreactive highly unreactive highly unreactive highly unreactive highly unreactive highly unreactive +