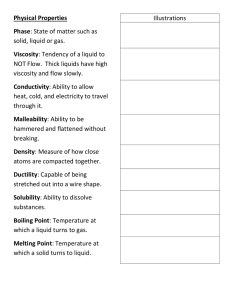
Viscosity Definition The viscosity of a fluid is a measure of its resistance to deformation at a given rate. For liquids, it corresponds to the informal concept of "thickness": for example, syrup has a higher viscosity than water INTRODUCTION Viscosity is a fundamental characteristic property of all liquids. When a liquid flows, it has an internal resistance to flow. Viscosity is a measure of this resistance to flow or shear. Viscosity can also be termed as a drag force and is a measure of the frictional properties of the fluid. Viscosity is a function of temperature and pressure. Although the viscosities of both liquids and gases change with temperature and pressure, they affect the viscosity in a different manner. In this book, we will deal primarily with viscosity of liquids and its change as a function of temperature. Viscosity is expressed in two distinct forms: a. Absolute or dynamic viscosity b. Kinematic viscosity Dynamic viscosity is the tangential force per unit area required to slide one layer (A) against another layer (B) as shown in Figure 1.1 when the two layers are maintained at a unit distance. In Figure 1.1, force F causes layers A and B to slide at velocities v1 and v2, respectively. Since the viscosity of a fluid is defined as the measure of how resistive the fluid is to flow, in mathematical form, it can be described as: Shear stress = η (Strain or shear rate) where η is the dynamic viscosity viscosity history Viscosity is a measurable quantity that denotes a liquid's thickness. A relatively thin liquid, such as water, has lower viscosity than a thicker liquid, such as honey or oil. The measurement was discovered by French physicist Jean Léonard Marie Poiseuille. Today, it is measured by the metric system in units of poise -- or poiseuille -- in honor of the physicist. Biography Born in Paris in 1799, Poiseuille began studying physics at university École Polytechnique in 1815, but left when the school closed the next year. He switched to medicine and his 1828 dissertation featured the invention of a device called the Utube mercury manometer, or hemodynamometer. It was used to measure the blood pressure of dogs and horses, and used in medical schools until the 1960s. Poiseuille focused on blood flow throughout the remainder of his career. The Discovery Poiseuille continued to focus on blood flow when he began as a practitioner in 1829. He devised an apparatus made of glass tubes that could be heated and cooled to experiment with liquids of varying thickness. He discovered that tube pressure, temperature, diameter and length all affected viscosity. He discovered an equation -now called Poiseuille's law -- to derive viscosity from all four factors. The equation can be used to determine the viscosity of everything from human blood to molten lava Measurement Viscosity is measured with various types of viscometers and rheometers. A rheometer is used for those fluids that cannot be defined by a single value of viscosity and therefore require more parameters to be set and measured than is the case for a viscometer. Close temperature control of the fluid is essential to acquire accurate measurements, particularly in materials like lubricants, whose viscosity can double with a change of only 5 °C.[21] For some fluids, the viscosity is constant over a wide range of shear rates (Newtonian fluids). The fluids without a constant viscosity (non-Newtonian fluids) cannot be described by a single number. Non-Newtonian fluids exhibit a variety of different correlations between shear stress and shear rate. One of the most common instruments for measuring kinematic viscosity is the glass capillary viscometer. In coating industries, viscosity may be measured with a cup in which the efflux time is measured. There are several sorts of cup – such as the Zahn cup and the Ford viscosity cup – with the usage of each type varying mainly according to the industry. The efflux time can also be converted to kinematic viscosities (centistokes, cSt) through the conversion equations. Also used in coatings, a Stormer viscometer uses loadbased rotation in order to determine viscosity. The viscosity is reported in Krebs units (KU), which are unique to Stormer viscometers. Newtonian and non-Newtonian fluids In solids Measurement Vibrating viscometers can also be used to measure viscosity. Resonant, or vibrational viscometers work by creating shear waves within the liquid. In this method, the sensor is submerged in the fluid and is made to resonate at a specific frequency. As the surface of the sensor shears through the liquid, energy is lost due to its viscosity. This dissipated energy is then measured and converted into a viscosity reading. A higher viscosity causes a greater loss of energy. Extensional viscosity can be measured with various rheometers that apply extensional stress. Volume viscosity can be measured with an acoustic rheometer. Apparent viscosity is a calculation derived from tests performed on drilling fluid used in oil or gas well development. These calculations and tests help engineers develop and maintain the properties of the drilling fluid to the specifications required. Causes of viscosity The viscosity arises from the forces of friction between the layers of the liquid while they are moving to each other (caused by the presence of gravitational forces (cohesion) between the fluid particles causing internal friction), and this effect is weak in solutions with low viscosity such as ethyl alcohol and water with a smooth (rapid) flow. Other solutions, such as bee honey or motor oils of high viscosity, will have a very slow flow. ❒ It can also be said that the friction of adjacent layers in glycerine is greater than friction of adjacent layers in water, and this is why the speed of flow of the two glycerines is less than the speed of water flow and the glycerine becomes more viscous than water. ❒ And if we assume that we put a liquid in a narrow tube and we affected it with some force, such as a difference in pressure between the ends of the tube, the fluid begins to flow into the tube, but the speed with which the layers of this fluid move varies from one layer to another, so the thin layer that touches the wall of the tube is still Roughly, the velocity of movement of the layers increases as we move away from the wall of the tube, and this speed reaches its maximum in the middle layers of the fluid, and then gradually decreases until it reaches the opposite wall of the tube (see the following figure) This friction between the layers of the liquid resists movement and leads to viscosity phenomenon Mathematical laws related to viscosity ❒ It has been found from scientific experiments that the friction forces (F) that counteract the relative motion as two adjacent layers are necessary in order for there to remain a constant difference from the velocity of the two successive layers proportionally proportional to the surface area of the contact surface between the two layers of liquid. Fα S Where (S) is the area of the contact surface and is proportional to the rate of change of velocity: dV / dxFα Where (V) is the velocity and (X) is the distance separating the two layers. By combining the previous two relationships, we find that: Where η is constant and is known as the viscosity coefficient. Friction forces = viscosity coefficient x area of contact surface x (velocity over area) Coefficient of viscosity and its units of measurement Definition of coefficient of viscosity (or viscosity) It is the force in dyne that affects between two parallel layers of liquid, the area of each section (1 cm2) and distance (1 cm2) from each other in order to keep the flow velocity of one layer relative to the other at a speed of (1 cm / s) The relationship between the viscosity coefficient and the liquid volume that passes through a fine (capillary) tube of diameter (r) and length (L) in time (t) per second under pressure (P) is given by a relationship of Poisellieh parallel When measuring the viscosity in the direct way using a parallel relationship where an Ostwald device called a viscometer is used, a modification can be made in a parallelic equation for such a purpose, where the derived force (hydrostatic pressure) present in a parallelic equation is replaced by (ghd) Viscosity factor units Factors affecting viscosity temperature: ❒ The viscosity decreases with increasing temperature, because the increase in temperature increases the movement of particles, so the gravitational forces between the particles decrease relatively, so if the attraction decreases, the viscosity decreases. Therefore car drivers are advised to use high viscosity oils in summer. And little viscosity in winter. It was found that the viscosity of the liquid decreased by (1-2%) for each rise in temperature by (1 C). the forces of attraction The forces of attraction between molecules are an initial measure of the viscosity of liquids, the greater the attraction between the molecules, the more difficult the movement of the particles becomes, and therefore the viscosity of the liquid increases. For example, fluids with polar molecules are more viscous. Thus, liquids whose molecules are characterized by high gravitational forces of high viscosity. The presence of dissolved substances The solute substances in the liquid affect the viscosity. For example, the presence of sugar in the water increases the viscosity of the water, while the presence of ionic salts in the water reduces the viscosity of the water. And the presence of suspended substances in the liquid increases its viscosity, for example blood is higher viscosity than water because of the presence of proteins and blood cells suspended in it. pressure As the pressure on the liquid increases, the gravitational forces increase between the fluid particles, and thus the viscosity increases somewhat. Form and structure of the molecule The shape and composition of the fluid particles play a large role in the amount of its viscosity. Liquids with large and irregular molecules (whose shape is constraining their movement) are more viscous than identical small molecules. How to measure viscosity There are two ways to measure: (1) Viscosity is usually measured by measuring the distance traveled by an iron ball during a specific time in a column of liquid, from which the velocity that is inversely proportional to the viscosity is measured (2) Or by measuring the distance a liquid travels during a certain time in a column containing this fluid (Figure B), and this experiment we will apply in the lab to determine the viscosity of some liquids. Discussion Why are liquids more viscous than others? Since the viscosity depend on the liquid molecules whether the liquid molecules connected to each strong liquid be less able to move if using a large ball is the viscosity will be change why the viscosity change if we change the temperature only if the temp. increased what will happen about the dynamic viscosity ? the distance between molecules increases so viscosity incresses