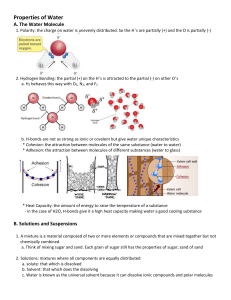
First Exercise: (7 points) Organic compounds The below list represents different chemical compounds. Observe them then answer the following questions: H2O, C2H6O, C3H6, CO2, CH3NH2, C4H10 1. Classify the compounds given above according to the table below: (Copy the table on your answer sheet). Organic compounds Hydrocarbons Non-hydrocarbons Inorganic compounds 2. Specify the families to which the above hydrocarbons belong and give the name of each. 3. Write the balanced chemical equation of the combustion reaction of C3H6. 4. C3H6 had two possible structural isomers, an open chain and a closed one. 4.1- Show the condensed structural formulas of the two possible structures of C3H6. 4.2- Give their names. 5. Name the following structures. a- b- c- 1 Second Exercise: (7 points) Alkanes: variation of boiling point temperature Refer to document 1 to answer the following questions: 1. Specify the class of compound (A) 2. Write all possible condensed structural formulas of compound (A) and give the IUPAC name of each. 3. Give the relation between these compounds (A) The boiling point of organic compounds having the same number of carbon atoms but differ in their structure increase when the number of branches increases. 4. Arrange the compounds of part 2 in increasing order of their boiling point Refer to document 2 to answer the following questions: 7. Choose the correct answer. Justify your choice. The boiling point of decane is: a. Equal to 130ºC b. Less than 130ºC c. Greater than 130ºC 150 boiling point of an alkane ºc 5. Give the boiling point of compound (A) 6. Show whether the following statement is true or false: “As the number of carbon atoms in a molecule of a straight chain alkane increases the boiling point temperature of the alkane decreases”. Document 1 100 50 0 1 2 3 4 5 6 7 8 -50 -100 -150 -200 Number of carbon atom in a molecule of an alkane Document 2 2 Third Exercise: (6 points) Acids and Bases Acids are usually corrosive and bases are usually used in making soap and cleaning detergents. Acids and bases react in a reaction to form water and salt. The structure of the four molecules that interferes in such a reaction are shown below. A B C D 1- Referring to the preceding structureS: 1.1- Identify the acidic substance 1.2- Identify the basic substance 1.3- Identify the neutral substances 2- List three properties of acids and three properties of bases. 3-What is the name of the reaction that forms when acids and bases react to form water and salt. 4- Sort the above compounds to show the reaction that forms when acids and bases react. Balance it if needed. Remark: you have to show their chemical formula. + + 5- Design an experiment that will enable us to identify the pH of acids, bases and salts. 3