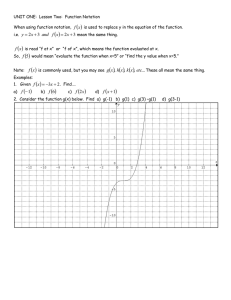
CHEM MODULE 1 Lesson 1: What is Chemistry? New vocabulary MATTER chemistry science hypothesis theory scientific law pure research applied research substance mass weight model MATTER chemistry science hypothesis theory scientific law pure research applied research substance mass weight model chemistry science 1 Compare and contrast mass and weight using the Venn diagram below. • does not reflect gravitational pull on matter • a measure of the effect of gravitational pull on matter • a measurement that reflects the amount of matter in an object MASS WEIGHT *Explain a chemical model by completing the following sentences. Use the words listed below The ____________, composition, and ____________of all matter can be explained on a____________ level. All that we observe depends on____________ and the____________ they undergo. ____________seeks to explain the submicroscopic events that lead to____________. One way to do this is by making a chemical model, which is a____________ of a ____________. Word choices Atoms Behavior Changes Chemistry Macroscopic observations Structure Submicroscopic Submicroscopic event Visual representation *Explain why scientists use mass instead of weight for their measurements. 2 Lesson 2: Measurement New vocabulary base unit second meter kilogram kelvin derived unit liter density scientific notation dimensional analysis base unit derived unit density scientific notation conversion factor dimensional analysis conversion factor Match the SI base units with their functions Meter Distance Liter Temperature Kilogram Time Kelvin Mass Second Volume The Metric System BASE UNITS Kilo Hecto Deca Meter Gram Deci Centi Milli Liter Larger Units Smaller Units The following pneumonic will help you remember the prefixes “ “ If you move to the _________ in the diagram, move the decimal to the________ If you move to the _________ in the diagram, move the decimal to the ________ 3 Using the pneumonic to remember the prefixes, draw the boxes for the metric system and answer the following questions: Practice Problem #1 1 meter = ___________________ centimeters? Practice Problem #2 1,000 millimeter = ___________________ centimeters? Practice Problem #3 16,093 meters = ___________________ kilometers? Practice Problem #4 40,000 meter = ___________________ kilometers? Practice Problem #5 4,000 meters = ___________________ kilometers? Practice Problem #6 4,000 centimeters = ___________________ meters? Practice Problem #7 5 meters = ___________________ centimeters? Practice Problem #8 0.3 kilometers = ___________________ meters? Practice Problem #9 0.04 kilometers = ___________________ millimeters? Derived Units/Density One of the derived units that we will be using is what? Also give the formula. For example, imagine a piece of an unknown metallic element that has a volume of 5.0 cm3 and a mass of 13.5 g. Find the density of the unknown element. What is the identity of the unknown element? A 147-g piece of metal has a density of 7.00 g/mL. A 50-mL graduated cylinder contains 20.0 mL of water. What is the final volume after the metal is added to the graduated cylinder? What is the volume of a sample that has a mass of 20 g and a density of 4g/mL? When a piece of aluminum is placed in a 25-mL graduated cylinder that contains 10.5 mL of water, the water level rises to 13.5 mL. What is the mass of the aluminum? Given the density of aluminum is 2.7 g/mL. 4 Scientific Notation Scientific notation: is a method of representing very _______ or very _____ numbers in the form “M” is called the __________ and is a number between _________ and_________ “n” is called the __________ and is either __________ or ___________ Standard Form: is when a number is written all the way out. Example:460,000,000,000,000,000,000,000 Scientific Notation: is when a numbers is written in the form M x 10n . Example 4.6 x 1023 Scientific Notation the EASY way • • • • • If number is larger than 1 your exponent will be positive #>1 • If your number is smaller than 1 your exponent will be negative. #<1 Then move your decimal to make it “happen” Step 1: Insert an understood decimal point Step 2: Decide where the decimal must end up so that only 1 number is to the left Step 3: Count how many places you move the decimal point Step 4: Rewrite in the form M x 10n 2,500,000,000 38,000 0.0000579 0.00602 Express each number in scientific notation 4,500,000 685,000,000,000 0.0054 0.0000000008 Express each quantity in regular notation along with its appropriate unit 3.6 x 105s 5.4 x 10-5 g/cm3 Write the following data in scientific notation. The diameter of the Sun is 1,392,000 km. 3.060 x 103 km 8.9 x 1010 Hz Write the following data in scientific notation. The density of the Sun’s lower atmosphere is 0.000000028 g/cm3. 5 Using Scientific Numbers with your calculator (5.63 × 10-5) + (2.32 × 10-4) (9 × 102) - (7 × 102) (2.23 × 10-5) x (3.43 × 10-4) (5.43× 105) ÷ (9.32 × 10-4) Dimensional Analysis/ CONVERSION FACTOR Dimensional analysis: is a systematic approach to _________ solving that uses ___________ factors to move, or convert, from one unit to another. Conversion Factor: Examples of conversion factors: 1 week = days 1 year = days 1 foot = 1 dozen = inches eggs A conversion factor MUST: Steps to solving conversion factor problems: a) Always take what is given and create a ____________. Don’t forget_____________ b) Write a new conversion factor that will __________ the original units with another one. c) Continue to do this until you are left with the ________________ you want Convert 35 days to weeks Convert 672 cm to inches (1in=2.540cm) Convert 1,892 ml to quarts (1 liter =1.06 qt) In ancient Egypt, small distances were measured in Egyptian cubits. An Egyptian cubit was equal to 7 palms, and 1 palm was equal to 4 fingers. If 1 finger was equal to 18.75 mm, convert 6 Egyptian cubits to meters. Write the conversion factors needed to determine the number of seconds in one year. How many seconds in 5 days? How many pizzas do you need to order if 32 people will attend a party, each person eats 3 slices of pizza, and each pizza has 8 slices? 6 CONVERSION FACTOR Distance/ Length Mass 1 Meter = 100 cm = 1000mm = .01 km 1 Gram = .001 kg = 1000 mg 12 inches 1 foot 16 ounces 1 pound 3 feet 1 yard 2000 pounds 1 ton 36 inches 1 yard 1 Newton 100 grams 1760 yards 1 mile 1 pound 454 grams 1 mile 1.61 kilometers 2.20 pounds 1 kilogram 1 inch 2.54 centimeters 1 kilogram 1000 grams 1 kilometer 1000 meters 5280 feet 1 mile Volume Time 1 Liter = 1000 ml = .01 kl 1.06 quarts 1 liter 1 gallon 3.78 liters 2 pints 1 quart 42 gallons 1 barrel 4 quarts 1 gallon 1000 Liters 1 kiloliter 1 year 1 week 1 day 1 hour 1 minute 1 decade 1 century 52 weeks 7 days 24 hours 60 minutes 60 seconds 10 years 100 years Temperature Formulas °𝐶 = (°𝐹 – 32) ∙ 5 9 °𝐹 = 9 5 °𝐶 + 32 (9/5 = 1.8) 𝐾 = °𝐶 + 273 7 Lesson 3: Uncertainty in Data New vocabulary accuracy precision error percent error significant figure accuracy precision error percent error is the __________ of an ________ to an _____________. significant figure Rules for significant figures • Rule 1: Nonzero numbers are always significant. • Rule 2: Zeros between nonzero numbers are always significant. • Rule 3: All final zeros to the right of the decimal are significant. • Rule 4: Placeholder zeros are not significant. To remove placeholder zeros, rewrite the number in scientific notation. • Rule 5: Counting numbers and defined constants have an infinite number of significant figures. Problem Determine the number of significant figures in the following masses. a. 0.00040230 g b. 405,000 kg c. 72.3 g d. 6.20 g e. 0.0253 g f. 4320 g Lesson 4: Representing Data New vocabulary graph graph independent variable dependent variable interpolation extrapolation independent variable dependent variable interpolation extrapolation 8 9