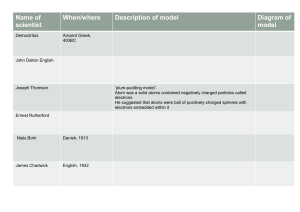
Niels Bohr and Bohr Diagrams Who is Niels Bohr? • Niels Bohr was a Danish scientist born in 1885. • Bohr received a Nobel Prize in Physics in 1922 for his contribution to the Atomic Model. • He proposed that electrons revolve in stable orbits(shells) around the nucleus of an atom based on the energy levels of its electrons. • This model expanded on Ernest Rutherford’s model of 1911. Comparing Rutherford and Bohr’s models of the atom Atomic Structure Review • Atoms have a nucleus that contains Protons and Neutrons • Electrons are contained in orbits or shells that surround the nucleus • An atom is made of mostly empty space • Protons have a positive (+) charge • Electrons have a negative (-) charge • Neutrons are Neutral Bohr Models are NOT Boring! How to Draw Bohr Diagrams Bohr Diagrams 1) Find the element on the periodic table. 2) Determine the number of electrons – How do we do this?. 3) This is how many electrons you will draw. Bohr Diagrams 1 • Find out which period (row) your element is in. 2 3 4 5 6 7 •Elements in the 1st period have one energy level. •Elements in the 2nd period have two energy levels, and so on. Bohr Diagrams 1) Draw a nucleus with the element symbol inside. C P+ = ? N0 = ? 2)Write the number of Protons and Neutrons the element has inside the nucleus. Put a + by the P and a little 0 by the N. Why do we do this? Note: Round mass to nearest 1 when figuring neutrons. The electron shells surrounding the nucleus each hold a particular number of electrons. Shells are named with letters: 1 = K shell 2 = L shell 3 = M shell 4 = N shell 5-7 = O, P, Q = 2 electrons = 8 electrons = 8 electrons = 18 electrons = up to 32 electrons Remember: The outer shell of an atom (no matter what letter) can only hold 8 electrons! What does this mean for Elements with NOPQ shells? Bohr Diagrams C P+ = 6 N0 = 6 1) Carbon is in the 2nd period, so it has two energy levels, or shells. 2) Draw the shells around the nucleus. Bohr Diagrams 1) C P+ = 6 N0 = 6 2 e- Add the electrons. 2) Carbon has 6 electrons. 3) The first shell can only hold 2 electrons. 4) Write the number of electrons that shell holds and e- for electrons an their charge. 5) How many electrons are in the outer shell? Bohr Diagrams 1) C P+ = 6 N0 = 6 2e- 4e- Since you have 2 electrons in the first shell, you need to add 4 more. 2) These go in the 2nd shell. 3) Again, write e- and the number of electrons. Bohr Diagrams 1) Check your work. C P+ = 6 N0 = 6 2e- 4e- 2) You should have 6 total electrons for Carbon. 3) Only two electrons can fit in the 1st shell. 4) The 2nd shell can hold up to 8 electrons. 5) The 3rd shell can hold 18, but the elements in the first few periods only use 8 electrons. But do electrons clump up like this model shows? Why/why not? Bohr Diagrams 1) 2) 3) Add the electrons. Carbon has 6 electrons. The first shell can only hold 2 electrons. To correctly draw the dots… Draw an X over the diagram and start at the far right working counterclockwise. Bohr Diagrams 1) 2) 3) Add the electrons. Carbon has 6 electrons. The first shell can only hold 2 electrons. To correctly draw the dots… Draw an X over the diagram and start at the far right working counterclockwise. Bohr Diagrams 1) 2) 3) Add the electrons. Carbon has 6 electrons. The first shell can only hold 2 electrons, and the electrons exist opposite one another. To correctly draw the dots… Draw an X over the diagram and start at the far right working counterclockwise. Bohr Diagrams 1) 2) 3) Since you have 2 electrons already drawn, you need to add 4 more. These go in the 2nd shell. Add one at a time -starting on the right side and going counter clock-wise. Bohr Diagrams 1) 2) 3) Since you have 2 electrons already drawn, you need to add 4 more. These go in the 2nd shell. Add one at a time -starting on the right side and going counter clock-wise. Bohr Diagrams 1) 2) 3) 4) 5) Check your work. You should have 6 total electrons for Carbon. Only two electrons can fit in the 1st shell. The 2nd shell can hold up to 8 electrons. The 3rd shell can hold 18, but the elements in the first few periods only use 8 electrons. Your turn! You will each be given an element from the first 20 elements of the Periodic table. You will need to draw the electron configuration of your element.