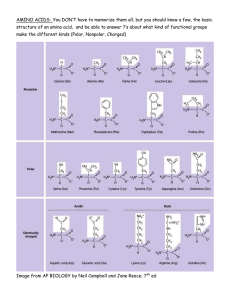
Theramine Innovative Pain Solutions Theramine® for the dietary management of pain syndromes Non-Addictive No Reported GI Bleeds Improve Pain Perception Over 38 million Individual Doses Administered without a Reported GI Bleed 1 Improve Clinical Outcomes Percent Reduction in CRP Levels * Reduction of Chronic Back Pain* 14 67.86 12 60 p<0.01 n=127 % Change 40 20 0 -20 Roland Morris Index 80 10 p<0.001 n=111 8 Before 6 After 4 -40 2 -60 -64.79 -80 Naproxen Theramine 0 Naproxen Theramine Double Blind Randomized Control Study - 28 days of therapy In this double blind multicenter trial of 127 subjects with chronic established back pain, a statistically relevant decrease in CRP was measured among subjects taking Theramine compared to those taking naprosyn (250mg) once daily.* In a 28 day double blind multicenter trial of 127 subjects with chronic established back pain, subjects taking Theramine experienced a 76% reduction in pain compared to once daily naprosyn (250mg)1 as measured by the Roland Morris Index.* *Shell, et al. Theramine and naproxen double blind clinical trial American Journal of Therapeutics; 2012 108-114 Safety Information Theramine® is contraindicated in an extremely small number of patients with hypersensitivity to any of the nutritional components of Theramine. ADVERSE REACTIONS: Ingestion of L-Tryptophan, L-Arginine, or Choline at high doses of up to 15 grams daily is generally well tolerated. The most common adverse reactions of higher doses — from 15 to 30 grams daily — are nausea, abdominal cramps, and diarrhea. Theramine contains less than 1 gram per dose of amino acids however, some patients may experience these symptoms at lower doses. The total combined amount of amino acids in each Theramine capsule does not exceed 300 mg. DRUG INTERACTIONS: Theramine Theramine may allow for lowering the dose of co-administered drugs under physician supervision. Theramine® and Targeted Cellular Technology TM Theramine is driven by Targeted Cellular Technology 1 Neurotransmitter Precursors Choline bitartrate, L-arginine, L-glutamine, L-histidine, 5-HTP, L-serine 2 Neuron Uptake Stimulator Cinnamon, Whey Protein Isolate 3 Neuron Activator Glutamine 4 Adenosine Antagonist Metabromine 5 Attenuation Inhibitor Grape-seed extract Amino Acids, Biogenic Amines, & Other Nutrients Rx Medical Food Per Recommended Dose** (Theramine) Gamma Aminobutyric Acid (GABA) 200 mg Choline Bitartrate (Choline Bitartrate from natural L(+)-tartaric acid) 125 mg Whey Protein Isolate (90%+ protein by weight) 75 mg L-Arginine HCL (Produced from plant derived materials) 75 mg L-Histidine HCL (Produced from plant derived materials) 50 mg L-Glutamine (Produced from plant derived materials) 50 mg (seed) (95% 5-HTP) (Standardized Extract) 32 mg L-Serine (Produced from plant derived materials) 25 mg Cocoa Extract (fruit) (6% theobromine) (Standardized Extract) 50 mg Grape Seed Extract (85% polyphenols) (Standardized Extract) 25 mg Cinnamon (bark) (Botanical Powder) 25 mg * Ingredient claims are based on current market availability. Actual claims may vary. Individual results, including those for microbiology, pesticides, and heavy metals including arsenic, cadmium, lead, and mercury are available to practitioners upon request. If you would like to learn more or provide feedback please email help@ptlcentral.com Theramine should be taken without food to increase the absorption of key ingredients. Theramine ® /ŶĚŝĐĂƟŽŶ͗ Theramine is indicated for the dietary management of pain syndromes that include chronic pain, ĮďƌŽŵLJĂůŐŝĂ͕ŶĞƵƌŽƉĂƚŚŝĐƉĂŝŶ͕ĂŶĚŝŶŇĂŵŵĂƚŽƌLJƉĂŝŶ͘Theramine is an Rx only GRAS/GRAE ŵĞĚŝĐĂůĨŽŽĚ͘ WŚĂƌŵĂĐŽĚLJŶĂŵŝĐƐ͗ dŚĞƉŚĂƌŵĂĐŽĚLJŶĂŵŝĐƉƌŽƉĞƌƟĞƐŽĨ TheramineĂƌĞĚŝƌĞĐƚůLJƌĞůĂƚĞĚƚŽƚŚĞĞīĞĐƚƐŽĨƚŚĞĂŵŝŶŽ ĂĐŝĚƉƌĞĐƵƌƐŽƌƐŽŶŶĞƵƌŽƚƌĂŶƐŵŝƩĞƌĂĐƟǀŝƚLJǁŚŝĐŚĂƌĞƌĞƐƉŽŶƐŝďůĞĨŽƌƚŚĞŝŶĚƵĐƟŽŶ͕ ĂŵƉůŝĮĐĂƟŽŶ͕ĂŶĚŵŝƟŐĂƟŽŶŽĨƉĂŝŶ͘ ŽƐŝŶŐ͗ The recommended dose of Theramine ŝƐϭŽƌϮĐĂƉƐƵůĞƐ͕ƚĂŬĞŶϭƚŽϰƟŵĞƐĚĂŝůLJĂƐĚŝƌĞĐƚĞĚďLJ ĂƉŚLJƐŝĐŝĂŶ͘ /ŶŐƌĞĚŝĞŶƚƐ͗ '͕ŚŽůŝŶĞŝƚĂƌƚƌĂƚĞ͕>ͲĂƌŐŝŶŝŶĞ͕tŚĞLJƉƌŽƚĞŝŶŚLJĚƌŽůLJƐĂƚĞ͕>ͲŚŝƐƟĚŝŶĞ͕>ͲŐůƵƚĂŵŝŶĞ͕DĞƚĂďƌŽŵŝŶĞ͕ ϱͲ,LJĚƌŽdžLJƚƌLJƉƚŽƉŚĂŶ͕'ƌĂƉĞ^ĞĞĚdžƚƌĂĐƚ͕>Ͳ^ĞƌŝŶĞ͕ŝŶŶĂŵŽŶďĂƌŬ͘ WƌĞĐĂƵƟŽŶƐĂŶĚŽŶƚƌĂŝŶĚŝĐĂƟŽŶƐ͗ Theramine ŝƐĐŽŶƚƌĂŝŶĚŝĐĂƚĞĚŝŶĂŶĞdžƚƌĞŵĞůLJƐŵĂůůŶƵŵďĞƌŽĨƉĂƟĞŶƚƐǁŝƚŚŚLJƉĞƌƐĞŶƐŝƟǀŝƚLJƚŽĂŶLJŽĨƚŚĞŶƵƚƌŝƟŽŶĂů components of Theramine͘ ADVERSE REACTIONS /ŶŐĞƐƟŽŶŽĨ>ͲdƌLJƉƚŽƉŚĂŶ͕>ͲƌŐŝŶŝŶĞ͕ŽƌŚŽůŝŶĞĂƚŚŝŐŚĚŽƐĞƐŽĨƵƉƚŽϭϱŐƌĂŵƐĚĂŝůLJŝƐŐĞŶĞƌĂůůLJǁĞůůƚŽůĞƌĂƚĞĚ͘dŚĞ ŵŽƐƚĐŽŵŵŽŶĂĚǀĞƌƐĞƌĞĂĐƟŽŶƐŽĨŚŝŐŚĞƌĚŽƐĞƐͶĨƌŽŵϭϱƚŽϯϬŐƌĂŵƐĚĂŝůLJͶĂƌĞŶĂƵƐĞĂ͕ĂďĚŽŵŝŶĂůĐƌĂŵƉƐ͕ĂŶĚ ĚŝĂƌƌŚĞĂ͘Theramine ĐŽŶƚĂŝŶƐůĞƐƐƚŚĂŶϭŐƌĂŵƉĞƌĚŽƐĞŽĨĂŵŝŶŽĂĐŝĚƐŚŽǁĞǀĞƌ͕ƐŽŵĞƉĂƟĞŶƚƐŵĂLJĞdžƉĞƌŝĞŶĐĞƚŚĞƐĞ ƐLJŵƉƚŽŵƐĂƚůŽǁĞƌĚŽƐĞƐ͘dŚĞƚŽƚĂůĐŽŵďŝŶĞĚĂŵŽƵŶƚŽĨĂŵŝŶŽĂĐŝĚƐŝŶĞĂĐŚdŚĞƌĂŵŝŶĞĐĂƉƐƵůĞĚŽĞƐŶŽƚĞdžĐĞĞĚϯϬϬŵŐ͘ DRUG INTERACTIONS Theramine ĚŽĞƐŶŽƚĚŝƌĞĐƚůLJŝŶŇƵĞŶĐĞƚŚĞƉŚĂƌŵĂĐŽŬŝŶĞƟĐƐŽĨƉƌĞƐĐƌŝƉƟŽŶĚƌƵŐƐ͘ůŝŶŝĐĂůĞdžƉĞƌŝĞŶĐĞŚĂƐƐŚŽǁŶƚŚĂƚ ĂĚŵŝŶŝƐƚƌĂƟŽŶŽĨTheramine ŵĂLJĂůůŽǁĨŽƌůŽǁĞƌŝŶŐƚŚĞĚŽƐĞŽĨĐŽͲĂĚŵŝŶŝƐƚĞƌĞĚĚƌƵŐƐƵŶĚĞƌƉŚLJƐŝĐŝĂŶƐƵƉĞƌǀŝƐŝŽŶ͘ OVERDOSE dŚĞƌĞŝƐĂŶĞŐůŝŐŝďůĞƌŝƐŬŽĨŽǀĞƌĚŽƐĞǁŝƚŚTheramine as the total amount of amino acids in a one month supply ;ϵϬĐĂƉƐƵůĞƐͿŝƐůĞƐƐƚŚĂŶϯϬŐƌĂŵƐ͘KǀĞƌĚŽƐĞƐLJŵƉƚŽŵƐŵĂLJŝŶĐůƵĚĞĚŝĂƌƌŚĞĂ͕ǁĞĂŬŶĞƐƐ͕ĂŶĚŶĂƵƐĞĂ͘ For more information, please visit www.ptlcentral.com Copyright © 2012 Physician Therapeutics®, a division of Targeted Medical Pharma Inc. All Rights Reserved. Ref Booklet 1 April 2012 Theramine ® ŵŝŶŽĐŝĚƐ͕ŝŽŐĞŶŝĐŵŝŶĞƐ͕ĂŶĚKƚŚĞƌEƵƚƌŝĞŶƚƐ ƉĞƌĂƉƐƵůĞ;нͬͲϭϬйͿ Gamma Aminobutyric Acid (GABA) 100 mg Choline Bitartrate (Choline Bitartrate from natural L(+)-tartaric acid) 62.5 mg Whey Protein Isolate (90%+ protein by weight) 37.5 mg L-Arginine HCL (Produced from plant derived materials) 37.5 mg >Ͳ,ŝƐƟĚŝŶĞ,> (Produced from plant derived materials) 25 mg L-Glutamine (Produced from plant derived materials) 25 mg 'ƌŝīŽŶŝĂdžƚƌĂĐƚ'ƌŝīŽŶŝĂ^ŝŵƉůŝĐŝĨŽůŝĂ(seed) (95% 5-HTP) (Standardized extract) 16 mg L-Serine (Produced from plant derived materials) 12.5 mg ĚĚŝƟŽŶĂůŽƚĂŶŝĐĂůƐ ƉĞƌĂƉƐƵůĞ;нͬͲϭϬйͿ Cocoa Extract dŚĞŽďƌŽŵĂĐĂĐĂŽ (fruit) (6% theobromine) (Standardized extract) 25 mg Grape Seed Extract sŝƚƵƐǀŝŶŝĨĞƌĂ (85% polyphenols) (Standardized extract) 12.5 mg Cinnamon ŝŶŶĂŵŽŵƵŵ͘ĐĂƐƐŝĂ (bark) (Botanical powder) 12.5 mg *Ingredient claims are based on current market availability. Actual claims may vary. Individual results, ŝŶĐůƵĚŝŶŐƚŚŽƐĞĨŽƌŵŝĐƌŽďŝŽůŽŐLJ͕ƉĞƐƟĐŝĚĞƐ͕ĂŶĚŚĞĂǀLJŵĞƚĂůƐŝŶĐůƵĚŝŶŐĂƌƐĞŶŝĐ͕ĐĂĚŵŝƵŵ͕ůĞĂĚ͕ĂŶĚ ŵĞƌĐƵƌLJĂƌĞĂǀĂŝůĂďůĞƚŽƉƌĂĐƟƟŽŶĞƌƐƵƉŽŶƌĞƋƵĞƐƚ͘/ĨLJŽƵǁŽƵůĚůŝŬĞƚŽůĞĂƌŶŵŽƌĞŽƌƉƌŽǀŝĚĞĨĞĞĚďĂĐŬ please email help@ptlcentral.com For more information, please visit www.ptlcentral.com Copyright © 2012 Physician Therapeutics®, a division of Targeted Medical Pharma Inc. All Rights Reserved. Ref Booklet 1 April 2012 www.ptlcentral.com ® 1 Acute Pain Chronic Pain Fibromyalgia Neuropathic Pain Inflammatory Pain www.ptlcentral.com Recommended R d d Dosing: D i 2 capsules b.i.d. • • • • • Theramine® is intended for the dietary management of pain syndromes that include: Indication & Dosing 2 1. 2. www.ptlcentral.com RussellIJ,MichalekJE,VipraioGA,FletcherEM,WallK.Serumaminoacidsinfibrositis/fibromyalgiasyndrome.JRheumatolSuppl1989;19:158Ͳ163 Shelletal.TheramineandIbuprofendoubleblindclinicaltrial,2010.Publicationpending. 3 In a double blind, multicenter trial, subjects with pain syndromes showed decreased levels of the amino acids required for production of pain modulating neurotransmitters, despite having a sufficient intake of 1 protein indicating that the need for these amino acids are selectively increased in these patients. Theramine® Improves Amino Acid Levels www.ptlcentral.com 1.Shell,etal.Theramineandnaproxendoubleblindclinicaltrial;AmericanJournalofTherapeutics;2012108Ͳ114 4 In a 28 day double blind, randomized, controlled, multicenter trial of 127 subjects with chronic established back pain, subjects taking Theramine experienced a 76% reduction in pain compared to 1 once daily naprosyn (250mg) Theramine® can Significantly Reduce Pain www.ptlcentral.com 1.Shell,etal.TheramineandnaproxendoubleblindclinicaltrialAmericanJournalofTherapeutics;2012108Ͳ114 Data from a 2009, 28 day double blind randomized controlled trial of 127 subjects with chronic established back pain shows that a low dose NSAID can be effective when co-administered with 1 Theramine. Effective as a Standalone Medication and as an Adjunct to a Low Dose NSAID Theramine® 5 www.ptlcentral.com 1. Shell, et al. Theramine and naproxen double blind clinical trial American Journal of Therapeutics; 2012 108-114 In this double blind multicenter trial of 127 subjects with chronic established back pain, a marked decrease in CRP was measured among subjects taking Theramine compared to subjects taking 1 naprosyn (250mg) once daily daily. 6 Theramine® is Effective at Reducing Inflammation 1. www.ptlcentral.com Shell et al. Theramine and Ibuprofen double blind clinical trial, 2010. Publication pending. In this double blind multicenter trial of 122 subjects with chronic established back pain, a marked decrease in CRP was measured among subjects taking Theramine compared to subjects taking 1 Ibuprofen(400mg) once daily daily. 7 Theramine® is Effective at Reducing Inflammation 1. www.ptlcentral.com Shell et al. Theramine and Ibuprofen double blind clinical trial, 2010. Publication pending. In this double blind multicenter trial of 122 subjects with chronic established back pain, a marked decrease in IL-6 was measured among subjects taking Theramine compared to subjects taking 1 Ibuprofen(400mg) once daily daily. 8 Theramine® is Effective at Reducing Inflammation * administrations is defined as number of pills sold since 2004 www.ptlcentral.com 1. Shell, et al. American Journal of Therapeutics; 2012 108-114 ; Theramine ibuprofen trial 2010, Unpublished. • Reduces inflammation1 • Reduces chronic back pain1 Effective Non-Addictive Pain Medication • No reported adverse CV effects • No reported GI bleeds No reported side effects in over 38 million administrations administrations* Why is Theramine® a Good Choice? 9 www.ptlcentral.com 1. Shell, et al. American Journal of Therapeutics; 2012 108-114 ; Theramine ibuprofen trial 2010, Unpublished. • Theramine can be used as a replacement therapy 10 • Theramine can be used as an adjunct to a low dose opiate Replace or Reduce Narcotic Pain Meds • Patients taking g Theramine can take a veryy low dose NSAID without loss of efficacy1 • Patients unable to take NSAIDs ( High BP BP, Over 65 65, CVD CVD, Taking Aspirin) Contraindicated Replace or Augment NSAID Therapies Clinical Applications of Theramine® Pain syndromes increase metabolic demand and the usual rate of synthesis is no longer sufficient and nonessential nutrients become conditionally essential, requiring that supplemental amounts be consumed. consumed • www.ptlcentral.com 11 (CH3)3N+CH2CH2OHXо These patients require additional amounts of arginine, choline, GABA glutamine, GABA, glutamine histidine histidine, 5 5-hydroxytryptophan, hydroxytryptophan and serine to restore homeostasis. • Example:IncreaseddemandsforCholine areassociatedwithpaindisordersandinflammation;normal p g y y amountsofcholineproducedendogenouslyareinsufficientandmustbeadministeredenterally. Theramine is a source of amino acids, biogenic amines, and other nutrients formulated for patients with certain types of pain y syndromes. • Theramine® (Nutritional Requirements) Theramine is formulated with Targeted Cellular Technology™ (TCT) ap patented integrated g molecular system y that delivers milligram g quantities of amino acids and other ingredients to targeted cells in a time sensitive manner and in specific ratios efficiently promote neurotransmitter production. • 12 The use off Theramine Th Th i in i managementt off pain i syndromes d iis supported by experimental and clinical data which have identified specific roles for each ingredient in the mechanism of pain reduction. • www.ptlcentral.com The Pharmacodynamic properties of Theramine® are directly related to the effects of amino acid and other precursors on neurotransmitter activity which are responsible for the induction, amplification, and mitigation of pain. • Pharmacodynamics www.ptlcentral.com 13 Ingestion of L-Tryptophan, L-Arginine, or Choline at high doses of up to 15 grams daily is generally well tolerated. The most common adverse reactions of higher doses — from 15 to 30 grams daily — are nausea, abdominal cramps, diarrhea Theramine contains less than 1 gram per dose of amino acids and diarrhea. however, some patients may experience these symptoms at lower doses. The total combined amount of amino acids in each Theramine capsule does not exceed 300 mg. ADVERSE REACTIONS Theramine® is contraindicated in an extremely small number of patients with hypersensitivity to any of the nutritional components of Theramine. PRECAUTIONS AND CONTRAINDICATIONS Safety Information www.ptlcentral.com 14 There is a negligible g g risk of overdose with Theramine as the total amount of amino acids in a one month supply (90 capsules) is less than 30 grams. Overdose symptoms may include diarrhea, weakness, and nausea. OVERDOSE Theramine® does not directly influence the pharmacokinetics of prescription drugs. Clinical experience has shown that administration of Theramine may allow for lowering the dose of co-administered drugs under physician supervision. supervision DRUG INTERACTIONS Safety Information Cont. 29. 30. 31. 32. 33. 24. 25. 26. 27. 28. 19. 20. 21. 22. 23. 17. 18. 15. 16. 14. 12. 13. 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. www.ptlcentral.com 15 Fields HL, Heinricher MM, Mason P. Neurotransmitters in nociceptive modulatory circuits. Annu Rev Neurosci 1991;14:219-245. Schaible HG, Ebersberger A, Von Banchet GS. Mechanisms of pain in arthritis. Ann N Y Acad Sci 2002;966:343-354. Zimmermann M. Pathobiology of neuropathic pain. Eur J Pharmacol 2001;429:23-37. Millan MJ. The induction of pain: an integrative review. Prog Neurobiol 1999;57:1-164. Elenkov IJ, Chrousos GP. Stress hormones, proinflammatory and antiinflammatory cytokines, and autoimmunity. Ann N Y Acad Sci 2002;966:290-303. Cuninkova L, Brown SA. Peripheral circadian oscillators: interesting mechanisms and powerful tools. Ann N Y Acad Sci 2008;1129:358 2008;1129:358-370. 370. Gaillard JM. Neurochemical regulation of the states of alertness. Ann Clin Res 1985;17:175-184. McGinty D, Szymusiak R. The sleep-wake switch: A neuronal alarm clock. Nat Med 2000;6:510-511. Fuller PM, Gooley JJ, Saper CB. Neurobiology of the sleep-wake cycle: sleep architecture, circadian regulation, and regulatory feedback. J Biol Rhythms 2006;21:482-493. Turek FW, Dugovic C, Zee PC. Current understanding of the circadian clock and the clinical implications for neurological disorders. Arch Neurol 2001;58:1781-1787. Belousov AB, O'Hara BF, Denisova JV. Acetylcholine becomes the major excitatory neurotransmitter in the hypothalamus in vitro in the absence of glutamate excitation. J Neurosci 2001;21:2015-2027. Farber L, Haus U, Spath M, Drechsler S. Physiology and pathophysiology of the 5-HT3 receptor. Scand J Rheumatol Suppl 2004;2-8. Dickenson AH, Chapman V, Green GM. The pharmacology of excitatory and inhibitory amino acid-mediated acid mediated events in the transmission and modulation of pain in the spinal cord. Gen Pharmacol 1997;28:633-638. Ernberg M, Lundeberg T, Kopp S. Pain and allodynia/hyperalgesia induced by intramuscular injection of serotonin in patients with fibromyalgia and healthy individuals. Pain 2000;85:31-39. Furst DE, Manning DC. Future directions in pain management. Clin Exp Rheumatol 2001;19:S71-S76. Linderoth B, Stiller CO, Gunasekera L et al. Release of neurotransmitters in the CNS by spinal cord stimulation: survey of present state of knowledge and recent experimental studies. Stereotact Funct Neurosurg 1993;61:157-170. Hogg RC, Raggenbass M, Bertrand D. Nicotinic acetylcholine receptors: from structure to brain function. Rev Physiol Biochem Pharmacol 2003;147:1-46. Abbadie C, Brown JL, Mantyh PW, Basbaum AI. Spinal cord substance P receptor immunoreactivity increases in both inflammatory and nerve injury models of persistent pain. Neuroscience 1996;70:201-209. Thomas RJ. Excitatory amino acids in health and disease. J Am Geriatr Soc 1995;43:1279-1289. Dickenson AH. Plasticity: implications for opioid and other pharmacological interventions in specific pain states. Behav Brain Sci 1997;20:392-403. Dickenson AH. NMDA receptor antagonists: interactions with opioids. Acta Anaesthesiol Scand 1997;41:112-115. Oliverio A, Castellano C, Puglisi-Allegra S. Psychobiology of opioids. Int Rev Neurobiol 1984;25:277-337. Ono T, Inoue M, Rashid MH, Sumikawa K, Ueda H. Stimulation of peripheral nociceptor endings by low dose morphine and its signaling mechanism. Neurochem Int 2002;41:399407. Ribeiro JA,, Sebastiao AM,, de MA. Adenosine receptors p in the nervous system: y pathophysiological p p y g implications. p Prog g Neurobiol 2002;68:377-392. ; Gallowitsch-Puerta M, Pavlov VA. Neuro-immune interactions via the cholinergic anti-inflammatory pathway. Life Sci 2007;80:2325-2329. Hancock CM, Riegger-Krugh C. Modulation of pain in osteoarthritis: the role of nitric oxide. Clin J Pain 2008;24:353-365. Holthusen H, Arndt JO. Nitric oxide evokes pain at nociceptors of the paravascular tissue and veins in humans. J Physiol 1995;487 ( Pt 1):253-258. Wahl SM, McCartney-Francis N, Chan J, Dionne R, Ta L, Orenstein JM. Nitric oxide in experimental joint inflammation. Benefit or detriment? Cells Tissues Organs 2003;174:2633. Efron DT, Barbul A. Modulation of inflammation and immunity by arginine supplements. Curr Opin Clin Nutr Metab Care 1998;1:531-538. Mori M. Regulation of nitric oxide synthesis and apoptosis by arginase and arginine recycling. J Nutr 2007;137:1616S-1620S. Budzinski M,, Misterek K,, Gumulka W,, Dorociak A. Inhibition of inducible nitric oxide synthase y in p persistent p pain. Life Sci 2000;66:301-305. ; Pelligrino DA, Baughman VL, Koenig HM. Nitric oxide and the brain. Int Anesthesiol Clin 1996;34:113-132. Cerra FB. Nutrient modulation of inflammatory and immune function. Am J Surg 1991;161:230-234. Select References Visit www.ptlcentral.com For More Information American Journal of Therapeutics 0, 000–000 (2010) A Double-Blind Controlled Trial of a Single Dose Naproxen and an Amino Acid Medical Food Theramine for the Treatment of Low Back Pain William E. Shell, MD,1* Elizabeth H. Charuvastra, RN,1 Marcus A. DeWood, MD,2 Lawrence A. May, MD,2 Debora H. Bullias, BS, CRA,1 and David S. Silver, MD2 To study the safety and efficacy of a new medical food (Theramine) in the treatment of low back pain, we performed a 28-day double-blind randomized controlled trial in 129 patients. Back pain was present for at least 6 weeks and was not mild. Patients were randomly assigned to receive medical food alone (n = 43), naproxen alone (250 mg/d, n = 42), or both medical food and naproxen (n = 44). All patients were assessed by using Roland–Morris Disability Questionnaire, Oswestry Low Back Pain Scale, Visual Analog Scale Evaluation and laboratory analysis performed at baseline and at 28 days for assessing the safety and impact on inflammatory markers, which included complete blood counts, C-Reactive protein (CRP), and liver function (alkaline phosphatase, aspartate transaminase, and alanine transaminase). At baseline, there were no statistically significant differences in low back pain when assessed by Roland–Morris function or Oswestry assessments nor were there differences in the blood indices of inflammation. At day 28, both the medical food group and combined therapy group (medical food with naproxen) were statistically significantly superior to the naproxen-alone group (P , 0.05). The medical food and naproxen group showed functional improvement when compared to the naproxen-alone group. The naproxen-alone group showed significant elevations in CRP, alanine transaminase, and aspartate transaminase when compared with the other groups. Medical food alone or with naproxen showed no significant change in liver function tests or CRP, with medical food potentially mitigating the effects seen with naproxen alone. The medical food (Theramine) appeared to be effective in relieving back pain without causing any significant side effects and may provide a safe alternative to presently available therapies. Keywords: amino acid formulation, Theramine, pain, NSAIDs, C-reactive protein, naproxen, medical food, low back pain, neurotransmitter, nitric oxide INTRODUCTION A large percentage of the population will experience low back pain during their lifetime.1 Low back pain can become chronic with considerable pain and debilitation. Long-term treatment adds additional costs to the healthcare system and time out of work is frequent and costly to society.2,3 1 Physician Therapeutics, Los Angeles, CA; and 2UCLA School of Medicine, Cedars-Sinai Medical Center, Los Angeles, CA. *Address for correspondence: 2980 Beverly Glen Circle, Suite 301, Los Angeles, CA 90077. E-mail: weshell@ptlcentral.com 1075–2765 Ó 2010 Lippincott Williams & Wilkins The treatments for both acute and chronic back pain include nonsteroidal anti-inflammatory agents(NSAIDS), cetaminophen, narcotics, surgical interventions, and physical therapy.4,5 Many of the drug treatment modalities have significant side effects, including gastrointestinal (GI) hemorrhage, kidney, and heart disease. The side effects of NSAIDS are related to the magnitude and frequency of the dose.6,7 Theramine, an amino acid formulation (AAF), has been developed and is used as a prescription medical food for the clinical dietary management of the metabolic processes associated with pain and inflammation.8 The formulation is designed to increase the production of serotonin,9–11 nitric oxide (NO),12–15 www.americantherapeutics.com 2 histamine,16–18 and gamma-aminobutyric acid by providing precursors to these neurotransmitters. The neurotransmitters addressed in this formulation have well-defined and specific roles in the modulation of pain and inflammation. For example, gut serotonin alters platelet aggregation, whereas gut NO specifically reduces erosions induced by NSAIDS.19–21 The formulation contains ingredients that are generally recognized as safe (GRAS) and is regulated by the Food and Drug Administration in the medical food category.22–24 A medical food that is GRAS and effective for its intended use and that has shown the ability to allow a reduction in the dose of NSAIDS used in the treatment of back pain, thereby reducing the side effects of these agents, would be of substantial use. The purpose of this randomized double-blind controlled clinical trial was to compare the effects of the AAF with and without low-dose naproxen in a 28-day study of 129 patients with chronic low back pain. MATERIALS AND METHODS The study involved 129 patients in a 3-arm doubleblind randomized trial comparing naproxen alone (n = 42), AAF alone (n = 43), or the combined use of AAF and naproxen (n = 44). During the washout period, patients taking oral anti-inflammatory or other analgesic medicines discontinued their medication for 5 half lives before randomization. Aspirin ingestion (#325 mg/d) for nonarthritic conditions was allowed and used as a stable background drug. Only acetaminophen (650–1000 mg every 4–6 hours) was used as rescue therapy for pain but never exceeded 4 gm daily. Protocol The study was conducted at 12 sites. At each site, informed consent was obtained, screening procedures were performed, and a washout period was begun. After the washout period, there was a baseline day-1 visit. At that time, a baseline Roland–Morris Disability Questionnaire, an Oswestry Low Back Pain Scale, and a Visual Analog Scale (VAS) evaluation were obtained. In addition, blood was sampled for assessing Creactive protein (CRP), blood count, and blood chemistries. On the day-1 visit, the patients were randomized to 1 of 3 groups: (1) naproxen-alone group, which was treated for 28 days with a 2-capsule dose of an amino acid–like placebo twice daily and naproxen 250 mg/d in the morning (2) AAF-alone group, which was treated with the active AAF at a 2-capsule dose twice daily and a single naproxen-like placebo in the morning, and (3) the combined group (both AAF and American Journal of Therapeutics (2010) 0(0) Shell et al naproxen), which was treated with active AAF at a 2-capsule dose twice daily and 250 mg of an active naproxen in the morning. The active and naproxen tablets were identical, and the AAF active and placebo capsules were identical. On days 7 and 14, the evaluation of VAS and patient medication usage was completed. On day 28, a Roland–Morris Disability Questionnaire, an Oswestry Low Back Pain Scale, a VAS Evaluation, and a patient medication usage evaluation were completed. Blood was again sampled for estimating CRP, blood count, and blood chemistries. Primary endpoints The primary endpoints of the study were pain and disability as measured by the Roland–Morris pain questionnaire and the Oswestry Disability Index.25–29 Patient selection Patients were identified in 12 separate physicians’ offices. Men and nonpregnant, nonlactating women aged between 18 and 75 years were recruited for the study. To be included in the study, patients were required to have back pain lasting ,6 weeks, with pain present on 5 of 7 days during each of the 2 weeks before screening. Patients with a Roland–Morris back pain index .40 of 100 mm on the VAS were included. Finally, patients being treated with psychoactive medication were considered eligible to participate provided the dose remained stable for 3 months before study entry. Exclusion criteria Patients with surgery in the previous 6 months were excluded as were patients with neurologic impairment. Patients with fracture of the spine within the past year and patients receiving oral, intramuscular, or soft tissue injection of corticosteroids within 1 month before screening were excluded. Patients were also excluded if they had a history of GI bleeding, gastric or duodenal ulcer as were patients receiving an epidural injection within 3 months before screening. Patients were also excluded for participation in a prior clinical trial within 1 month of screening for the present study. Finally, patients who used controlled substances or opiate analgesics for .5 days in the month before screening were considered ineligible to participate. Statistical analysis The primary measure of efficacy was the change in awakening stiffness and pain scores obtained from the Roland–Morris Lower Back Pain Scale and the Oswestry Disability Index questionnaire evaluation.25–30 Scores were assigned on study entry (day 0) www.americantherapeutics.com A Double-Blind Trial for Treatment of Back Pain and at study end (day 28). Assuming that larger values are worse, a negative value for the change from baseline score indicates an improvement in the score, and positive values indicate a worsening in the score in percent. Analysis of variance was used (ANOVA) to determine statistical differences among the 3 groups on the study entry and at the completion of treatment. Statistical significance was defined as P # 0.05. An intension to treat analysis was utilized. Of the 129 patients who entered the trial, 126 completed the study. Patients who did not complete were carried forward as an intention to treat. As is shown in Table 1, none of the 3 study groups was statistically different on entry into the trial. Likewise, the laboratory responses assessed in each of the 3 study groups we measured, including CRP and hemoglobin (Hgb), alkaline phosphatase (alk phos), aspartate transaminase (AST), and alanine transaminase (ALT), were not statistically significantly different (Table 1) at baseline. CRP was chosen because it is an acute phase marker of inflammation. The liver enzymes (alk phos, AST, and ALT) were monitored to assess possible liver toxicity due to NSAIDs. Safety There were no adverse events or complications among any of the groups during this 28-day study. There were no GI side effects observed in this cohort. RESULTS 3 For example, The Roland–Morris Index if fell by 65%, and the Oswestry Disability index fell 61% between baseline and day 28 in the AAF/naproxen group. In the AAF-alone group, there was a significant reduction in back pain. Thus, if the AAF was used as either primary therapy or an adjunct to naproxen, low back pain was significantly improved. Low-dose naproxen had no appreciable effect on chronic back pain in 28 days. Similar results were seen on using the VAS scale. C-Reactive protein In the single daily dose of naproxen (Table 3), CRP rose significantly (P , 0.001). In the AAF-alone group, the CRP level fell by 16.7% (P , 0.05). In the group treated with both the AAF and single daily dose of 250 mg of naproxen, CRP fell 78.6% (P , 0.001). Laboratory measurements For participants of the study, no significant differences were found in Hgb, Alk Phos, alanine transaminase, aspartate transaminase among the 3 groups, as shown in Table 1. Throughout the study (Table 3), neither Hgb nor Alk Phos changed significantly among the groups. Both ALT and AST values rose significantly in the naproxen-alone group compared with those in the AAF alone group or the AAF/naproxen-treated cohort. Although there was no clinical deterioration evident, there was laboratory evidence of hepatocellular inflammation if naproxen was used in the absence of active AAF. DISCUSSION Significant changes were observed among the 3 groups after 28 days (Table 2). The Naproxen group remained unchanged from baseline to 28 days when assessed by either the Oswestry Low Back Pain Scale or the Roland–Morris rating scale. There were significant differences in pain reduction in both the AAF-alone group and the amino acid/naproxen treated groups. The data in this study indicate that addressing the dietary management of pain syndromes could allow for the dose reduction of NSAIDs without affecting therapeutic efficacy. Dietary management of disease is an underappreciated option for patients, although it has been in existence for .100 years. Osler31 Table 1. Clinical characteristics at study entry. Mean 6 SD Naproxen alone (n = 42) Oswestry Disability Index Roland–Morris Pain Scale Hgb CRP Alk. Phos. ALT AST www.americantherapeutics.com 29.19 12.90 13.61 1.9 75.04 24.85 20.85 6 6 6 6 6 6 6 7.49 5.14 3.92 1.90 27.1 10.64 7.49 AAF alone (n = 43) 24.21 10.97 13.93 2.36 73.7 25.69 21.84 6 8.09 6 5.42 61.52 6 3.3 6 29.99 6 15.46 6 11.3 Both (n = 44) P value 6 6 6 6 6 6 6 NS NS NS NS NS NS NS 27.13 12.38 13.85 3.53 74.2 30.53 25.69 8.19 5.31 1.51 5.73 19.16 28.13 15.46 American Journal of Therapeutics (2010) 0(0) 4 Shell et al Table 2. Primary endpoints percent change from baseline. % Change from baseline Oswestry disability index Roland–Morris Pain index 23.4 232.94 260.47 ,0.05 2.95 244 265 ,0.05 Naproxen AAF Both P value ANOVA prominently emphasized the value of nutrition in his textbooks. Advances in science mandate inclusion of nutrient management of symptoms and disease. Because nutrient management of disease has existed since therapeutic medicine began, evidence-based examples of more modern observations would be useful. For example, Tepaske et al32 administered an arginine-based preparation to patients before cardiac surgery. The clinical outcomes were found to be improved, specifically postoperative creatine clearance and immune function. Fonarow and coworkers33 and Tepaske et al32 demonstrated that administration of amino acid neurotransmitter precursors in patients with congestive heart failure improved clinical outcomes. These are 2 examples of recent observations of the importance of nutrient management of disease. The AAF of neurotransmitter precursors used in this study is designed to elicit neurotransmitter production. The amino acid precursors support the production of neurotransmitters that modulate pain and inflammation. The precursors of serotonin, NO, histamine, and gamma-aminobutyric acid are supplied in this formulation as 5-hydroxytryptophan, arginine, histidine, and glutamine, respectively. These neurotransmitters modulate nociception and inflammation.34–48 Histidine, for example, is converted to histamine, which elicits adrenocorticotropic hormone/cortisol release.49,50 In this study, a single daily dose of 250 mg of naproxen had no effect on chronic back pain over 28 days, a nonsignificant 2.95% increase in the Roland– Morris Index measure of pain was found. The AAF Table 3. Toxicity data percent change from baseline. % Change from baseline CRP Naproxen 184.5 AAF 216.7 Both 278.6 P value ANOVA ,0.01 HB Alk Phos 21.6 21 21.49 ,0.01 1.68 4.75 0.51 NS American Journal of Therapeutics (2010) 0(0) ALT AST 7.4 20.24 2 1.37 214.1 9.96 ,0.05 ,0.05 alone produced a 44% reduction in the Roland–Morris Index and a 33% reduction in the Oswestry index. The AAF with 250 mg of naproxen administered once a day resulted in a 65% reduction in the Roland–Morris Index and a 61% reduction in the Oswestry Index. Back pain is a common concern, affecting up to 90% of people during their lifetime. Nonsteroidal antiinflammatory drugs are the most commonly used drugs in the treatment of pain and inflammation.51–59 However, their use is limited by adverse drug side effects notably GI toxicity.6,60,61–64 The adverse effects of NSAIDS are dose related.65–72 The current advice of the American Geriatrics Society is to restrict or even eliminate NSAIDS in older people. This demographic with the highest incidence of osteoarthritis, back pain, and spinal stenosis is at greatest risk for adverse events. For many of these patients, the only alternative to NSAIDS may be addictive narcotics. The study included 129 patients from 12 sites. The differences in the data were highly statistically significant, but the subjects were limited to 129 patients. Because the ingredients of the AAF are GRAS, a large safety trial would appear to be unnecessary. The single daily dose of naproxen is unlikely to cause liver or kidney damage. Whether the low dose of naproxen would be cardioprotective or whether the low dose of naproxen combined with the AAF would reduce the incidence of GI side effects was not examined. It is interesting to note that tryptophan induces an increase in platelet aggregability, and NO production in the GI tract is known to reduce NSAID-induced mucosal erosion. Anti-inflammatory nonsteroidal drugs with NOproducing precursors attached (NO–NSAIDs) are a new class of drugs.73–77 These compounds have been shown to retain the anti-inflammatory, analgesic, and antipyretic activity with reduced GI toxicity.20,73,78 The use of an NO moiety with an NSAID has been shown in studies to inhibit in vitro T-cell proliferation79 and cytokine production.79 Moreover, NO–NSAIDS have been shown to be GI protective in several models. The AAF used in our study produces NO similarly to the NO–NSAIDS. If the reduction of inflammation and the alteration of nociception in chronic back pain syndromes seen in this study are also associated with the reduction of GI side effects associated with the NO– NSAIDS, the use of an AAF, with or without low-dose naproxen therapy may be useful in the management of back pain. A single daily dose of naproxen increased the CRP by 185%, whereas the administration of AAF reduced the CRP. The AAF administered with naproxen reversed the elevation of CRP. There is a paucity of reported data on the effects of low-dose naproxen on www.americantherapeutics.com A Double-Blind Trial for Treatment of Back Pain CRP. NSAIDs alter the prostaglandin inflammatory cascade but have little effect on other components such as cytokine release and T-cell activation. The ingredients in the AAF are defined by the Food and Drug Administration as GRAS, and in this formulation, the doses fall within the acceptable daily dose for GRAS. The study, however, is underpowered to detect any potential deleterious interaction between the amino acids and naproxen. We could only detect an event of 1 in 129 exposures. We have examined a large number of subjects exposed to the AAF and various NSAIDS, and this manuscript is in preparation. In addition, additional double-blind trials will be necessary to detect potential deleterious interactions. There are limited data, however, to indicate that the provision of neurotransmitter precursors alters the efficiency of pharmaceuticals. The data in this study indicate that the provision of amino acid precursors in a formulation to facilitate neurotransmitter production results in improving the efficiency of pharmaceutical therapy. We postulate that the mechanism is related to improving intracellular metabolic function rather than having any effect on the drug itself. This may be a new approach to a long-standing therapy. REFERENCES 1. Cassidy JD, Cote P, Carroll LJ, et al. Incidence and course of low back pain episodes in the general population. Spine. 2005;30:2817–2823. 2. Sleed M, Eccleston C, Beecham J, et al. The economic impact of chronic pain in adolescence: methodological considerations and a preliminary costs-of-illness study. Pain. 2005;119:183–190. 3. van Leeuwen MT, Blyth FM, March LM, et al. Chronic pain and reduced work effectiveness: the hidden cost to Australian employers. Eur J Pain. 2006;10:161–166. 4. McCarberg BH. Acute back pain: benefits and risks of current treatments. Curr Med Res Opin. 2010;26:179–190. 5. Last AR, Hulbert K. Chronic low back pain: evaluation and management. Am Fam Phys. 2009;79:1067–1074. 6. Hunt RH, Harper S, Watson DJ, et al. The gastrointestinal safety of the COX-2 selective inhibitor etoricoxib assessed by both endoscopy and analysis of upper gastrointestinal events. Am J Gastroent. 2003;98:1725–1733. 7. Uribe A, Johansson C, Slezak P, et al. Ulcerations of the colon associated with naproxen and acetylsalicylic acid treatment. Gastroint Endosc. 1986;32:242–244. 8. Shell W, Bullias D, Charuvastra E, et al. A randomized, placebo-controlled trial of an amino acid preparation on timing and quality of sleep. Am J Ther. 2010;17:133–139. 9. McCleane G. Antidepressants as analgesics. CNS Drugs. 2008;22:139–156. 10. Gajraj NM. Pregabalin: its pharmacology and use in pain management. Anesth Analg. 2007;105:1805–1815. www.americantherapeutics.com 5 11. Dworkin RH, O’Connor AB, Backonja M, et al. Pharmacologic management of neuropathic pain: evidence-based recommendations. Pain. 2007;132:237–251. 12. Tepper SJ, Rapoport A, Sheftell F. The pathophysiology of migraine. Neurolog. 2001;7:279–286. 13. Chevlen E. Opioids: a review. Curr Pain Headache Rep. 2003;7:15–23. 14. Goldberg SH, Von Feldt JM, Lonner JH. Pharmacologic therapy for osteoarthritis. Am J Orthop. 2002;31:673–680. 15. Dixit RK, Bhargava VK. Neurotransmitter mechanisms in gabapentin antinociception. Pharmacology. 2002;65:198–203. 16. Oluyomi AO, Hart SL. Involvement of histamine in naloxone-resistant and naloxone-sensitive models of swim stress-induced antinociception in the mouse. Neuropharmacology. 1991;30:1021–1027. 17. Russell IJ, Michalek JE, Vipraio GA, et al. Serum amino acids in fibrositis/fibromyalgia syndrome. J Rheumatol Suppl. 1989;19:158–163. 18. Gross PM. Cerebral histamine: indications for neuronal and vascular regulation. J Cereb Blood Flow Metab. 1982;2: 3–23. 19. Nemmani KV, Mali SV, Borhade N, et al. NO-NSAIDs: gastric-sparing nitric oxide-releasable prodrugs of nonsteroidal anti-inflammatory drugs. Bioorg Med Chem Lett. 2009;19:5297–5301. 20. Wallace JL. Nitric oxide, aspirin-triggered lipoxins and NO-aspirin in gastric protection. Inflamm Aller Drug Targets. 2006;5:133–137. 21. Walley M, Hotz-Behofsits C, Simpson R, et al. Nitric oxide: potential role for reducing gastro-enteropathy. Inflammopharmacology. 2003;11:429–436. 22. Morgan SL, Baggott JE. Medical foods: products for the management of chronic diseases. Nutr Rev. 2006;64: 495–501. 23. Czap A. The FDA, medical foods, and patents. Altern Med Rev. 2002;7:367–368. 24. Mueller C, Nestle M. Regulation of medical foods: toward a rational policy. Nutr Clin Pract. 1995;10:8–15. 25. Bannwarth B, Allaert FA, Avouac B, et al. A randomized, double-blind, placebo controlled triphosphate in study of oral adenosine subacute low back pain. J Rheumatol. 2005; 32:1114–1117. 26. Bess S, Boachie-Adjei O, Burton D, et al. Pain and disability determine treatment modality for older patients with adult scoliosis, while deformity guides treatment for younger patients. Spine (Phila Pa 1976). 2009;34: 2186–2190. 27. Cambron JA, Gudavalli MR, Hedeker D, et al. One-year follow-up of a randomized clinical trial comparing flexion distraction with an exercise program for chronic low-back pain. J Altern Compl Med. 2006;12:659–668. 28. Chen C, Hogg-Johnson S, Smith P. The recovery patterns of back pain among workers with compensated occupational back injuries. Occup Environ Med. 2007;64:534–540. 29. Demoulin C, Ostelo R, Knottnerus JA, et al. What factors influence the measurement properties of the RolandMorris disability questionnaire? Eur J Pain. 2010;14: 200–206. American Journal of Therapeutics (2010) 0(0) 6 30. Frost H, Lamb SE, Stewart-Brown S. Responsiveness of a patient specific outcome measure compared with the Oswestry Disability Index v2.1 and Roland and Morris Disability Questionnaire for patients with subacute and chronic low back pain. Spine (Phila Pa 1976). 2008;33: 2450–2457. 31. Osler W. The Principles and Practice of Medicine (1882). Leslie B. Adams, JR, MA, JD, UD. Delanco, NJ. Copyright 1978. 32. Tepaske R, Velthuis H, Oudemans-van Straaten HM, et al. Effect of preoperative oral immune-enhancing nutritional supplement on patients at high risk of infection after cardiac surgery: a randomised placebo-controlled trial. Lancet. 2001;358:696–701. 33. Kalantar-Zadeh K, Anker SD, Horwich TB, et al. Nutritional and anti-inflammatory interventions in chronic heart failure. Am J Cardiol. 2008;101:E89–E103. 34. Abbott FV, Young SN. The effect of tryptophan supplementation on autotomy induced by nerve lesions in rats. Pharmacol Biochem Behav. 1991;40:301–304. 35. Aghabeigi B. The pathophysiology of pain. Br Dent J. 1992;173:91–97. 36. Akunne HC, Soliman KF. Serotonin modulation of pain responsiveness in the aged rat. Pharmacol Biochem Behav. 1994;48:411–416. 37. Batmanghelidj F. Pain: a need for paradigm change. Anticancer Res. 1987;7:971–989. 38. Ceccherelli F, Costa C, Ischia S, et al. Cerebral tryptophan metabolism in humans in relation to malignant pain. Funct Neurol. 1989;4:341–353. 39. Contino MI, Bausano G. [Nutritional approach to the therapy of pain: recent findings]. Recent Prog Med. 1985; 76:472–475. 40. Delgado PL. Common pathways of depression and pain. J Clin Psychiatry. 2004;65(Suppl 12):16–19. 41. Aley KO, McCarter G, Levine JD. Nitric oxide signaling in pain and nociceptor sensitization in the rat. J Neurosci. 1998;18:7008–7014. 42. Anbar M, Gratt BM. Role of nitric oxide in the physiopathology of pain. J Pain Sympt Manag. 1997;14:225–254. 43. Devulder JE. Could nitric oxide be an important mediator in opioid tolerance and morphine side effects? J Clin Anesth. 2002;14:81–82. 44. Fiorucci S, Antonelli E, Burgaud JL, et al. Nitric oxidereleasing NSAIDs: a review of their current status. Drug Saf. 2001;24:801–811. 45. Kindgen-Milles D, Arndt JO. Nitric oxide as a chemical link in the generation of pain from veins in humans. Pain. 1996;64:139–142. 46. Bennett A. The role of biochemical mediators in peripheral nociception and bone pain. Cancer Surv. 1988;7:55–67. 47. Dray A. Inflammatory mediators of pain. Br J Anaesth. 1995;75:125–131. 48. Keele CA. The chemistry of pain production. Proc R Soc Med. 1967;60:419–422. 49. Feigelson M. Hypophyseal regulation of hepatic histidase during postnatal development and adulthood. II. Pituitary-estrogen interrelationship. Biochem Biophys Acta. 1971;230:309–318. American Journal of Therapeutics (2010) 0(0) Shell et al 50. Kansal VK, Wagle DS. Studies on the action of hormones on the intestinal transport of l-histidine. Ind J Physiol Pharmacol. 1975;19:28–33. 51. Barkin RL. Nonopiod analgesics for osteoarthritis: availability of topical NSAIDs. J Pain Palliat Care Pharmacother. 2010 ;24:180–181. 52. Chou R. Pharmacological management of low back pain. Drugs. 2010;70:387–402. 53. Bach SM, Holten KB. Guideline update: what’s the best approach to acute low back pain? J Fam Pract. 2009;58:E1. 54. Romano CL, Romano D, Bonora C, et al. Pregabalin, celecoxib, and their combination for treatment of chronic low-back pain. J Orthop Traumatol. 2009;10:185–191. 55. Hancock MJ, Maher CG, Latimer J, et al. Can predictors of response to NSAIDs be identified in patients with acute low back pain? Clin J Pain. 2009;25:659–665. 56. Kroenke K, Krebs EE, Bair MJ. Pharmacotherapy of chronic pain: a synthesis of recommendations from systematic reviews. Gen Hosp Psychiatry. 2009;31:206–219. 57. Kuritzky L. Current management of acute musculoskeletal pain in the ambulatory care setting. Am J Ther. 2008; 15(Suppl 10):S7–S11. 58. Rossignol M, Abouelfath A, Lassalle R, et al. The CADEUS study: burden of nonsteroidal anti-inflammatory drug (NSAID) utilization for musculoskeletal disorders in blue collar workers. Br J Clin Pharmacol. 2009;67:118–124. 59. Roelofs PD, Deyo RA, Koes BW, et al. Non-steroidal antiinflammatory drugs for low back pain. Cochrane Database Syst Rev. 2008;(1):CD000396. 60. Chan FK, Chung SC, Suen BY, et al. Preventing recurrent upper gastrointestinal bleeding in patients with Helicobacter pylori infection who are taking low-dose aspirin or naproxen. N Engl J Med. 2001;344:967–973. 61. Laine L, Smith R, Min K, et al. Systematic review: the lower gastrointestinal adverse effects of non-steroidal anti-inflammatory drugs. Aliment Pharmacol Ther. 2006;24: 751–767. 62. Mahajan SK, Vaidya P, Sood BR, et al. Duodenal diverticular haemorrhage in a patient taking NSAID. J Assoc Phys India. 2003;51:416–418. 63. Papich MG. Principles of analgesic drug therapy. Sem Vet Med Surg (Small Anim). 1997;12:80–93. 64. Sweet BV, Townsend KA, Tsai CY. Risk assessment of NSAID-induced gastrointestinal toxicity in ambulatory care patients. Am J Health Syst Pharm. 2004;61:1917–1921. 65. Baum K, Engels B, Vogtle-Junkert U, et al. High dose ibuprofen in low back pain. Fortschr Med. 1997;115: 51–52. 66. Becker MC, Wang TH, Wisniewski L, et al. Rationale, design, and governance of Prospective Randomized Evaluation of Celecoxib Integrated Safety versus Ibuprofen Or Naproxen (PRECISION), a cardiovascular end point trial of nonsteroidal antiinflammatory agents in patients with arthritis. Am Heart J. 2009;157:606–612. 67. Curtis SP, Ng J, Yu Q, et al. Renal effects of etoricoxib and comparator nonsteroidal anti-inflammatory drugs in controlled clinical trials. Clin Ther. 2004;26:70–83. www.americantherapeutics.com A Double-Blind Trial for Treatment of Back Pain 68. Devogelaer JP, Dreiser RL, Abadie E, et al. Guidelines for clinical studies assessing the efficacy of drugs for the management of acute low back pain. Clin Exp Rheumatol. 2003;21:691–694. 69. Gotzsche PC. Review of dose–response studies of NSAIDs in rheumatoid arthritis. Dan Med Bull. 1989;36: 395–399. 70. Katz N, Ju WD, Krupa DA, et al. Efficacy and safety of rofecoxib in patients with chronic low back pain: results from two 4-week, randomized, placebo-controlled, parallel-group, double-blind trials. Spine (Phila Pa 1976). 2003;28:851–858. 71. Lanas A. Nonsteroidal anti-inflammatory drugs, low-dose aspirin, and potential ways of reducing the risk of complications. Eur J Gastroenterol Hepatol. 2001;13:623–626. 72. Porter RS. Factors determining efficacy of NSAIDs. Drug Intell Clin Pharm. 1984;18:42–51. 73. Fiorucci S, Antonelli E. NO-NSAIDs: from inflammatory mediators to clinical readouts. Inflamm Allergy Drug Targets. 2006;5:129–131. www.americantherapeutics.com 7 74. Cirino G, Distrutti E, Wallace JL. Nitric oxide and inflammation. Inflamm Allergy Drug Targets. 2006;5: 115–119. 75. Whittle BJ. Nitric oxide-modulating agents for gastrointestinal disorders. Expert Opin Invest Drugs. 2005;14: 1347–1358. 76. Velazquez C, Praveen Rao PN, Knaus EE. Novel nonsteroidal antiinflammatory drugs possessing a nitric oxide donor diazen-1-ium-1,2-diolate moiety: design, synthesis, biological evaluation, and nitric oxide release studies. J Med Chem. 2005;48:4061–4067. 77. Whittle BJ. Cyclooxygenase and nitric oxide systems in the gut as therapeutic targets for safer anti-inflammatory drugs. Curr Opin Pharmacol. 2004;4:538–545. 78. Wallace JL. Building a better aspirin: gaseous solutions to a century-old problem. Br J Pharmacol. 2007;152:421–428. 79. Cicala C, Ianaro A, Fiorucci S, et al. NO-naproxen modulates inflammation, nociception and downregulates T cell response in rat Freund’s adjuvant arthritis. Br J Pharmacol. 2000;130:1399–1405. American Journal of Therapeutics (2010) 0(0) David S. Silver et al. / Journal of Pharmacy Research 2012,5(5),2806-2809 Research Article ISSN: 0974-6943 Available online through www.jpronline.info Theramine (A Medical Food) Versus Non-Steroidal Anti Inflammatory Agents in Elderly Patients: A Pharmacoeconomic Analysis *David S. Silver MD,Elizabeth H. Charuvastra RN,Lawrence May MD,Stephanie L. Pavlik CRA,William E. Shell MD Targeted Medical Pharma,2980 N. Beverly Glen Circle,Suite 301,Los Angeles, CA 90077, USA Received on:11-01-2012; Revised on: 17-02-2012; Accepted on:19-04-2012 ABSTRACT Non-steroidal anti-inflammatory drugs are the leading cause of drug induced morbidity and mortality in the United States. Gastrointestinal hemorrhage is the most common concern, with hypertension, edema, renal complication and cardiovascular risk other considerations. These complications occur at higher rates in elderly patients. Strategies to reduce these risks have had mixed results. Theramine, a prescription only medical food, is used to treat pain and inflammation without risk of gastrointestinal or other side effects. We undertook a pharmacoeconomic analysis of Theramine versus NSAIDs in elderly patients; specifically examining the additional cost burden of the strategies to prevent GI side effects and complications. The higher acquisition costs for Theramine are offset by the reduction in side effects and need for testing and other protective medications in patients over the age of sixty-five taking NSAIDs. Theramine should be the preferred choice over NSAIDs in elderly patients. Key words: Theramine, NSAIDs, Side effects, Cost Analysis, Elderly population, Pain management INTRODUCTION Non-steroidal anti-inflammatory drugs (NSAIDs) are a mainstay treatment of pain from a variety of inflammatory and non-inflammatory conditions. They are a recommended treatment for a wide variety of disease states such as, rheumatoid arthritis, systemic lupus erythematosis, and osteoarthritis, as well as nonsystemic acute and chronic muscle, joint and ligament discomforts. With more than 100 million prescriptions annually, NSAIDs are the most commonly prescribed drug class (1). Concerns regarding safety have led to reduced use in older and high-risk patients, who are often the patients most in need of pain relief. Billions of dollars are spent each year on NSAIDs, the majority over the counter purchases with approximately 10% by prescription. The introduction of COX-2 inhibitors in the late 1990’s was intended to reduce the incidence of NSAID induced bleeds while preserving equivalent efficacy (6,7). Although COX-2 inhibitors did show a reduction in gastrointestinal side effects, the incremental cost and concerns about cardiovascular safety have limited their use (8,9). Two popular agents (Vioxx and Bextra) were voluntarily withdrawn from the market. Further investigation revealed that the COX-2 inhibitors were probably no different from non selective NSAIDs in terms of cardiovascular risk (10, 11, 12). Physician and public awareness of the potential toxicity of the class was increased. These drugs simply provide symptomatic relief making the search for alternative agents imperative. NSAIDs are associated with significant drug induced morbidity and mortality. In the late 1990’s it was estimated 16,500 died and over 100,000 were hospitalized from NSAID induced GI bleeds (2). This complication accounted for a third of the total cost of arthritis care (3, 4). Patients over the age of 65, with concomitant medications and disease states are the most likely to suffer serious consequence side effects (5). NSAIDs are associated with other toxicities as well. Patients with impaired renal or hepatic function must exercise caution when taking NSAIDs or should not take them at all. NSAIDs precipitate blood pressure elevation and fluid retention. Approximately a fifth of the population cannot tolerate NSAIDs due to esophageal reflux, dyspepsia or diarrhea and GI side effects independent of hemorrhage (5,13). Table 1. Risk factors for NSAID induced upper gastrointestinal bleed The addition of proton pump inhibitors to NSAIDs has been shown to decrease GI bleeds and dyspepsia by up to 50% (14). However, the additional cost and other potential complications such as pneumonic reduced calcium absorption, resulting in osteoporosis, (15) and B12 deficiency from changes in gut flora are problematic (16, 17). Reduced compliance occurs due to the number of pills taken and frequency in dosing (18-20). New agents combining a NSAID with a Nitric Oxide moiety reduce GI bleeds by 20-30% and lower the risk of hypertension, but have yet to be approved by the FDA and will be costly if they reach the market (21). Age of 65 years and over. Previous history of gastroduodenal ulcer and gastrointestinal bleeding. Concomitant use of medications that are known to increase the likelihood of uppergastrointestinal adverse events (anticoagulants, aspirin, including low-dose aspirin, and corticosteroids). Presence of serious co-morbidity, such as cardiovascular disease, renal or hepatic impairment, diabetes, or hypertension. Prolonged duration of NSAID use. Use of the maximum recommended doses of NSAIDs. The presence of Helicobacter pylori infection. Alcohol use. Smoking. *Corresponding author. David S. Silver MD, Targeted Medical Pharma, 2980 N. Beverly Glen Circle, Suite 301,Los Angeles, CA 90077, USA Alternatives to NSAIDs for pain management also have challenges. Narcotic analgesics, although effective, are sedating, cause constipation, urinary retention and have potential for addiction. Tricyclic antidepressants, dual reuptake inhibitors, anti epileptics, and others have a separate set of side effects and can also be costly. Theramine Theramine is intended for use in the management of pain syndromes including Journal of Pharmacy Research Vol.5 Issue 5.May 2012 2806-2809 David S. Silver et al. / Journal of Pharmacy Research 2012,5(5),2806-2809 fibromyalgia, acute and chronic, neuropathic or inflammatory pain. Theramine is a medical food that must be used under the active or ongoing supervision of a physician. Medical foods address the altered physiologic requirements and distinctive nutritional needs resulting from metabolic disorders, chronic diseases, injuries, premature birth, other medical conditions or drug therapies. (22) The nutrient requirements that are most crucial for patients with pain syndromes are the amino acids which are essential for the synthesis of neurotransmitters which transmit pain signals and mediate their perception. (23) The concept that nutrient requirements are modified in disease has long been recognized, and is supported by studies of plasma, urinary, and tissue levels of nutrients associated with changes in physiological endpoints, symptoms or decreases. (24). These requirements can be estimated by determining the level of intake at which a physiological response is normalized, indicating that the balance between intake and metabolic demand has been restored. For example, improvement in perceived intensity of back pain following consumption of supplemental amounts of 5-hydroxytryptophan, arginine, and glutamine from Theramine suggests an additional need for tryptophan, arginine, and glutamate in individuals suffering with pain syndromes. (25-28) Many peer-reviewed publications support increased requirements of arginine, tryptophan, choline, glutamine, serine, and histidine in pain syndromes. Patients suffering with pain syndromes have decreased blood levels of these amino acids despite sufficient protein intake indicating amino acid needs are selectively increased in pain patients. (29 -31) This observation may be explained by the competitive demands for these amino acids by metabolic pathways which decrease the supply available to moderate the pain process. Low blood levels of tryptophan and/or altered tryptophan metabolism are reported in patients with pain disorders. (32) These patients also exhibit reduced blood levels of 5-hydroxytryptophan (5-HTP), arginine, choline, GABA, histidine, and serine. (33 - 35) Moreover, they respond to oral administration of amino acid formulations by showing favorable changes in physiologic endpoints and improvements in clinical symptoms supporting a need for increased amounts of those amino acids which are reduced in the blood of patients with pain disorders. (36,37). Theramine Clinical Trials Two double blind multicenter randomized trials which compared Theramine to low dose Naproxen and Ibuprofen respectively have been performed (38,39). Each study involved 120 patients in 12 centers around the US. Patients were randomized to receive Theramine alone, 2 capsules twice daily, NSAID, or a combination of both. In both studies, Theramine showed statistically significantly greater pain relief than either Naproxen, 250mg daily or Ibuprofen 400mg daily. The combination of Theramine and NSAID produced better pain relief than either alone. Importantly, Theramine lowered CRP while increased CRP was seen in the Naproxen and Ibuprofen groups. No side effects from Theramine were reported in either trial. The available clinical data estimates 1-4% of patients over the age of 65 taking NSAIDs will experience a GI bleed annually (40-42). A significant percentage of patients in this age group will take aspirin 81mg every day or every other day for cardiovascular prophylaxis, further increasing the risk (43). The estimated cost of hospitalization for a GI bleed is $50,000 (44). It is recommended that high risk patients take proton pump inhibitors (PPIs) with the NSAID to prevent GI events (45-47). This includes patients over the age of 65 or patients with a previous history of ulcer or GI hemorrhage which represent over half of all chronic users of NSAIDS. The costs of additional PPIs are approximately $120 per month. Compliance with combination therapy, especially in populations with already complicated medical regimens, is diminished. Most commonly, if patients are asymptomatic, they will continue to take the NSAID for pain relief and not take the PPI, although a preparation that combines naproxyn with prilosec is available in a single pill. Alternatively, the use of Celecoxib, the only available COX-2 selective inhibitor, would cost approximately $200. These costs do not account for the other complications including renal, hepatic, and others associated with the use of NSAIDs. Biannual blood screening to monitor potential side effects of NSAIDs is estimated at $100 and must be considered as well. Finally, evaluation costs for patients who present with dyspepsia and undergo gastroenterological workup. Estimates range from 25-50% of all patients taking NSAIDs will have evidence of endoscopic ulceration in the stomach or duodenum, but only a fraction of them will be scoped in clinical settings (48,49). The following assumptions are based on clinical evidence available with an implied bias in favor of NSAIDs, as they are presently the preferred treatment. 1. 2. 3. 4. 5. 6. Observational reporting after over 60,000,000 doses of Theramine revealed no GI bleeds. Although underreporting may have occurred, the data implies GI risk in patients taking Theramine is unlikely. Therefore, an increased risk of gastrointestinal bleeds based on the formulation would not be expected. 7. 8. Cost Analysis Pharmacy and therapeutic formulary committees are charged with balancing efficacy and safety concerns versus cost of medication acquisition. Analysis of economic factors is necessary before placing a more expensive medication on formulary. In an ideal world, only safety and efficacy would drive decisions. The burden of escalating health care costs and limited resources demand a pharmacoeconomic analysis. The costs of side effects can be challenging to quantify but must be undertaken to determine cost benefit rates of Theramine compared to a generic NSAID. 9. The annual risk of GI bleed in patients over the age of 65 taking regular NSAIDs is 2.5%. Theramine does not cause gastrointestinal bleeds. The cost of generic NSAID is $10 per month. The insurance reimbursement of Theramine averages $176 per month at the most commonly and clinical trial dose. The dollar amount is based on the average insurance reimbursement for Theramine. Laboratory screen of complete blood count (CBC) and complete metabolic panel semi-annually costs $100 total per year. Estimate of 40-65% of NSAID users are considered at high GI risk for bleed and should receive a PPI or COX-2 inhibitor. All patients in the 65 and older age group are considered at high risk. It is recommended that high risk patients should be either on a COX-2 inhibitor or a PPI with a standard NSAID. The average cost estimate would be $40 to $120 per month per patient and will prevent 50% of all NSAID induced GI bleeds. Assume 10% of patients taking NSAIDs will require GI work up for dyspepsia costing $1200 for upper endoscopy, Helicobacter Pylori testing, consultation and endoscopies. Mortality for upper gastrointestinal bleed is approximately 16% in high risk patients (2,49). Calculations of cost per 100 patients treated per year will be performed as well as cost per life saved. The additional costs of cardiovascular, renal and hepatic toxicity due to NSAIDs may be substantial but have not been calculated. The figures do not include patients in whom NSAID therapy is contraindicated, specifically patients with renal insufficiency, previous history of GI bleed, congestive heart failure, peripheral edema, hepatic failure, poorly controlled hypertension and aspirin sensitivity. These patients now have an option for non-narcotic pain relief. Journal of Pharmacy Research Vol.5 Issue 5.May 2012 2806-2809 David S. Silver et al. / Journal of Pharmacy Research 2012,5(5),2806-2809 MATERIALS AND METHODS Risk data and rates of gastrointestinal bleeding, morbidity and mortality data were derived from the Medicare database and peer reviewed literature. Medication acquisition costs were determined based on listed average wholesale pricing (AWP). Procedural and hospitalization rates were obtained from the Medicare fee schedule. RESULTS The annual cost of Theramine for 100 patients treated is $211,200. A generic NSAID for 100 patients would be $12,000 if all patients were given generic NSAID’s; the cost of branded prescription NSAIDs would obviously add to this cost. The additional costs include: $75,000 for costs of GI bleeds per 100 patients treated per year, medication acquisition costs of $144,000 for high risk patients, laboratory testing of $10,000 for all patients, cost of GI evaluation of symptomatic patients $14,500. Excluding cost of treatment for hepatic, renal and cardiovascular side effects generic NSAIDs will cost $241,500 per 100 patients treated. When examining actual costs, Theramine is cost savings when comparing total impact of NSAIDs. Cost per life saved analysis was not performed as there is no incremental overall cost increase to use of Theramine. DISCUSSION NSAIDs are the most commonly utilized drug class. Despite efficacy in treating symptoms of inflammatory and non inflammatory conditions, their use has been reduced greatly due to the side effect profile. Physicians must make difficult decisions either accepting the risk or prescribing as needed with therapeutic efficacy reduced. Patients are either exposed to significant side effects or suffer with inadequate pain relief. Recent guidelines for pain management in the elderly have recommended only rare use of NSAIDS, despite the fact that over 50% of individuals over the age of 65 suffer from chronic pain (50). Efforts to diminish the GI side effects of NSAIDs include co-administration of a PPI or use of COX-2 inhibitor increase cost. They do not eliminate the gastrointestinal effects or impact hepatic, renal and cardiovascular toxicities at all. Theramine, a prescription only medical food, has been shown to be an effective and safe anti-inflammatory pain reliever without the concerning side effects of NSAIDs. Although more expensive than generic NSAIDs, the use of Theramine is cost neutral using conservative estimates of the economic impact of NSAID side effects based on best care practice guidelines. Cost neutrality favors the use of Theramine because of the reduction in morbidity and mortality. Theramine, unlike generic NSAIDs plus a PPI or COX-2 inhibitors, eliminates all NSAID induced side effects. Despite the initial upfront costs, even when low risk patients are included, Theramine lowers cost of care, and there is no risk of GI bleed, renal or hepatic toxicity, hypertension, peripheral edema or heart attack. Based on this analysis, Theramine should be preferred in all cases instead of NSAIDs. If Theramine does not provide adequate therapeutic response, use of NSAIDs can be considered and administered at the lowest dose and for the shortest time when added to Theramine. The economic reducibility or likely cost saving in addition to the reduced burden of morbidity and mortality make Theramine preferred over NSAID for treatment of pain syndromes. ACKNOWLEDGEMENTS During the preparation of this manuscript, Elizabeth Charuvastra passed away after a brief but brave battle with pancreatic cancer. Her contributions to this paper, and the development of safer and more effective medical treatments and her tireless devotion to patients are incalculable. She will be sorely missed by all that have known and worked with her. Conflict of Interest The authors of this manuscript are owners, employees or consultants of Targeted Medical Pharma, Inc. (TMP). TMP has been the sponsor for all data compilation and preparation related to this manuscript. REFERENCES: 1. http://www.medscape.com/viewarticle/538039 2. Wolff M, Lichtenstein D, Singh G. Gastrointestinal toxicity of non-steroidal anti-inflammatory drugs. N Engl J Med. 1999; 340(24):1888-99. 3. Johnson R, Hornbrook M, Hooker R, et al. Analysis of the costs of NSAID-associated gastropathy. Experience in a US health maintenance organization. Pharmacoeconomics. 1997; 12(1):7688. 4. Sheen C, MacDonald T. Gastrointestinal side effects of NSAIDspharmacoeconomic implications. Expert Opin Pharmacotherapy. 2002; 3(3):265-9. 5. Burmester G, Lanas A, Biasucci L, et al. The appropriate use of non-steroidal anti-inflammatory drugs in rheumatic disease: opinions of a multidisciplinary European expert panel. Ann Rheum Dis. 2011; 70(5):818-22. 6. Bombardier C, Laine L, Reicin A, et al. Comparison of upper gastrointestinal toxicity of rofecoxib and naproxen in patients with rheumatoid arthritis. VIGOR Study Group. N Engl J Med. 2000; 343(21):1520-8. 7. Silverstein F, Faich G, Goldstein J, et al. Gastrointestinal toxicity with celecoxib vs nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis: the CLASS study: A randomized controlled trial. JAMA 2000; 284(10):1247-55. 8. FitzGerald G, Cheng Y, Austin S. COX-2 inhibitors and the cardiovascular system. Clin Exp Rheum 2001; 19(6 suppl 25):S316. 9. Mukherjee D, Nissen S, Topol E. Risk of cardiovascular events associated with selective COX-2 inhibitors. JAMA 2001; 286(8):954-9. 10. http://www.fda.gov/ForConsumers/ConsumerUpdates/ ucm107856.htm 11. White W, Faich G, Borer J, Makuch R. Cardiovascular thrombotic events in arthritis trials of the COX-2 inhibitor celecoxib. Am J Cardiol. 2003; 92(4):411-8. 12. Kearney P, Baigent C, Godwin J, et al. Do selective COX-2 inhibitors and traditional NSAIDs increase the risk of atherothrombosis? Meta-analysis of randomized trials. BMJ. 2006; 332(7553):1302-8. 13. Thiefin G, Schaeverbeke T, Barthelemy P, et al. Upper gastrointestinal symptoms in patients treated with nonsteroidal anti-inflammatory drugs: prevalence and impact-the COMPLAINS study. Eur J Gastro Hepatol. 2010; 22(1):81-7. 14. Moore R, Derry S, Phillips C, McQuay H. NSAIDs, COX-2 selective inhibitors (coxibs) and gastrointestinal harm: review of clinical trials and clinical practice. BMC Musculoskeletal Disorders. 2006; 7:2474-9 15. Gray S, LaCriox A, Larson J, et al. Proton pump inhibitor use, hip fracture, and change in bone mineral density in postmenopausal women: results from the Women’s Health Initiative. Arch Int Med. 2010; 170(9):765-71. 16. Ito T, Jensen R. Association of long-term proton pump inhibitor therapy with bone fractures and effects on absorption of calcium, vitamin B12, iron, and magnesium. Curr Gastro Rep. 2010; 12(6):448-57. 17. Thiefin G, Schwalm M. Underutilization of gastroprotective drugs in patients receiving non-steroidal anti-inflammatory drugs. Dig Liver Dis. 2011; 43(3):209-14. Journal of Pharmacy Research Vol.5 Issue 5.May 2012 2806-2809 David S. Silver et al. / Journal of Pharmacy Research 2012,5(5),2806-2809 18. Helsper C, Smeets H, Numans M, et al. Trends and determinants of adequate gastroprection in patients chronically using NSAIDs. Pharmacoepidemiol Drug Saf. 2009; 18(9):800-6. 19. Laine L, Connors L, Griffin M, et al. Prescription rates of protective co-therapy for NSAID users at high GI risk and results of attempts to improve adherence to guidelines. Aliment Pharmcol Ther. 2009; 30(7):767-74. 20. Schnitzer T, Kivitz A, Frayssinet H, Duquesroix B. Efficacy and safety of naproxcinod in the treatment of patients with osteoarthritis of the knee: a 13-week prospective, randomized, multicenter study. Osteoarthritis Cartilage. 2010; 18(5):629-36. 21. Borgonio A, Witte K, Stahrenberg R, Lemmer B. Influence of circadian time, ageing, and hypertension on the urinary excretion of nitric oxide metabolites in rats. Mech Ageing Dev 1999;111:23-37. 22. Wurtman RJ, Hefti F, Melamed E. Precursor control of neurotransmitter synthesis. Pharmacol Rev 1980;32:315-335. 23. Modern Nutrition in Health and Disease. 10th ed. Philadelphia: Lipincott Williams & Wilkin, 2006. 24. Brown DW. Abnormal fluctuations of acetylcholine and serotonin. Med Hypotheses 1993;40:309-310. 25. Carlsson A. Interaction between dopaminergic and serotonergic systems. Clin Neuropharmacol 1992;15 Suppl 1 Pt A:616A-617A. 26. Lehnert H, Wurtman RJ. Amino acid control of neurotransmitter synthesis and release: physiological and clinical implications. Psychother Psychosom 1993;60:18-32. 27. Young SN, Teff KL. Tryptophan availability, 5HT synthesis and 5HT function. Prog Neuropsychopharmacol Biol Psychiatry 1989;13:373-379. 28. Fernstrom JD. Effects of the diet on brain function. Acta Astronaut 1981;8:1035-1042. 29. Fernstrom JD. Can nutrient supplements modify brain function? Am J Clin Nutr 2000;71:1669S-1675S. 30. Fernstrom JD. Dietary amino acids and brain function. J Am Diet Assoc 1994;94:71-77. 31. Yunus MB, Dailey JW, Aldag JC, Masi AT, Jobe PC. Plasma tryptophan and other amino acids in primary fibromyalgia: a controlled study. J Rheumatol 1992;19:90-94. 32. 149 Field CJ, Johnson I, Pratt VC. Glutamine and arginine: immunonutrients for improved health. Med Sci Sports Exerc 2000;32:S377-S388. 33. de Koning TJ, Klomp LW. Serine-deficiency syndromes. Curr Opin Neurol 2004;17:197-204. 34. Histidine: An essential amino acid for normal adults. Nutr Rev 1975;33:200-202. 35. Fernstrom JD. Effects of precursors on brain neurotransmitter synthesis and brain functions. Diabetologia 1981;20 Suppl:281289. 36. Regulation of serine dehydratase and phosphoglycerate dehydro- 37. 38. 39. 40. 41. 42. 43. 44. 45. 46. 47. 48. 49. 50. genase by proteins and essential amino acids. Nutr Rev 1974;32:8889. Shell W, Charuvastra E, DeWood M, et al. A double-blind controlled trail of a single dose naproxen and an amino acid medical food Theramine for the treatment of low back pain. Am J Ther. 2010; 21. Information of file. Targeted Medical Pharma. SCORE tool for assessment of upper gastrointestinal bleeds as found at: https://www.hpsm.org/documents/SCORE_for_ NSAID_Risk_Tool.pdf Stocki K, Cyprien L, Chang EY. Gastrointestinal bleeding rates among managed care patients newly started on cox-2 inhibitors or nonselective NSAID’s. J Manag Care Pharm. 2005; 11(7):550-8. Hernanadez-Diaz S, Rodriguez L. Association between nonsteroidal anti-inflammatory drugs and upper gastrointestinal tract bleeding/perforation: an overview of epidemiological studies published in the 1990’s. Arch Int Med. 2000; 160(14):2093-9. Serensen H, Mellemkjaer L, Blot W, et al. Risk of upper gastrointestinal bleeding with use of low-dose aspirin. Am J Gastro. 2000; 95(9):2218-24. Silver D. Use of COX-1-Sparing Agents in the Federal Health System. Pharmacy and Therapeutics. 2004; 29(7). Pilotto A, Franceschi M, Maggi S, et al. Optimal management of peptic ulcer disease in the elderly. Drugs Aging. 2010; 27(7):54558. Spiegel B, Chiou C, Ofman J. Minimizing complications from nonsteroidal anti-inflammatory drugs: cost-effectiveness of competing strategies in varying risk groups. Arth Rheum. 2005; 53(2):185-97. Bardou M, Barkun A. Preventing the gastrointestinal adverse effects of nonsteroidal anti-inflammatory drugs: from risk factor identification to risk factor intervention. Joint Bone Spine. 2010; 77(1):6-12. Hawkey C, Laine L, Simon T, et al. Comparison of the effect of rofecoxib (a cyclooxygenase 2 inhibitor), ibuprofen, and placebo on the gastroduodenal mucosa of patients with osteoarthritis: a randomized, double-blind, placebo-controlled trial. The Rofecoxib Osteoarthritis Endoscopy Multinational Study Group. Arth Rheum. 2000; 43(2):370-7. Hawkey C, Laine L, Harper S, et al. Influence of risk factors on endoscopic and clinical ulcers in patients taking rofecoxib or ibuprofen in two randomized controlled trials. Aliment Pharmacol Ther. 2001; 15(10):1593-601. Jureidini Z, Abdul Walid O. Upper gastrointestinal bleeding: an age based comparative study. IJGE 2001; 1(1):22-8. Barkin R, Beckerman M, Blum S, et al. Should nonsteroidal antiinflammatory drugs (NSAIDs) be prescribed to older adults. Drugs Aging. 2010; 27(10):775-89. Source of support: Nil, Conflict of interest: None Declared Journal of Pharmacy Research Vol.5 Issue 5.May 2012 2806-2809 -60 -40 -20 0 20 40 60 80 -70 -60 -50 -40 -30 -20 -10 0 10 -50.3 -63.1 p<0.01 for the comparison of Theramine or combination versus ibuprofen Combination n = 38 Theramine n = 41 Ibuprofen n = 43 60.1 -35.99 -50 -40 -30 -20 -10 0 10 20 -70 -60 -50 -40 -30 -20 -10 0 12.65 -4.5 -62.2 -23.55 -43.1 Figure 4. Interleukin- 6 -41.9 Figure 2. Oswestry Pain Index Percent Change in Amino AcidAcid Blood Concentration DayDay 1 and 28 Figure 5. Percent Change in Amino Serum Concentrationatfrom 1 to Day Day 28 Versus Normal -47.05 p<0.01 for the comparison of Theramine or combination versus ibuprofen. Combination n = 38 Theramine n = 41 Ibuprofen n = 43 Figure 3. High Sensitivity C-Reactive Protein 0.73 Figure 1. Roland-Morris Pain Scale Results Affiliation: Targeted Medical Pharma, Inc. p<0.01 for the comparison of Theramine and combination versus ibuprofen Combination n = 38 Theramine n = 41 Ibuprofen n = 43 p<0.01 for the comparison of Theramine or combination versus ibuprofen. Ibuprofen n = 43 Theramine n = 41 Combination n = 38 Ibuprofen 77 Theramine 19 Combination 12 Figure 6. Percent Subject Use of Rescue Medication (APAP 6501000mg prn) at Study Completion p<0.01 n=122 Combination Theramine Ibuprofen * All authors are employees of Targeted Medical Pharma, Inc. which manufactures and distributes the medical food, Theramine under subsidiary brand Physician Therapeutics. Theramine is an effective analgesic in the treatment of chronic back pain. Theramine reduces inflammatory markers. Theramine may provide a safer pharmacologic alternative to traditional pharmaceuticals prescribed in the management of chronic back pain. Conclusions Both the Theramine alone group and the combination group showed statistically significant reductions in pain when compared to baseline. (p<0.01) The ibuprofen group alone showed no difference. (Figures 1 and 2) Laboratory blood tests confirmed Theramine reduced inflammatory markers as measured by hsCRP and Il-6. (Figures 3 and 4) Administration of Theramine resulted in an increase in blood concentrations of the amino acids associated with the neurotransmitters involved in the modulation of pain. (Figure 5) Theramine subjects required statistically significantly less rescue medications when compared to ibuprofen. (p<0.01) (Figure 6) 0 10 20 30 40 50 60 70 80 90 Publication pending . Previous study entitled, “A Double Blind Placebo Study to determine the effectiveness of Theramine on the management of chronic back pain”, American Journal of Therapeutics (2011) A total of 127 patients were randomized (122 completed) to one of three treatment arms: ibuprofen 400mg (n=42); Theramine (n=42); or the combination of Theramine and ibuprofen (n=43) for a 28-day period. Pain was assessed using the Roland-Morris Pain Scale, Oswestry Disability Index and Visual Analog Scale. Blood samples were taken at baseline and Day 28 to measure Complete Blood Count (CBC) and liver function. The inflammatory markers high sensitivity CReactive Protein (hsCRP) and Interleukin-6 (Il-6) were measured at baseline and Day 28. 25 subjects were randomly selected for serum amino acid level sampling, measured at baseline and Day 28. Materials & Methods condition. Many current drug treatments such as non-steroidal anti-inflammatory drugs (NSAIDs) and narcotic analgesics are associated with significant side effects, including gastrointestinal (GI) hemorrhage, renal and cardiovascular complications and have an increased potential for addiction. The primary objective of this study was to compare prescription-only Theramine, an amino-acid based prescription medical food/old drug, to low dose ibuprofen in the treatment of chronic low back pain. Theramine has been shown in a previous double blind clinical trial to be effective in the treatment of chronic low back pain when compared with naproxen, as well as reducing levels of inflammatory mediators. Chronic low back pain is a common debilitating Background Authors: William E. Shell M.D., David S. Silver M.D., Fred McCall-Perez Ph.D., Stephanie Pavlik C.R.A.* THERAMINE VERSUS IBUPROFEN FOR THE SAFE AND EFFECTIVE MANAGEMENT OF CHRONIC LOW BACK PAIN AND INFLAMMATION Percent Change Percent Change Percent Change Percent Change Percent Change Theramine® Product Information Theramine®ProductInformation Indication Theramine® is intended for use in the management of pain syndromes that include acute pain, chronic pain, fibromyalgia, neuropathic pain, and inflammatory pain. Theramine is a medical food that must be used under the active or ongoing supervision of a physician. Medical foods are developed to address the different or altered physiologic requirements that may exist for individuals with distinctive nutritional needs arising from metabolic disorders, chronic diseases, injuries, premature birth associated with inflammation and other medical conditions, as well as from pharmaceutical therapies.1 Pain is a complex process initiated by pain-inducing or noxious stimuli interacting with pain receptors (nociceptors) which triggers a series of action potentials that are transmitted by neurotransmitters from peripheral afferent neurons to the spinal cord and higher nerve centers in the brain. The presence of inflammation exacerbates the intensity of signals from these stimuli by sensitization of the peripheral neurons in the surrounding area. Patients with pain syndromes benefit from increased availability of choline, arginine, glutamine, histidine, 5-hydroxytryptophan, and serine to restore homeostasis. Theramine is designed to provide a balance of neurotransmitters with well-defined roles in the modulation of pain and a blend of antioxidants, anti-inflammatory agents, and immunomodulators to moderate the effects of inflammation on the pain response. Ingredients Theramine is a proprietary blend of neurotransmitters (gamma-aminobutyric acid [GABA], Lglutamate, L-glutamine) and neurotransmitter precursors (choline bitartrate, L-arginine, L-glutamine, L-histidine, 5-hydroxytryptophan, and L-serine); stimulators of precursor uptake (cinnamon); polyphenolic antioxidants (grape seed extract, cinnamon bark, cocoa powder); anti-inflammatory and immunomodulatory peptides (whey protein hydrolysate); adenosine antagonists (cocoa powder, metabromine); and an inhibitor of the attenuation of neurotransmitter production associated with precursor administration (grape-seed extract). The neurotransmitters and neurotransmitter precursors have been carefully selected based on scientific support for their roles in the physiological processes involved in reduction of pain. These roles are summarized in this monograph in the section, Scientific Support for Use of Theramine in Management of Pain Syndromes. The other ingredients in the formulation are involved in neurotransmitter metabolism or are functional components of the Targeted Cellular Technology® system. All of the ingredients included in Theramine are classified as generally recognized as safe (GRAS) by the United States Food and Drug Administration (FDA). To qualify for GRAS status, a substance that is added to a food, including a medical food, has to be supported by data demonstrating that it is safe when consumed in amounts from these foods, as they are typically ingested or prescribed. 1 As defined in the guidelines issued by the Center for Food Safety and Nutrition, United States Food and Drug Administration (FDA). 1|P a g e Revised03Mar2011 Theramine® Product Information TargetedCellularTechnology® Theramine has been formulated using Targeted Cellular Technology, an integrated molecular system that facilitates the uptake and utilization of neurotransmitter precursors by target cells within the nervous system. This 5-component patented system consists of (1) specific neurotransmitter precursors; (2) a stimulus for the neuronal uptake of these precursors by specific neurons; (3) an adenosine antagonist that blocks the inhibitory effect of adenosine on neuronal activity (adenosine brake); (4) a stimulus to trigger the release of the required neurotransmitters from targeted neurons; and (5) a mechanism to prevent attenuation of the precursor response, a well known phenomenon associated with precursor administration. Use of Targeted Cellular Technology improves the metabolic efficiency of neurotransmitter synthesis, thereby reducing the amounts of precursors needed to correct neurotransmitter imbalances. Use of Targeted Cellular Technology also ensures that the appropriate amounts of neurotransmitter precursors are delivered to the target neurons with the appropriate timing. As such, Targeted Cellular Technology synchronizes the availability of the precursor supply with the fluctuating demand for the corresponding neurotransmitters, which is especially important for processes which are regulated by circadian rhythms and are therefore sensitive to the timing of the synthesis and release of neurotransmitters such as acetylcholine, serotonin, nitric oxide, and histamine (1-5). Previous attempts to provide an exogenous source of precursor amino acids and other biogenic amines in the quantities required to support neurotransmitter synthesis for individuals with specific needs necessitated that large amounts of amino acids be added to the formulations. For patients whose precursor requirements were considerably higher than normal, the amounts of exogenous amino acids that were needed were not practical to consume on a daily basis. Moreover, ingestion of large quantities of amino acids increases the potential for adverse effects. Metabolic efficiency is also decreased when large amounts of amino acids are delivered to the cells at one time because intestinal membrane transport receptors would be rapidly saturated resulting in a reduction in fractional amino acid absorption and thus attenuation of the tissue response to the supplemental amounts provided. Improving metabolic efficiency in uptake and utilization of neurotransmitter precursors by target neurons with Targeted Cellular Technology allows ingestion of smaller amounts of amino acids to elicit the same responseas larger amounts, making daily dosing more feasible and reducing the potential for tolerance. Unlike pharmaceutical agents which are not innate components of the pain process, and thus may lose their effectiveness in a relatively short period of time, the effectiveness of Theramine is not attenuated. Metabolism Theramine is a source of amino acids,biogenic amines, and other nutrients formulated for patients with certain types of pain syndromes. These patients require additional amounts of arginine, choline, GABA, glutamine, histidine, 5-hydroxytryptophan, and serine to restore homeostasis. Under normal physiological conditions, glutamine, arginine, serine, and choline are considered nonessential because endogenous synthesis is sufficient to satisfy metabolic demand. When needs are altered by conditions that increase metabolic demand, the usual rate of synthesis is no longer sufficient and these nutrients become conditionally essential, requiring that supplemental amounts be consumed. Histidine has also been 2|P a g e Revised03Mar2011 Theramine® Product Information considered a nonessential amino acid for adults because blood levels can be maintained by breakdown of skeletal muscle and hemoglobin; however, there is no evidence of de novo histidine synthesis in mammalian tissues and thus an exogenous supply is important, especially during times of increased need to preserve muscle mass and plasma hemoglobin concentration. In contrast to the nutrients which are nonessential under normal conditions, tryptophan is an essential amino acid that must always be consumed from exogenous sources, as the enzymes required for its synthesis are absent in humans. Because it is an essential amino acid, the amount of tryptophan consumed determines the amount available for utilization by multiple pathways. Tryptophan is a precursor of the neurotransmitter serotonin, as well as of the coenzymes nicotinamide adenine dinucleotide (NAD+) and nicotinamide adenine dinucleotide phosphate (NADP) (Figure 1). The competition between these and other metabolic pathways for the supply of tryptophan available restricts the amount of serotonin that can be produced from supplemental amounts of the amino acid. Figure 1. Competing Pathways of Tryptophan Metabolism To overcome this limitation, Theramine provides 5 hydroxytryptophan, an intermediate metabolite in the pathway of tryptophan conversion to serotonin, thus bypassing the rate-limiting step dependent on tryptophan availability (6) (Figure 2). Unlike tryptophan, this intermediate cannot be shunted into production of niacin or protein which eliminates competition by other metabolic pathways for the amount available. Consequently, an increase in 5-hydroxytryptophan lessens the dependence of serotonin levels on the amount of tryptophan consumed. By facilitating production of serotonin without requiring consumption of large amounts of tryptophan, Theramine ensures that adequate amounts of serotonin are produced without compromising synthesis of other important compounds derived from tryptophan, thus improving metabolic efficiency. 3|P a g e Revised03Mar2011 Theramine® Product Information Figure 2. Structure of 5-Hydroxytryptophan As nonessential amino acids, glutamine, arginine, and serine are not normally dependent on exogenous sources, thus metabolic competition for these amino acids develops only under conditions of increased demand. Under normal conditions, glutamine is synthesized from glutamate in virtually all tissues by the transfer of an amino group. Glutamine can therefore function as a carrier of amino groups which are utilized in the synthesis of a number of cellular compounds including the antioxidant glutathione (Ȗ-glutamylcysteinylglycine) as well as purines, pyrimidines, and urea (Figure 3). Deamination of glutamine regenerates glutamate, the major excitatory neurotransmitter of the central nervous system which is also utilized as a precursor for the synthesis GABA, the major inhibitory neurotransmitter of the central nervous system. In addition, glutamine also functions as a neurotransmitter at NMDA receptor sites in the brain. Figure 3. Competing Pathways of Glutamine Metabolism 4|P a g e Revised03Mar2011 Theramine® Product Information For individuals with inflammatory pain, the requirement for glutamine is greater than normal because additional amounts are needed to support the increase in demands for glutamate and GABA. The competition for glutamine by other pathways that utilize it will limit the amount of glutamate that can be produced, and subsequently, the amount of GABA. A decrease in the supply of glutamine will draw on the available supply of glutamate and thus can compromise other important glutamate-dependent functions. Theramine improves metabolic efficiency by providing supplemental glutamine to ensure that there will be sufficient glutamate available to support its metabolic activity without drawing from the supply of glutamine needed by competing pathways. Additional GABA further ensures that there is a sufficient amount of this neurotransmitter while conserving the available supplies of both glutamate and glutamine. The metabolic pathways which generate arginine are also normally sufficient to ensure an adequate supply of this amino acid. Because arginine can be synthesized from glutamine and glutamate, it is not considered an essential nutrient. A critical role for arginine is utilization as precursor of nitric oxide, a neurotransmitter which also functions as a vasodilator, immunomodulator, and intercellular/intracellular messenger. Arginine is also utilized as a precursor of polyamines, urea, and the high-energy storage compounds creatine and creatine phosphate (Figure 4). When the demand for nitric oxide is increased, arginine is diverted away from the synthesis of these other compounds. To compensate a decrease in arginine available for utilization by competing pathways, glutamate and glutamine are metabolized to ornithine and citrulline which are then converted to arginine in the urea cycle. Theramine improves metabolic efficiency by ensuring that there is a sufficient amount of arginine available to satisfy the demands of competing pathways and prevent depletion of the glutamate body pool which would upset neurotransmitter balance (nitric oxide, glutamate, and GABA). Supplemental arginine will also ensure that there is sufficient glutamine available to conserve the existing supply of glutamate. 5|P a g e Revised03Mar2011 Theramine® Product Information Figure 4. Competing Pathways of Arginine Metabolism Choline is also considered a nonessential nutrient under normal physiological conditions. When the demand for choline is increased to supply precursor for synthesis of acetylcholine, supplemental amounts of choline are needed. Acetylcholine is produced from choline in an acetylation reaction catalyzed by choline acetyltransferase with acetyl coenzyme A (CoA) as the acetyl group donor (Figure 5). Figure 5. Biosynthesis of Acetylcholine The primary source of choline normally utilized in the synthesis of acetylcholine is phosphatidylcholine (lecithin), a membrane phospholipid which serves as a reservoir to supply choline for short-term needs (Figure 6). When the demand for acetylcholine exceeds the amount of choline that can be supplied by the hydrolysis of phosphatidylcholine from the membrane pool, dietary choline becomes an increasingly more important source. Theramine provides additional amounts of choline to meet the increased needs for acetylcholine when demand is elevated for an extended time period. By supplying an exogenous 6|P a g e Revised03Mar2011 Theramine® Product Information source of choline, Theramine prevents the depletion of membrane phosphatidylcholine and thus preserves the structural integrity of the cell. Figure 6. Sources of Acetylcholine Serine is a nonessential amino acid which functions as a neurotransmitter/neuromodulator in the brain and spinal cord in its D isomeric form (5). Since amino acids can only be incorporated into human protein as L isomers, D-serine is utilized solely as a neurotransmitter. Adequate amounts of L-serine are normally synthesized from 3-phosphoglycerate, an intermediate of glucose metabolism. To produce the neurotransmitter, L-serine is converted to D-serine by serine racemase which is localized primarily in specific neurons in the central nervous system (Figure 7).These neurons normally obtain L-serine from the plasma because they lack the enzyme needed for the synthesis of this amino acid. When demand for D-serine is high, plasma L-serine levels may not be adequate to provide sufficient quantities of precursor to these neurons, thus additional amounts must be obtained from nearby astrocytes. As a result, astrocyte production of L-serine becomes rate-limiting to the supply of D-serine during periods of increased needs. Theramine provides additional L-serine to ensure that there is sufficient D-serine available to meet these needs. 7|P a g e Revised03Mar2011 Theramine® Product Information Figure 7. Conversion of L-Serine to D-Serine Dosage The recommended dose of Theramine is 1 or 2 capsules, taken 1 to 4 times daily as directed by a physician. Theramine is designed to reduce pain and support the function of pain medications by minimizing side effects. Patients who are taking pharmaceutical agents to relieve pain may continue to take these medications with Theramine. As with any medical food, the best dosing protocol should be determined by assessment of individual needs. There are no known interactions between Theramine and any medication. Theramine can be taken with pain medications such as once daily low dose aspirin (32 mg) or other nonsteroidal anti-inflammatory drugs (NSAIDs) such as low dose naproxen (250 mg) or tramadol (50 mg daily). If pain relief is obtained when Theramine is taken in combination with other pain medications, the drug dosage may be further tapered to lower levels under medical supervision. Theramine can also be used to manage the effective dose and dose-related side effects of pain medications. A randomized crossover study of patients with pain syndromes who were treated with naproxen in combination with Theramine demonstrated a 75% decrease in the effective dose of naproxen needed to achieve the same degree of pain reduction from 4 times to once daily. This study is described in greater detail in this monograph in the section, Clinical Validation of Theramine. The amounts of each ingredient consumed at the recommended doses of Theramine for pain reduction are provided in Table 1. 8|P a g e Revised03Mar2011 Theramine® Product Information Table 1. Theramine Composition mg/kg body weight1 Ingredient 1 į-aminobutyric acid (GABA) 1.5 – 12.0 choline bitartrate 1.0 – 7.7 L-arginine 0.6 – 4.6 Whey protein hydrolysate 0.6 – 4.6 L-histidine 0.4 – 3.1 L-glutamine 0.4 – 3.1 Metabromine 0.4 – 3.1 5-Hydroxytryptophan (griffonia seed, 95% w/w) 0.2– 1.9 Grape seed extract 0.2 – 1.5 L-serine 0.2 – 1.5 Cinnamon bark 0.2 – 1.5 Cocoa powder 0.2 – 1.5 Dosing range of 1 to 4 capsules daily SideEffectsandContraindications As with any amino acid therapy, headache, upset stomach, or dry mouth may be experienced in some people after beginning treatment with Theramine. These symptoms are mild and temporary, and readily managed by increasing fluid intake. The development of side effects from Theramine can be minimized by careful titration of the dosage. The ingredients in Theramine are regularly consumed in amounts normally found in foods or dietary supplements; therefore, adverse reactions associated with administration of Theramine are not expected to occur. Theramine is contraindicated in patients who may be hypersensitive to any component of an argininecontaining preparation. Theramine contains L-arginine which has been associated with side effects when consumed as a supplement; however, these effects are not observed when consumed at low doses as part of a formulation containing other amino acids. The adverse effects associated with L-arginine supplementation also appear to be dependent on the dosage regimen and are not observed when doses are divided instead of given as a single dose (7). A 2-capsule dose of Theramine contains 126 mg of L-arginine provided in a balanced formula with other amino acids and dietary factors. Side effects specific to oral supplementation with L-arginine have been reported at doses between 3 and 100 g/d, approximately 12 to 400 times the amount provided in Theramine in the recommended daily dose (126 mg/2 capsules) (7). L-arginine is generally well-tolerated at intakes of up to 15 g/d. The most common adverse reactions have been observed at the range of intakes between 15 30 g/d and include 9|P a g e Revised03Mar2011 Theramine® Product Information nausea, abdominal cramps, diarrhea, and vomiting. Some patients may experience symptoms at lower doses. Most of the side effects associated with L-arginine supplements have been reported at single doses >9 g (>140 mg/kg), particularly when consumed in regimens where total daily doses amount to >30 g/d. L-arginine supplements must not be taken alone by patients testing positive for HIV-1 infection, but may be consumed by these patients in combination with other amino acids and dietary factors as provided in Theramine. Long-term safety studies have not been conducted with L- arginine. Because it may stimulate growth hormone production, pregnant women and nursing mothers should avoid L-arginine supplementation. Individuals with renal or hepatic failure should also exercise caution with the use of supplemental L-arginine. Oral supplements of arginine and citrulline can increase local nitric oxide production in the small intestine which may be harmful under certain circumstances. AbbreviationsandDefinitionofTerms The abbreviations and terms used frequently in this monograph are summarized in Table 2. Table 2. Abbreviations and Definitions of Terms Term/Abbreviation Definition Adenosine Ubiquitous homeostatic neuromodulator; responsible for fine tuning neuronal function; exerts inhibitory effects on neuronal activity Anti-inflammatory Inhibitory effect on the synthesis and release of chemical and hormonal stimuli which initiate and sustain inflammation Antinociceptive Moderates nociceptor responsiveness to noxious stimuli which reduces pain Antioxidants Molecules or enzyme systems that inhibit injury to cells from reactive oxygen or nitrogen species Biogenic amine Biologically active substance that contains an amine group but does not have the characteristic structure of an amino acid, i.e., alpha carbon binding both an amino and carboxyl group Central sensitization Increased responsiveness of the spinal cord to all incoming signals which contributes to hyperexcitability of spinal cord neurons; originates from the release of excessive amounts of neurotransmitters from sensitized peripheral neurons C-fibers Small diameter afferents in peripheral nerves which convey information about pain and temperature to the dorsal horn of the spinal cord Cholinergic Neurons that synthesize, package, and release acetylcholine Dorsal horn Section of the spinal cord which is the primary center for processing nociceptive information; receives input from peripheral afferent fibers where it is encoded and transmitted for processing by higher nerve centers in the thalamus and cortex Excitatory Neurotransmitters Molecules released from presynaptic cells at terminal nerve endings which transmit action potentials to adjacent neurons by depolarization of postsynaptic cell membranes resulting in a decreased stimulus threshold for firing which increases the frequency and rate of transmission of action potentials 10|P a g e Revised03Mar2011 Theramine® Product Information Term/Abbreviation Definition GABAergic Neurons that synthesize, package, and release gamma-aminobutyric acid Glutamatergic Neurons that synthesize, package, and release glutamate Glutathione Potent cellular antioxidant synthesized from glutamate, cysteine, and glycine Inflammatory Mediators Serum proteins and cellular products released from activated inflammatory cells (lymphocytes, leukocytes, platelets) and damaged tissues which modulate the inflammatory response, e.g, cytokines and prostaglandins Inhibitory Neurotransmitters Molecules released from presynaptic cells at terminal nerve endings which transmit action potentials to adjacent neurons by hyperpolarization of postsynaptic cell membranes resulting in a increased stimulus threshold for firing which decreases the frequency and rate of transmission of action potentials Neuropeptides Polypeptides which function as neurotransmitters but are more widely diffused and have longer-lasting effects; may also function as hormones, e.g., prolactin, vasopressin Neurotransmitters Amino acids, biogenic amines, and other molecules that facilitate communication between the peripheral nervous system, spinal cord, and brain by generating a series of action potentials which are transmitted between neurons NMDA Receptors N-methyl-D-aspartate; subfamily of glutamatergic receptors which require a co-agonist for activation; implicated in generation and maintenance of central (spinal) states of hypersensitivity through enhancing transmission of nociceptive information; mediates events that are critical components of pathological and/or prolonged pain states Nociception Initiation, transmission, processing, and perception of pain Nociceptors Receptors at the terminal endings of peripheral afferent nerves which transduce noxious stimuli to receptor potentials Noxious stimuli Chemical, physical, or electrical signals that stimulate nociceptors in the area of tissue damage to initiate the sequence of electrochemical events that send the nociceptive information over afferent pathways to the brain Neuropeptides Polypeptides which function as neurotransmitters but are more widely diffused and have longer-lasting effects; may also function as hormones, e.g., prolactin, vasopressin Peripheral sensitization Increased responsiveness of primary afferent neurons as a consequence of factors that lower the threshold level for nociceptive stimulation Pronociceptive Effect that enhances the intensity and duration of pain by increasing nociceptor responsiveness to noxious stimuli Prostaglandins Inflammatory mediators synthesized from 20-carbon fatty acids by COX enzymes; may have proinflammatory (omega-6 fatty acids) or anti-inflammatory (omega-3 fatty acids) Serotoninergic Neurons that synthesize, package, and release serotonin (5-hydroxytryptamine) Substance P Sensory neuropeptide released from NMDA receptors at the terminal ending of C-fibers in response to increased electrical activity in nociceptors Targeted Cellular Technology™ A patented process that facilitates endogenous production, uptake, and utilization of neurotransmitter precursors 11|P a g e Revised03Mar2011 Theramine® Product Information MechanismofAction Theramine has been formulated to provide a balance of neurotransmitters with well-defined roles in the physiological mechanisms underlying the initiation and perception of pain. Pain is a sensory response to tissue damage which protects the area from additional stimulation in order to optimize healing and prevent infection. It is the outcome of a complex series of events originating with an interaction between unspecialized nerve endings (nociceptors) and noxious stimuli at or near the site of the injury (8-9). The noxious stimuli are transduced to receptor potentials by the nociceptors triggering a series of action potentials that are transmitted over peripheral afferent pathways to the spinal cord and higher nerve centers in the brain (8-12). The presence of inflammation increases the intensity of pain by sensitizing the nociceptors in the surrounding area to noxious stimuli causing them to fire more rapidly and frequently (11, 13-14). Transmission of nociceptive information by peripheral afferent fibers to the pain processing centers in the dorsal horn of the spinal cord is mediated by neurotransmitters. In the dorsal horn, these signals are encoded and output transmitted to the higher nerve centers in the thalamus and cerebral cortex. Neurotransmitters are amino acids, biogenic amines, or amino acid derivatives which function as mediators of physiological responses to physical, chemical, or electrical stimuli. Neurotransmitters are released from storage vesicles in presynaptic neurons in response to action potentials at the distal nerve endings where they bind to receptors on postsynaptic neurons (Figure 8). Neurotransmitter binding alters the resting membrane potential of postsynaptic neurons generating an action potential which is transmitted to the terminal ending of the neuron where the sequence of electrochemical events is repeated until the signal reaches specific processing centers in the brain. The same mechanism of neurotransmittermediated electrochemical events is involved in transmission of output from the brain to the target effector tissues or organs, and in transmission of signals originating within different regions of brain over the internal circuits between these regions. Figure 8. Neurotransmitter Activity in Presynaptic and Postsynaptic Neurons The rate of signal transmission between presynaptic and postsynaptic neurons in the central and peripheral nervous systems is dependent on the chemical nature of the neurotransmitter involved. Excitatory neurotransmitters released from presynaptic nerve terminals depolarize postsynaptic cell 12|P a g e Revised03Mar2011 Theramine® Product Information membranes which lowers the stimulus threshold for firing and increases the frequency and rate of transmission. Inhibitory neurotransmitters have the opposite effect of hyperpolarizing postsynaptic membranes which raises the stimulus threshold and decreases the frequency and rate of transmission. Although neurotransmitters can be classified as excitatory or inhibitory based on the primary effects they have on resting membrane potentials, these classifications do not always predict the response of the effector tissue or organ. Excitatory neurotransmitters can suppress a response by activation of inhibitory mechanisms and inhibitory neurotransmitters can activate a response by suppression of these mechanisms. Imbalances caused by deficiencies in one or more of the excitatory and inhibitory neurotransmitters, or changes in their binding affinities to postsynaptic receptors, will determine the intensity and duration of the signals transmitted (16-20). The primary neurotransmitters involved in the transmission of nociceptive signals are glutamate, GABA nitric oxide, acetylcholine, histamine, and D-serine. Glutamate is the major excitatory neurotransmitter of the central nervous system while GABA is the primary inhibitory neurotransmitter. Serotonin has excitatory activity while acetylcholine, nitric oxide and histamine can each exhibit both excitatory and inhibitory effects in the central and peripheral nervous systems depending upon the specific type and location of the respective receptors. D-serine influences nociceptive and inflammatory processes as an excitatory neurotransmitter in the brain. Imbalances caused by deficiencies in one or more of the excitatory and inhibitory neurotransmitters, or changes in their binding affinities to postsynaptic receptors, will determine the intensity and duration of the nociceptive signals generated (21-24). The amount of pain experienced resulting from tissue injury is influenced by nociceptor activity at or near the site of the injury. The responsiveness of neurons to pain signals is amplified by the presence of chemical or electrical phenomena which sensitize them to incoming signals (25-29). The accumulation of leukocytes, lymphocytes, and platelets at the site of tissue damage triggers the release of inflammatory mediators (cytokines and prostaglandins) which activates the corresponding receptors in a proportion of nerve fibers (13-14, 27, 30-31). Maintenance of inflammation after tissue damage has been repaired will ultimately lead to peripheral sensitization, which lowers the threshold level for nociceptive stimulation. Within the first hour after onset of inflammation, sensitization of peripheral neurons occurs and can persist over weeks in chronic inflammation (10). Sensitization of the central nervous system arises from the release of excessive amounts of neurotransmitters from sensitized peripheral neurons (11-12, 28, 32). The responsiveness of the spinal cord to all incoming signals is then amplified which contributes to hyperexcitability of spinal cord neurons. This hyperexcitable state potentiates the responsiveness of dorsal horn neurons to noxious mechanical and chemical stimuli (hyperalgesia), reduces the pain threshold of spinal nociceptors (allodynia), and increases the sensitivity of normal tissue to mechanical stimulation (8, 25-27, 32-35). Central sensitization is mediated by various transmitter/receptor systems on spinal cord neurons which include N-methyl-D-aspartate (NMDA) receptors. NMDA receptors are a subfamily of glutamatergic receptors concentrated in the dorsal horn of the spinal cord which increase hyperalgesia following tissue damage, nerve dysfunction, and surgery by potentiating spinal nociception (23, 31). NMDA receptors differ from other nociceptive receptors in that the ion 13|P a g e Revised03Mar2011 Theramine® Product Information channels involved in generation of action potentials are blocked by magnesium and therefore cannot be activated until the block is removed. Activation of NMDA receptors requires a co-agonist such as glutamate or acetylcholine to remove the magnesium block which triggers a massive depolarization that markedly increases neuron excitability resulting in enhancement of nociceptive signal transmission (23, 36). Although not directly involved in transmission of nociceptive information, NMDA receptors facilitate and sustain transmission of these signals and thus NMDA-mediated events are important contributors to pathological and/or prolonged pain states. Since the effects of NMDA receptor activity are slow and long-lasting, they contribute to all persistent clinically important types of pain including neuropathic and inflammatory pain. The activation of NMDA receptors is the key event in the development and maintenance of peripheral and central sensitization and a factor in modulation of the intensity of the nociceptor response to noxious stimuli (11-13, 28, 35, 37). Sensitized neurons establish the physiological basis for persistent or ongoing pain by spontaneously discharging at greater frequencies over extended periods. Persistent pain is an outcome of the hyperexcitability of dorsal horn neurons originating with severe or prolonged tissue or nerve injury (11, 27-28, 35, 38). Activation of nociceptors by sensitizing agents not only amplifies cellular responsiveness to pain stimuli, but also attenuates neuron sensitivity to antinociceptive receptor stimulants such as endogenous opioids (endorphins, dynorphins, and enkephalins) or exogenously administered opiates such as morphine (39-43). NMDA receptor activation further enhances the transmission of nociceptive information in the spinal cord by stimulating the release of nitric oxide and prostaglandins in the dorsal horn (29, 34, 40, 44). D-serine influences central nociceptive activity by effects on NMDA receptor activity in the brain (45-46). The release of glutamate following the activation of NMDA receptors increases the neuronal discharge rate and stimulates the release of substance P from the central terminals of spinal nociceptors. The release of substance P is triggered in response to an increase in electrical activity of NMDA receptors on the terminals of C-fibers, which are small-diameter peripheral afferents that receive input from peripheral nociceptors (32, 34, 37, 47-51). Substance P is a sensory neuropeptide which functions as a primary afferent neurotransmitter and mediator of central transmission with effects that are more widely diffused and longer-lasting than those of other neurotransmitters (32, 34, 37, 47, 49-50, 52-53). It facilitates the expression of nociceptive inputs which signal the intensity of noxious or aversive stimuli, promotes vasodilation and swelling, and stimulates the release of histamine from mast cells, which prolongs pain by sustaining inflammation-mediated neuron sensitivity (34, 37, 47, 49-53). Substance P is also believed to be an integral part of central nervous system pathways involved in pain associated with psychological stress (39, 50, 52). Central sensitization which is mediated by NMDA receptor activity is enhanced by increased levels of substance P and proinflammatory prostaglandin E2 (PGE2) in the spinal cord (37 49, 51-52, 54). Prostaglandins enhance the sensitivity of high threshold pain fibers by increasing neuron excitability, lowering the pain threshold, and potentiating the action of pain-producing stimuli (55-56). PGE2 is the dominant prostaglandin in the spinal nociceptive pathway (57). PGE2 binds to receptors at presynaptic endings of primary afferent neurons promoting the release of substance P and to receptors on postsynaptic spinal cord neurons where it produces changes in spinal cord responsiveness similar to peripheral 14|P a g e Revised03Mar2011 Theramine® Product Information inflammation (Schaible 2002 9). The release of pronociceptive neurotransmitters triggered by PGE2 binding depolarizes spinal cord neurons and blocks the effects of inhibitory neurotransmitters (57-58). Binding of prostaglandins to receptors on peripheral terminals of primary sensory nerves increases the sensitivity of high threshold pain fibers by enhancing neuron excitability which lowers the pain threshold thus potentiating the action of pain-producing stimuli. PGE2 levels are regulated by changes in the activity of the inducible form of cyclooxygenase (COX-2), which is increased by macrophages at sites of inflammation (56, 58-59). COX-2 is also expressed constitutively in small amounts in the central nervous system by glutamatergic neurons where it may participate in glutamate-mediated neurotransmission (57, 59). The effects of NMDA receptor activation and the release of substance P on induction, enhancement, and maintenance of pain suggest that treatments which target these effects may be useful in ameliorating persistent pain (28, 43, 47, 60-62). Central sensitization of the responses of spinothalamic tract neurons can be prevented by spinal cord administration of NMDA-glutamate receptor antagonists or substance P receptor antagonists (32). Achieving balance between excitatory and inhibitory neurotransmitters such as nitric oxide, glutamate, and D-serine can also prevent the sensitization of nerve fibers triggered by NMDA stimulation of spinal nociceptors (23). Because central sensitization facilitates efferent neuronal processes through which the nervous system influences the inflammatory response, regulation of NMDA receptor activity can be effective in management of pain resulting from inflammation. Successful pain management is complicated by the multifaceted relationships among different neurotransmitters and further complicated by the different types of pain often present in various combinations in different pain syndromes. Acute pain is a nociceptive phenomenon comprising visceral, somatic, and referred pain mechanisms whereas chronic pain is non-nociceptive dominated by neuropathic and psychogenic mechanisms which contribute to the physical and mental suffering and disability characteristic of this type of pain syndrome (10, 34, 63-65). The transformation of acute pain to chronic pain involves changes in neuronal pathways (plasticity) which include sensitization to stimuli and increased signal transmission in the central nervous system (12, 44, 66). Oral agents such as gabapentin which target GABAergic activity have been proven effective in treating neurogenic pain states (67-69). GABAergic receptors are concentrated in the pain-processing areas of the thalamus where they are expressed by approximately 10-25% of the neuron population (13, 68). Gabapentin is a widely prescribed analgesic which exerts its antinociceptive effects by selectively acting on NMDA receptors located in GABAergic neurons, thereby increasing the inhibitory activity of GABA. Selective serotonin reuptake inhibitors, which increase serotoninergic activity, have also been used for treatment of neurogenic pain, while D-serine facilitates analgesic effects by sensitizing opioid receptor systems to opioids, opioid-like agents, and other analgesics (8, 45, 69). 15|P a g e Revised03Mar2011 Theramine® Product Information The Inflammatory Cascade Inflammation is a response to cellular injury initiated by infection or trauma and mediated by serum proteins and cellular products released from accumulated phagocytic leukocytes, platelets, and lymphocytes at the site of the injury. Among these cellular products are cytokines (bradykinin), lymphokines, (interleukins and interferons), eicosanoids (prostaglandins and leukotrienes), histamine, chemotaxins, platelet aggregating factors, and neuropeptides. These substances are responsible for a wide range of effects including changes in vascular permeability and modification of the electrochemical properties of cell membranes which contribute to the cellular and physiological effects of inflammation. Clinical manifestations of inflammation are redness, swelling, loss of function, and increased body temperature. Inflammation is a part of the normal innate immune response, but can cause excessive damage to host tissue when it is uncontrolled and sustained after tissue repair is complete. Hyperexpression of cellular adhesion molecules, sequestration of leukocytes in areas where they are not normally found, and overproduction of inflammatory mediators are responsible for the destructive effects of inflammation. Although intended to be protective as an acute response, chronic inflammation is implicated in the pathology of autoimmune diseases (multiple sclerosis, rheumatoid arthritis, type I diabetes, lupus erythematous), cardiovascular disorders (angioplasty re-stenosis, by-pass graft occlusion, transplant vasculopathy), and other disorders (fibromyalgia, asthma, inflammatory bowel disease, and transplant rejection). ScientificSupportforUseofTheramineinManagementofPainSyndromes The use of Theramine in management of pain syndromes is supported by experimental and clinical data which have identified specific roles for each ingredient in the mechanism of pain reduction. An optimal balance between the activities of excitatory and inhibitory neurotransmitters involved in the transmission of nociceptive information is essential to management of pain (23, 25). Inflammation disrupts this equilibrium by chemical modification of nociceptive activity which recalibrates the levels of neurotransmitters required to achieve balance. The relationship between the excitatory effects of glutamate and inhibitory effects of GABA are primary determinants of the level of pain transmission. To reduce pain, the balance between neurotransmitters with pronociceptive effects and those with the opposing antinociceptive effects must be restored (23). Recent evidence suggests that central pain originating with central nervous system damage may be attributed to a derangement in neurotransmission between the sensory thalamus and sensory cortical areas involving GABA (24). In addition, loss of GABAergic activity in spinal cord neurons contributes to the allodynia and hyperalgesia observed after peripheral nerve injury (68). Central pain attributed to relative hypofunction of GABAergic inhibition at both the thalamic and cortical levels may be controlled by either potentiating GABAergic neurotransmission or by inhibiting the opposing effects of glutamatemediated transmission. If enhanced nociceptor sensitivity to noxious stimuli is not due to increased activity of excitatory neurotransmitters such as glutamate, loss of inhibitory controls would be the likely factor involved (23). Apoptosis of neurons in the peripheral and central nervous systems induced by loss of inhibitory systems and the resultant neuronal sensitization is often an outcome of nerve injury (neuropathic) and other nervous system diseases (neurogenic) (12). The spread of local nerve injuries to distant parts of the peripheral and central nervous systems is facilitated by attenuation of central pain 16|P a g e Revised03Mar2011 Theramine® Product Information inhibitory mechanisms in addition to upregulation of nitric oxide synthesis in axotomized neurons, deafferentation hypersensitivity of spinal neurons following afferent cell death, and long-term potentiation of spinal synaptic transmission. The nociceptive effects of neurotransmitters co-localized within the same neurons are also influenced by balance in the relative amounts released (38, 64). Some of these neurotransmitters may act as co-agonists with synergies that amplify the effects of each and others may act as antagonists with opposing activities that mitigate these effects. Balance is also important in the regulation of neurotransmitter activity by mechanisms involving feedback inhibition. The increase in nociceptor sensitivity to noxious stimuli which is mediated by release of glutamate from NMDA receptors in the spinal cord and brain is controlled by the subsequent increase GABAergic activity also mediated by glutamate (38, 67, 70). This increase in GABAergic activity inhibits the release of substance P from NMDA receptors as well as the additional release of glutamate from NMDA receptors (34, 47, 64, 70). Pain reduction is optimized by achieving a balance between the pronociceptive effects of excitatory neurotransmitters which are normally opposed by the antinociceptive effects of inhibitory neurotransmitters (64). The loss of inhibitory controls may enhance nociceptor sensitivity to noxious stimuli even in the absence of increased excitatory activity. Different pain states may not respond similarly to changes in inhibitory transmitter systems since the activity of these systems can be influenced by variations in opioid, adenosine, and GABA transmission in the spinal cord. For example, hyperalgesia might be balanced by inhibitory effects of neurotransmitter systems in inflammatory conditions, but not in neuropathic states associated with nerve damage. The effects of serotonin on nociceptive transmission are largely mediated by the activity of the 5-HT3 receptor in the central and peripheral nervous systems which modulates the release of acetylcholine, GABA and substance P, as well as serotonin itself (22, 71). The pronociceptive activity of serotonin results from stimulation of peripheral afferent fibers and enhancement of the transmission of nociceptive information to the spinal cord and pain perception centers in the brain (34, 72-73). The antinociceptive effects of serotonin are mediated by inhibition of the release of substance P and amplified by stimulation of GABA, acetylcholine activity, and the release of adenosine from peripheral afferent fibers (C-fibers) (13, 22, 28, 47, 74-76). Adenosine receptors in the spinal cord exhibit antinociceptive activity in acute nociceptive, inflammatory, and neuropathic pain tests (77). Endogenous adenosine systems also contribute to the antinociceptive properties of caffeine, opioids, noradrenalin, tricyclic antidepressants, and transcutaneous electrical nerve stimulation. The antinociceptive activity of acetylcholine is due in part to cholinergic-mediated stimulation of the synthesis and release of serotonin (64, 78). Acetylcholine also inhibits the activity of receptors localized on peripheral afferent nerve terminals and intrinsic dorsal horn neurons, decreases neuronal sensitivity and firing, inhibits NMDA receptor activity and production of substance P, and suppresses the production of proinflammatory cytokines by stimulating the parasympathetic nervous system (34, 37, 64). The parasympathetic-cholinergic anti-inflammatory pathway represents a physiological neuroimmune mechanism for regulation of innate immune function and control of inflammation (78). 17|P a g e Revised03Mar2011 Theramine® Product Information The effects of nitric oxide on nociception are dose-dependent with exacerbation of pain at high doses and inhibition at low doses (79). Nitric oxide is synthesized from arginine in a hydrolytic reaction catalyzed by one of three isoforms of nitric oxide synthase (NOS); however, inducible NOS (iNOS) is the only isoform which is active in inflammatory processes (26, 80-84). The activity of this enzyme is induced in neurons by inflammatory mediators concentrated in the area of inflammation (79). The synthesis of nitric oxide in dorsal horn neurons is increased by the induction of iNOS by NMDA receptor-mediated stimulation of C fibers and neuronal nitric oxide synthase (nNOS) (34, 40). The amount of nitric oxide produced by NOS is dependent upon the amount of arginine available to the enzyme and is controlled by upregulation of arginine transport and the extent of competition between this enzyme and arginase (Figure 4) (82-83). Nitric oxide modulates spinal cord nociception through effects on the transmission of pain signals from dorsal horn neurons to cerebral centers in the brain (85). It blocks the transmission of afferent pain signals in the spinal cord, activates natural opioids, and stimulates the production of anti-inflammatory prostaglandins (26, 82, 85-86). Pain can thus be modulated by NMDA-mediated changes in neuronal NOS activity which control nitric oxide production (13). Nitric oxide also influences the regulation, proliferation, survival, and differentiation of neurons (82). Although at high concentrations nitric oxide is deleterious to all cells, physiological amounts have anti-inflammatory effects and potent cytoprotective effects which are especially pronounced in neurons (87-90). While nitric oxide influences the efficiency of NMDA-mediated synaptic transmission, a deficiency blocks the neurotrophic and neuroprotective effects of NMDA receptor activity which has a profound impact on neuronal function. Glutamine plays an important role in modulation of pain as a precursor of glutamate especially during periods of increased demand because elevated levels of glutamate can be toxic to the central nervous system (91-93). This function of glutamine is particularly critical if nerve damage is present since extracellular glutamate levels are further increased by the additional glutamate which is released into the extracellular space from the damaged nerves. Glutamine also modulates the inflammatory response by attenuation of pro-inflammatory cytokine production (94). Glutamine and glutamate are also utilized as precursors of glutathione, a major intercellular antioxidant which protects against DNA damage from accumulation of the large amounts of reactive oxygen species produced by inflammatory cells during cytotoxic activities (95). Under certain conditions, glutathione can serve as an intracellular reservoir of nitric oxide in its nitrosylated form (S-nitrosoglutathione). Glutamate exhibits nociceptive effects in the brain and dorsal horn and modulates inflammatory processes through activation of the hypothalamus-pituitary-adrenal (HPA) axis which triggers the synthesis and release of glucocorticoids (54, 59, 95). Interactions between glutamate and acetylcholine have been identified in mediation of many neurological functions (21, 36, 96-100). Glutamatergic neurons in the central nervous system are concentrated in areas of high cholinergic activity. Under conditions where glutamatergic receptor activity is inhibited, cholinergic transmission is stimulated and its receptors upregulated in the hypothalamus (36). Histamine has both antinociceptive and pronociceptive activity as well as proinflammatory and antiinflammatory effects depending upon the particular histamine receptor involved (3, 101-102). Histamine 18|P a g e Revised03Mar2011 Theramine® Product Information is produced from histidine in a decarboxylation reaction catalyzed by histidine decarboxylase. Many neuropeptides rely on histaminergic transmission by H2 receptors for their analgesic effects and H1 receptors for nociceptive effects (13). Antinociception and stress-induced analgesia in the central histamine system are mediated by activation of H2 and H3 receptors. Histamine decreases nociceptive signal transmission by inhibition of NMDA receptor activity and acting synergistically with nitric oxide while it promotes nociception by stimulating the release of substance P from peripheral afferent nerve terminals (103). Neurogenic inflammation is also controlled by the H3 receptor through local feedback loops linking neurons to mast cells (3, 104). Whey protein hydrolysate comprises several proteins and peptides with anti-inflammatory, immunomodulatory, and antioxidant properties (105-106). In particular, Į-lactalbumin and ȕlactoglobulin interact with opioid receptors indicating that these proteins also have anti-nociceptive effects (105-107). Whey is a high biological value protein derived from milk which contains all 22 amino acids necessary for human protein synthesis and metabolism including the neurotransmitter precursors, tryptophan, arginine, and histidine. A summary of the roles of these and other ingredients provided in Theramine in the management of pain, is presented in Table 3. Table 3. Ingredient Effector Molecule GABA Role of Theramine Ingredients in Nociception Effects Roles GABA Inhibitory neurotransmitter; Antinociceptive Dampens pain signals in the spinal cord and brain; inhibits NMDA receptor activity; inhibits release of substance P and glutamate from dorsal horn neurons Choline Acetylcholine Inhibitory neurotransmitter; Antinociceptive; Anti-inflammatory Promotes synthesis and potentiates the effects of nitric oxide and serotonin; inhibits peripheral afferent nerve terminal receptor activity; reduces neuronal sensitivity and firing; inhibits NMDA receptor activity and production of substance P; suppresses proinflammatory cytokines by activation of the parasympathetic nervous system Glutamine Glutamine Facilitator of neurotransmitter precursor uptake Promotes synthesis of neurotransmitters; source of glutamate; protects against neurotoxicity by preventing extracellular glutamate accumulation in the central nervous system Glutamate Excitatory neurotransmitter; Pronociceptive Primary neurotransmitter of the NMDA receptor; Inhibits NMDA receptors through activation of GABAergic receptors; precursor of GABA ; interacts with acetylcholine Glutathione Antioxidant; Immunomodulator Prevents tissue oxidative damage due to the effects of inflammation 19|P a g e Revised03Mar2011 Theramine® Product Information Ingredient Effector Molecule Effects Roles 5-OH-tryptophan Serotonin Excitatory neurotransmitter; Antinociceptive; Pronociceptive Decreases pain signals in the spinal cord and brain; stimulates nociceptive peripheral afferent fibers; ; inhibits NMDA receptor activity; inhibits release of substance P; increases adenosine production by C-fibers which release substance P; acts synergistically with GABA and acetylcholine to amplify antinociception; stimulates release of histamine and serotonin from mast cells Serine D-Serine Neurotransmitter; Antinociceptive Regulates NMDA receptor activity in the brain; sensitizes opioid receptor systems to opioids, opioid-like agents, and other analgesics (natural opioids include endorphins, enkephalins, and dynorphins and synthetic opioids include morphine) Arginine Nitric Oxide Inhibitory and excitatory neurotransmitter; Antinociceptive; Pronociceptive; Neuroprotective; Anti-inflammatory Inhibits pain at low doses and exacerbates pain at high doses; inhibits transmission of afferent pain signals in the spinal cord; inhibits NMDA receptor activity; activates natural opioids; stimulates production of anti-inflammatory prostaglandins; neuroprotective at physiological levels Histidine Histamine Excitatory neurotransmitter; Antinociceptive; Proinflammatory; Anti-inflammatory Receptor dependent effects on nociception; active in the spinal cord and brain; stimulates NMDA receptor activity; acts synergistically with nitric oxide; modulates inflammation through increasing production of glucocorticoids Cocoa Powder Caffeine Adenosine antagonist Binds to adenosine receptors to disinhibit the adenosine brake which promotes the inhibitory effect of adenosine on neuronal activity (76, 108111) Grape seed extract Polyphenols Antioxidant; Anti-inflammatory Protects against tissue oxidant damage due to inflammation; prevents attenuation of the response to increased availability of neurotransmitter precursors (112 -115) Whey Protein Hydrolysate Į-lactalbumin, ȕ-Lactoglobulin, Glycomacropeptide, Lactoferrin Antinociceptive Immunomodulator Antioxidant Anti-inflammatory Į-lactalbumin and ȕ-lactoglobulin reduce pain through interactions with opioid receptors; other peptides reduce the effects of inflammation on pain (105-107, 109-111) Metabromine Caffeine, Theobromine, Procyanidins Adenosine antagonist Interacts with adenosine receptors localized on axon terminals of excitatory neurons; disinhibits the adenosine brake which promotes the inhibitory effects of adenosine on neuronal activity (108-110) NutritionalRequirementsinPainSyndromes The nutritional requirements of most interest to patients with pain syndromes are the nutrients and dietary factors which support the synthesis and activities of neurotransmitters involved in the transmission and perception of nociceptive information (38, 110, 116-120). These include arginine, choline, GABA, glutamine, histidine, 5-hydroxytryptophan, and serine which modulate nociceptor responsiveness to 20|P a g e Revised03Mar2011 Theramine® Product Information noxious stimuli. Theramine is formulated to ensure the availability of the neurotransmitters acetylcholine, nitric oxide, glutamate, serotonin, D-serine, histamine that modulate pain by optimizing nociceptive activity in the central and peripheral nervous systems using Targeted Cellular Technology to control the timing of the release of these ingredients. Balance in production and release of neurotransmitters is important to neurotransmission because it is the highly integrated functions of these neurotransmitters and the complexity of the multiple feedback loops between them that determine the net input received by the brain. These interactions explain why an imbalance in the intake of a nutrient or dietary factor which supports the synthesis or activity of any one neurotransmitter can influence the activities of the others, potentially inducing absolute and relative deficiencies (121-124). The concept that nutrient requirements are modified by disease has been recognized for more than 30 years, and is supported by studies which have shown changes in plasma, urinary, and tissue levels of nutrients with modified intakes of these nutrients that correspond to changes in physiological endpoints reflective of specific pathologies (Table 4) (125). These requirements can be estimated by determining the level of intake at which a physiological response is improved indicating that the balance between intake and metabolic demand has been favorably modified. The nature of the pathological characteristics of a disease will determine the relative amounts of nutrients needed to restore balance between intake and demand (118 -120, 125-129). For example, improvement in perceived intensity of back pain following consumption of supplemental amounts of 5-hydroxytryptophan, arginine, and glutamine from Theramine suggests that additional allowances for tryptophan and the other amino acids are needed by individuals with pain syndromes (Figure 9). (4, 124-129). The degree of coordination between the activities of different neurotransmitters is an important consideration in assessing the amounts of dietary precursor needed (110, 119, 130-134). Diseases with pathologies that involve imbalances in neurotransmitters will increase the requirements for certain amino acids and other dietary precursors to restore homeostasis (4, 120, 125 131). For most amino acids and dietary precursors of neurotransmitters, neuronal uptake is a concentration-driven process; therefore, intakes of precursors must be high enough to increase the extracellular to intracellular ratio to a level that will drive a rapid rate of uptake (116, 120, 130, 131- 132, 135-136). The rate of precursor uptake by target neurons is important to neurotransmitter synthesis because the enzymes involved are found only in these neurons and thus the amount of substrate available is the limiting factor in neurotransmitter production (120, 136). As blood levels of dietary precursors rise in response to increased intakes, the concentration-dependent rate of precursor uptake by target neurons is increased, making more substrate available for neurotransmitter production and subsequent release (132, 137-139). Changes in intakes of dietary precursors of these neurotransmitters will therefore influence physiological responses by affecting neurotransmitter availability (116, 121-122, 126, 128-129, 132, 135-142). A large body of peer-reviewed published data supports the basis for increased requirements of choline (120, 143-144), arginine (83, 86, 145-146, 148-149), tryptophan (117, 127, 150-151), glutamine (144, 149), serine (148, 152), and histidine (150, 153-154) in conditions which depend on neurotransmitter balance. Patients with pain disorders show decreased blood levels of specific amino acids despite having a sufficient intake of protein indicating that the needs for these amino acids are selectively increased in these patients (117, 149, 155-159). This observation may be explained by the competitive demands for 21|P a g e Revised03Mar2011 Theramine® Product Information certain amino acids by different metabolic pathways which decreases the supply of neurotransmitters available to function in the pain process (See Section on Metabolism in this monograph). Certain physiologic and biochemical mechanisms must exist in order for nutrient consumption to affect neurotransmitter synthesis (136). These conditions are listed below. The extent to which neurotransmitter synthesis in any particular aminergic neuron is affected by changes in precursor availability will vary directly with the firing frequency of the neuron. Consequently, precursor administration can produce selective physiologic effects by enhancing neurotransmitter release from some but not all of the neurons potentially capable of utilizing the precursor for the particular effect. It is also useful in predicting when administering the precursor might be useful for amplifying a physiologic process, or for treating a pathologic state. Conditions that Support Effects of Dietary Precursors on Neurotransmitter Synthesis 1. Absence of significant feedback control of plasma precursor levels 2. Ability of plasma precursor levels to control influx into or efflux from the central nervous system 3. Presence of a low-affinity (unsaturated) transport system mediating the flux of precursor between blood and brain 4. Low-affinity kinetics of enzyme that initiates conversion of precursor to neurotransmitter 5. Lack of in vivo end-product enzyme inhibition by the neurotransmitter Syndromes associated with musculoskeletal pain have been the most extensively studied with regard to identifying the increased needs for certain amino acids in moderation of pain. Patients with these syndromes and other types of pain disorders exhibit reduced blood levels of tryptophan, arginine, glutamate, histidine, and serine (117, 142, 154, 158). Measurements of total serum tryptophan (p = 0.002) as well as 6 other amino acids including histidine (p = 0.001) and serine (p = 0.028) yielded significantly lower values for patients with fibromyalgia (fibrositis syndrome) than for controls (117). Plasma histidine and serine levels were also found to be significantly lower (p < 0.01) in another study of patients with this condition (151). Fibromyalgia patients also had significantly lower plasma levels of total essential amino acids compared with controls suggesting that a generalized defect in amino acid homeostasis may be present among affected individuals (157). The possibility that relative deficiencies in certain amino acid precursors are associated with pain syndromes is also supported by clinical observations. In a study of patients with secondary fibromyalgia syndrome, individual measures of pain intensity assessed by the tender point index (TPI), an examinationbased measure, co-varied with glutamine levels (158). Severe neurological symptoms attributable to deficiencies in serine have also been reported underscoring the importance of this amino acid in brain 22|P a g e Revised03Mar2011 Theramine® Product Information tissue, particularly as a precursor of phospholipids and D-serine (152). Low blood levels of tryptophan accompanied by altered tryptophan metabolism have been frequently reported in patients with pain disorders and have also been associated with decreased brain serotonin concentration (127, 130, 137-138, 150, 155-156, 160-161, 163-165). These patients also commonly exhibit reduced blood levels of 5hydroxytryptophan, arginine, choline, GABA, histidine, and serine. Moreover, they respond to oral intake of amino acids with favorable changes in physiologic endpoints and improvements in clinical symptoms associated with pain, thus supporting the increased need for specific amino acids to normalize blood levels in patients with pain disorders (3, 86, 119-120, 125, 137-138, 143, 149, 156-157, 159-161, 163165). In conditions where tryptophan metabolism is altered, intake of 5-hydroxytryptophan, the metabolic intermediate in the conversion pathway of tryptophan to serotonin, would be a more effective approach for restoring balance in serotonin levels than administration of tryptophan (Figure 1) (See section on Metabolism in this monograph). Therapeutic administration of 5-hydroxytryptophan has been shown to be effective in treating a wide variety of conditions that involve serotoninergic activity including depression and insomnia (6). Oral 5-hydroxytryptophan is well-absorbed with approximately 70% of the dose measured in blood following intake. This molecule crosses the blood-brain barrier and effectively increases central nervous system synthesis of serotonin. The appearance in cerebrospinal fluid of increased amounts of 5-hydoxyindolacetic acid, the primary metabolite of the breakdown of serotonin, following oral-administration of 5-hydroxytryptophan confirms that supplemental intake of this precursor not only increases production of serotonin but that the neurotransmitter is released by serotoninergic neurons (129, 137-138). By affecting the production and release of neurotransmitters, changes in intakes of precursor nutrients can influence the physiological functions dependent on these neurotransmitters (116, 120, 123, 126, 128-133, 135, 138, 143, 149, 152,166-169). Some dietary precursors such as arginine which depend on membrane transporters for cellular uptake are not as affected by extracellular concentrations as those dependent on a favorable concentration gradient, but instead depend on factors that compete for binding sites on the transport proteins (126, 147, 169). The primary determinant of plasma arginine levels and thus nitric oxide production is dietary arginine because endogenous synthesis does not increase sufficiently to compensate for depletion, increased turnover, increased requirement, or inadequate supply of the amino acid (148, 170-171). Arginine requirements are therefore influenced by metabolic utilization and factors that affect rates of de novo synthesis (30-31, 89, 147, 166). Utilization of arginine is increased by citrulline which upregulates iNOS and eNOS activity and by glutamate which increases iNOS and nNOS activity in the brain and other nervous system tissues (85). In a study of stable short-bowel patients, a decrease in plasma arginine levels was observed after 5 days of consuming an arginine-free diet and was accompanied by a decrease in levels of citrulline, indicating that synthesis of arginine from citrulline in the urea cycle had been increased, but the rate was not sufficient to maintain plasma arginine levels when intake was inadequate (147, 172). Thus an increase in the demand for arginine to support increased nitric oxide synthesis would require an increase in arginine intake to satisfy the demand. The arginine body pool is also influenced by intake of glutamine and glutamate which contribute to levels of ornithine and citrulline, intermediates of the urea cycle which can be converted to arginine (Figure 3 23|P a g e Revised03Mar2011 Theramine® Product Information and Figure 4). Glutamine also plays a role in regulation of whole body arginine homeostasis through inhibitory effects on arginine utilization that reduce nitric oxide synthesis (172). It also increases in arginine utilization for synthesis of urea as the primary donor of amino groups to arginine. Despite being the most abundant amino acid in the blood, glutamine levels are rapidly depleted by catabolic processes. Plasma glutamine levels do not appear to be affected by the accelerated release of glutamine from skeletal muscle during periods of increased demand which suggests that glutamine availability is reduced by competition for tissue uptake and dietary intake becomes rate limiting for glutamine-dependent processes (94, 173). Although a clearly defined glutamine deficiency syndrome has not been described, endogenous production is not sufficient to meet the increased and altered tissue demands imposed by trauma, sepsis, infection, and inflammation (94). Since most naturally-occurring food proteins contain 4% to 8% of their amino acid residues as glutamine, an average of less than 10 g of dietary glutamine is likely to be consumed daily. Studies in stressed patients indicate that considerably larger amounts of glutamine (2040 g/day) may be necessary to maintain glutamine homeostasis (173). The need for glutamine is also increased by a demand for glutathione to protect against the increased production of reactive oxygen during inflammatory activities. As a precursor of glutathione, glutamine supports the activity of the glutathione peroxidase antioxidant enzyme system (174-175). Supplemental glutamine is important for maintenance of tissue glutathione levels that protects against cellular damage from oxidative stress (175). Supplemental glutamine is also protective against damage due to oxidative stress by effects that inhibit inflammation. The need for supplemental histidine intake is also increased by inflammation to satisfy the increased demand for histamine production. After 8 weeks of consuming a low histidine (10 mg/day), low nitrogen (6.3 g/day) diet, a significant decrease in 24-hour urinary free histidine was observed in 7 healthy men associated with a reduction in serum hemoglobin concentration indicating that the contribution of hemoglobin to the histidine body pool is limited (167). Addition of histidine to the diet resulted in an increase in serum hemoglobin concentration to baseline levels within 2 weeks associated with a corresponding increase in urinary histidine. Acetylcholine is produced in the terminal endings of cholinergic neurons and in regions of the brain where choline acetyltransferase is concentrated (Figure 5). Under steady state conditions, the brain enzyme is not completely saturated, thus the rate of acetylcholine production is driven by the availability of choline and acetyl CoA (139, 176). Dietary choline is the primary contributor to plasma choline levels accounting for a greater proportion of the plasma concentration than de novo synthesis (155, 161, 163, 168, 177-178). The rate of choline transport across the blood brain barrier is increased by an amount proportional to the increase in serum concentration and is followed by an increase in the release of acetylcholine from cholinergic neurons (139). In the brain, choline is incorporated into the membrane phosphatidylcholine pool and released when the demand for acetylcholine is increased; however, the appearance of choline in cerebrospinal fluid confirms that there is a pool of free choline in the brain (179-180). In a normal physiological state, most of the choline utilized for acetylcholine synthesis is obtained from hydrolysis of phosphatidylcholine (179, 24|P a g e Revised03Mar2011 Theramine® Product Information 181-183). When demand for acetylcholine is increased over prolonged periods, dietary choline becomes an increasingly more important source of precursor. If a supplemental source of choline is not provided to meet these increased demands, loss of membrane phosphatidylcholine will eventually compromise cell membrane function and trigger apoptosis (178-179, 182-187). The importance of dietary precursor intake to plasma levels and thus availability of precursor for neurotransmitter synthesis was demonstrated by a study showing an increase in serum choline concentration following treatment with choline chloride. Other studies have confirmed that exogenous choline can be utilized by central cholinergic neurons as a precursor for acetylcholine synthesis (180). An increase in plasma choline promotes the expression of high affinity choline transporters on cholinergic neurons which regulate the synaptic availability of choline and facilitate the release of acetylcholine from these neurons (155, 178-179). Synaptic acetylcholine levels are regulated by a negative feedback mechanism in which accumulation of the neurotransmitter inhibits transporter activity on cholinergic neurons to prevent further uptake of choline. Anticholinergic drugs such as chlorpromazine, atropine, and cholinesterase inhibitors decrease acetylcholine release by inhibition of these transporters (183, 188-189). Although serum choline levels are decreased by a choline-free diet, brain choline levels remain relatively stable indicating that the brain is given metabolic priority at the expense of other tissues when the amount of free choline available is limited (180). Brain phosphatidylcholine levels decrease in parallel with the decrease in serum choline which further suggests that brain choline concentration is maintained within narrow limits at the expense of larger tissue pools of phosphatidylcholine and other phospholipid precursors (serine and ethanolamine) (168, 177, 179, 182). Data from an experimental study in rats showed that brain choline concentration increased within 5 hours following oral administration of choline chloride (180). The consumption of a choline-free diet for 7 days lowered serum choline and brain phosphatidylcholine concentration suggesting that choline kinase, the controlling enzyme in phospholipid synthesis, is unsaturated with substrate in vivo and thus may serve as a modulator of the response of brain choline concentrations to alterations in the supply of circulating choline. Clinical evidence of a human choline deficiency was first reported in adults receiving total parenteral nutrition (TPN) (190-191). These patients exhibited hepatic morphologic and aminotransferase abnormalities which were reversed by choline-supplemented TPN. The effects of inadequate choline intakes on physiological endpoints are rapidly observed. Low blood levels of choline indicate that the requirements for the dietary precursors are not being met at current levels of intake (116, 139, 177, 179). Clinical signs of deficiency were documented in men with otherwise normal nutritional status after consuming a choline-deficient diet for a period of < 3 weeks (192). Changes in blood and urine markers of organ dysfunction (muscle and liver enzymes) were also been reported in these men. Decreases in plasma levels of choline and phosphatidylcholine accompanied by elevated alanine aminotransferase, a biochemical marker of liver damage, and elevated creatine kinase, a biological marker of muscle damage, have also been observed with a dietary choline deficiency (185, 192 -196). Serum choline levels are more responsive to increased choline intake than to a choline deficiency with increases of as much as 52% observed with choline supplementation compared with decreases of 20% with a choline-deficient diet (126, 179). 25|P a g e Revised03Mar2011 Theramine® Product Information Specific neurotransmitter deficiency syndromes related to inadequate intakes of amino acid precursors have been identified for GABA and serotonin. A syndrome characterized by a basic depressive state, sleep disorders, nuchal headache, and other clinical symptoms was attributed to a GABA deficiency based on the observation of a rapid reversal of these symptoms following administration of an enzyme inhibitor of GABA catabolism (197). Several findings also raise the possibility that inadequate intakes of tryptophan may be related to a brain serotonin deficiency in patients with fibromyalgia/fibrositis. An inverse relationship between blood tryptophan concentration and severity of musculoskeletal pain which was accompanied by significantly lower levels of serum tryptophan (p = 0.002) has been reported in patients with these conditions compared with healthy adults suggesting that a functional deficiency of serotonin may be involved in the pathology of fibromyalgia-related pain (150-151). A possible link between inadequate intake of tryptophan and a deficiency of serotonin is also supported by evidence of altered tryptophan metabolism associated with low blood tryptophan levels (170). A trend towards lower levels of plasma tryptophan was associated with a significantly lower tryptophan membrane transport ratio (p<0.01) in patients with primary fibromyalgia/fibrositis compared with controls indicating that insufficient amounts of tryptophan were reaching target tissues when plasma levels were below normal (151). A specific neuroendocrine response (i.e., prolactin release with tryptophan infusion) suggestive of postsynaptic serotonin receptor supersensitivity was associated with a 15 to 20% reduction in fasting total plasma tryptophan levels in 22 healthy subjects consuming a tryptophan-restricted diet (198). A summary of the scientific support for the increased requirements of specific amino acids for patients with pain disorders is found in Table 4. Table 4. Observations Supporting Increased Nutrient Needs in Pain Disorders Clinical Observations and Associated Biochemical Findings Nutrient Blood/Tissue/Urinary Levels Glutamine/Glutamate Low blood levels Increased pain intensity in patients with secondary fibromyalgia; low tissue glutathione levels; GABA deficiency characterized by a basic depressive state, sleep disorders, nuchal headache; loss of synaptic inhibition; seizures Tryptophan Low blood and brain levels Low membrane transport ratio; postsynaptic serotonin receptor supersensitivity; inverse relationship with severity of musculoskeletal pain; depression, behavioral changes, fibromyalgia/fibrositis syndrome; increased serotonin metabolites in cerebrospinal fluid following supplementation; serotonin deficiency Arginine Low plasma levels Low serum levels of arginine metabolites (e.g., ornithine, citrulline); decreased rate of nitric oxide synthesis; increase in plasma arginine and nitrates, and exhaled nitric oxide with supplementation Histidine Low 24-hour urine levels Decreased serum hemoglobin (source of histidine); fibromyalgia/fibrositis syndrome Low blood levels 26|P a g e Revised03Mar2011 Theramine® Product Information Clinical Observations and Associated Biochemical Findings Nutrient Blood/Tissue/Urinary Levels Serine Low blood levels Loss of sensitivity to both natural and synthetic inhibitors of pain Choline Low plasma levels Decreased activity of NMDA receptors; diminished responses to GABA and serotonin; decreased parasympathetic autonomic nervous system activity; increased creatine phosphokinase and alanine transaminase; myocyte and lymphocyte apoptosis ClinicalValidationofTheramine The relationship between intakes of dietary precursors and production of the corresponding neurotransmitters has been validated by observations of improvements in neurotransmitter-mediated clinical outcomes with supplemental intakes of these dietary factors (38, 119, 126-128, 133, 137-139, 143, 155, 198-204). A change in the levels of a neurotransmitter in the blood and/or its metabolites in cerebrospinal fluid following ingestion of a dietary precursor from a medical food reflect the uptake and utilization of the nutrient or dietary factor for synthesis of the neurotransmitter by target cells, thus demonstrating the biological availability of dietary precursors and the clinical utility of the medical food as a source of these precursors (118 -122, 125, 132-134, 137-138, 143-144, 149, 155-156, 160-163, 165, 177-178, 180, 182, 184-187, 203-210). The clinical benefits which may be obtained from medical foods can be validated by the observed changes in biological, physiological, and clinical endpoints following ingestion by individuals with specific conditions. If an individual with a chronic pain disorder ingesting a medical food containing 5hydroxytryptophan shows an increase in serum levels of the molecule following ingestion (biological availability) which is associated with increased concentrations of serotonin metabolites in cerebrospinal fluid (physiological response) and an improvement in pain measurement (clinical response), the clinical benefit of this medical food as a source of precursors for serotonin production has been validated. Theramine has been formulated with specific ratios of arginine, choline, GABA, glutamine, histidine, 5-hydroxytryptophan, and serine using Targeted Cellular Technology to control the timing of the release of each ingredient. If sufficient amounts of these nutrients are not available, or their availability is not well-synchronized with demand, imbalances in neurotransmitter activity contributing to acute and chronic pain disorders may result (21-24). 27|P a g e Revised03Mar2011 Theramine® Product Information BiologicalAvailability The biological availability of 5-hydroxytryptophan and L-arginine which are provided in Theramine has been demonstrated by changes in blood levels of the respective neurotransmitters observed following ingestion of these supplemental amino acids. Within 60 minutes after administration of 2000 mg of 5-hydroxytryptophan, blood serotonin levels increased by more than 3-fold, confirming that 5-hydroxytryptophan is well-utilized by target tissues as a precursor of serotonin (Figure 9). Figure 9. Effect of 5-Hydroxytryptophan Supplementation on Blood Serotonin Levels 28|P a g e Revised03Mar2011 Theramine® Product Information The biological availability of supplemental arginine has also been demonstrated by assessment of the appearance of nitric oxide in expired air following ingestion of a 15 mg dose. Within 15 minutes of administration, nitric oxide concentration in expired air increased 50% from baseline levels (Figure 10). This finding confirms that supplemental arginine is utilized as a precursor in the synthesis of nitric oxide. Figure 10. Effect of Supplemental Arginine on % Change in Nitric Oxide Concentration in Expired Air 29|P a g e Revised03Mar2011 Theramine® Product Information ClinicalResponse Theramine has been clinically tested in crossover studies of patients with fibromyalgia, trigeminal neuralgia, back pain, headache, osteoarthritis, tendonitis, and post herpetic neuropathic pain. Physiologic and symptomatic pain was assessed in a controlled crossover study of 14 patients using visual analogue scales and Likert numeric scales as outcome measures. After 3 days of treatment with Theramine, patients reported a statistically significantly reduction in symptoms of back pain compared with the period prior to treatment (p<0.01) (Figure 11). Based on the results assessed by the Likert scale, symptoms were reduced by more than half in these patients after treatment with Theramine. Figure 11. Effects of Theramine Administration on Back Pain SelectedReferences 1. Borgonio A, Witte K, Stahrenberg R, Lemmer B. Influence of circadian time, ageing, and hypertension on the urinary excretion of nitric oxide metabolites in rats. Mech Ageing Dev 1999;111:23-37. 2. Tangphao O, Chalon S, Coulston AM et al. L-arginine and nitric oxide-related compounds in plasma: comparison of normal and arginine-free diets in a 24-h crossover study. Vasc Med 1999;4:27-32. 3. Brown RE, Stevens DR, Haas HL. The physiology of brain histamine. Prog Neurobiol 2001;63:637-672. 4. Brown DW. Abnormal fluctuations of acetylcholine and serotonin. Med Hypotheses 1993;40:309-310. 5. Wolosker H, Dumin E, Balan L, Foltyn VN. D-amino acids in the brain: D-serine in neurotransmission and neurodegeneration. FEBS J 2008;275:3514-3526. 30|P a g e Revised03Mar2011