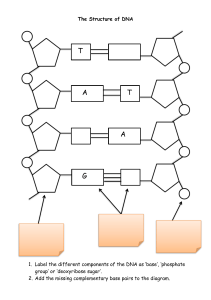
SHS Biotechnology Lab Book v 4.0 Name: ___________________________________ [Type text] Page 1 # 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 Assignment Lab Skills Unit Vocabulary Notes: Metric System /Scientific Notation Lab: Mass & Volume with SI Measurements Notes: Micropipettes Lab: Micropipette Notes: Spectrophotometers Lab: Using the Spectrophotometer Notes: Microscopes Lab: Immersion Oil Microscopy Bacteriology & Microbiology Unit Vocabulary Notes: History of Biotechnology Lab: Making Yogurt Lab: Microbial Flora of the Mouth… …Dental Caries Notes: Media Preparation & Plate Pouring Lab: Media Preparation and Plate Pouring Notes: Bacteria Culture Notes: Aseptic Technique Notes: Gram Staining Lab: Gram Staining Lab: Aseptic Technique and Streaking for Isolation Notes: Colony Morphology Lab: Bacterial Identification Supplemental Notes: Antibiotic Resistance Lab: Antibiotic Resistance Biochemistry Unit Vocabulary Resource: Periodic Table Resource: Chemical Formulas Notes: Chemistry Review Lab: Kool-Aid Chemistry (Mixing Solutions) Notes: Acids and Bases SHS Biotechnology Revised 2015 - 2016 Table of Contents 31 Lab: Creating a pH Scale Pg. 3 4 5 10 12 17 18 26 27 30 34 37 43 45 47 49 51 53 54 57 61 63 71 74 76 77 78 79 80 82 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 57 58 59 60 61 Lab: Chemical Solutions DNA Basics Vocabulary Unit Notes: History of DNA Notes: Nucleic Acids Notes: DNA Extraction Lab: DNA Extraction From Cheek Cells Lab: Strawberry DNA Extraction DNA Profiling Unit Vocabulary Notes: DNA Profiling Making Electrophoresis Gels Lab Lab: Case of the Invisible Bands Lab: Candy Electrophoresis Lab: Paternity Testing Lab: Restriction Digestion of Lambda DNA Lab: DNA Fingerprinting Using Restriction Enzymes DNA Replication & PCR Unit Vocabulary Notes: Replication of DNA Notes: PCR Lab: PCR of Chromosome 16: PV92 Gene Lab: DNA Sequencing Proteins & Genetic Engineering Unit Vocabulary Notes: RNA and Protein Synthesis Notes: Protein Synthesis Notes: DNA Mutation Notes: Size Exclusion Chromatography Lab: Size Exclusion Chromatography Rainbow Transformation Lab Notes: Genetic Engineering Transformation with BFP and GFP Lab Purification & Size Determination of GFP & BFP Lab 83 87 90 92 94 96 98 100 104 106 108 109 111 117 119 127 134 135 137 139 149 152 153 155 157 158 159 163 166 168 176 Page 2 Lab Skills Unit Vocabulary Define the following terms using your textbook. There will be a quiz on these words on ______________________. Assay Bioengineering Bioethics Biohazard Biological Products Bioremediation Biotechnology Centrifugation Food Biotechnology GEM GEO GMO In Vitro In Vivo SHS Biotechnology Revised 2015 - 2016 Page 3 Metric System Basics & Scientific Notation I. II. III. IV. V. Used in nearly every country in the world, the Metric System was devised by French scientists in the late 18th century. The goal of this effort was to produce a system that used the decimal system rather than fractions as well as a single unified system that could be used throughout the entire world. The metric system is based on a base unit that corresponds to a certain kind of measurement. a. Length = meter b. Volume = liter c. Weight (Mass) = gram Prefixes plus base units make up the metric system a. King Henry Drinks Milk During Castle Meetings b. Kilo Hecto Deca Meter Deci Centi Milli Converting with the Metric System a. When converting within the metric system it is simply a measure of moving the decimal in the appropriate direction. i. For each “step” to right, you are multiplying by 10 ii. For example, let’s go from a base unit to centi 1. 1 liter = 10 deciliters = 100 centiliters 2. 2 grams = 20 decigrams = 200 centigrams iii. An easy way to move within the metric system is by moving the decimal point one place for each “step” desired. iv. So for every “step” from the base unit to kilo, we moved the decimal 1 place to the left. b. If you move to the left in the diagram, move the decimal to the left c. If you move to the right in the diagram, move the decimal to the right Scientific Notation a. Step 1: Move decimal left. Leave only one number in front of decimal b. Step 2: Write number without zeros c. Step 3: Count how many places you moved decimal. Make that your power of ten i. 93,000,000 -- Standard Form ii. 9.3 x 107 --- Scientific Notation [Type text] Page 4 Mass & Volume with SI Measurements Lab Pre-Lab Reading Measurements in laboratory work are an essential part of making reliable observations. Measurements that can be made include those of mass, volume, temperature. For laboratory work, three basic types of measurement using the balance should be mastered. These are: measuring mass directly, “measuring out” a specific mass of a substance, and determining mass by difference. Oddly, the metric standard for mass is the gram. The liter is the standard unit of volume in the metric system, but that is generally too large for our purposes. We will measure volume in milliliters (mL). There is an equivalence between mL and cm 3 so that 1 mL = 1 cm3 Generally, our measurements of volume will be made with graduated cylinders. For measuring very specific small volumes, the micropipette will be the tool of choice. In this lab, you will practice how to properly make and record measurements taken with both analog and digital measurement devices with the maximum precision. You will review calculations of volumes of regularly shaped objects from their dimensions. Determining Volume from the Dimensions of an Object The volume (V) of a cuboid (a box-like 6-sided shape with all angles being right angles) is calculated from its length (l), width (w), and height (h) by the equation: V = l w h. Determining Volume by Displacement of a Liquid Another way to determine the volume of a solid is to measure the volume of a liquid that is displaced by the solid. This is particularly useful for irregularly shaped objects. This type of measurement is usually done in a graduated cylinder. The solid object must fit in the graduated cylinder, and be more dense than the liquid. Enough liquid to completely cover the solid is added to the graduated cylinder. The volume of the liquid is measured to the level of precision allowed by the glassware, and then the solid sample is added to the cylinder, causing the liquid level to increase. A second volume measurement is made, which gives the total volume of the liquid and the solid. The volume of the solid is determined by difference: Solid Volume = Total Volume - Liquid Volume Density Density (d) is defined as the ratio of the mass (m) of a sample to its volume (V): d m V Even though the mass and volume of a sample vary with the sample size, the density of a particular substance is the same for any size sample; it is an intensive property. This is true of all “ratio” properties, making them useful for sample identification. Density is useful for a number of reasons. The density of a sample can be used by scientists or technicians to find the identity of the sample. Density is also used to interconvert between mass and volume. If we consider the formula above, we can see that if the density is known, the mass of an object can be determined from its volume by doing some simple algebra. SHS Biotechnology Revised 2015 - 2016 Page 5 Mass & Volume with SI Measurements Lab – Process Lab Purpose: To practice the various techniques of measuring mass and volume using laboratory tools. Materials for Parts A, B & C: Graduated cylinder Beaker with 50 ml H20 3 beakers with water (red, blue, and yellow) 3 irregularly shaped objects Pipette 1 eyedropper calculator 3 stones Ruler Balance 6 test tubes labeled A, B, C, D, E, and F pencil Part A: Count your drops 1. 2. 3. 4. 5. 6. Find the number of drops in 1 milliliter of water. Remember to read the bottom of the meniscus when you are reading the volume of a liquid in a graduated cylinder. Fill a small graduated cylinder with 10 ml of water. Count the number of drops it takes to raise the water to 11 ml. Record the number in the chart. Leave the water in the graduated cylinder and count the number of drops it takes to raise the water to 12ml. Record the number in the chart. Leave the water in the graduated cylinder and count the number of drops it takes to raise the water to 13ml. Record the number in the chart. Calculate your average and round to the nearest tenth. # Drops to 11 mL # Drops to 12 mL # Drops to 13 mL Avg. # Drops in 1 mL Based on your average, how many drops would it take to make 1 liter? _______ Part B: Water Displacement Follow the directions to find the volume of three stones using water displacement. 1. 2. 3. Add 20 ml of water to a 100 ml graduated cylinder. Record this amount in the chart. Add three stones to the cylinder and measure the volume. Record this amount in the chart. Find the difference between the two measurements and record in the chart. The difference between the two measurements will be the volume of the three marbles. Volume of Water Before Adding Stones (ml) SHS Biotechnology Revised 2015 - 2016 Volume of Water After Adding Stones (ml) Difference in Volume (ml) Volume of 3 Stones Page 6 Part C: Mass Mania 1. 2. 3. 4. Zero the balance before placing an object on it. Place your metric ruler on the pan and read & record the ruler's mass. After resetting the balance to Zero, measure and record the mass of the empty 50-ml graduated cylinder and then the 3 stones. Reset the balance to ZERO when all items have been massed. Mass of Metric Ruler (g) Mass of Empty 50-ml graduated cylinder (g) Mass of 3 stones (g) Part D: Color Challenge Perform each step outlined below using accurate measurements. 1. Measure 17 ml of RED water from the beaker and pour into test tube A. 2. Measure 21 ml of YELLOW water from the beaker and pour into test tube C 3. Measure 22 ml of BLUE water from the beaker and pour into test tube E. 4. Measure 5 ml of water from test tube A and pour it into test tube B. 5. Measure 6 ml of water from test tube C and pour it into test tube D. 6. Measure 8 ml of water from test tube E and pour it into test tube F. 7. Measure 5 ml of water from test tube C and pour it into test tube B. 8. Measure 2 ml of water from test tube A and pour it into test tube F. 9. Measure 4 ml of water from test tube E and pour it into test tube D. Test Tube Color Final Volume (ml) (total of what you added to each test tube) A B C D E F SHS Biotechnology Revised 2015 - 2016 Page 7 Part E: Estimation, Density & Measuring by Displacement For solids that have an irregular shape, the displacement method must be used to determine their volume. When using the displacement method, you must first measure the starting volume of the liquid. Then add the object and record the change in volume. This gives the volume of the irregularly-shaped object. 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. Obtain 3 irregularly shaped objects that will sink in water. Estimate the mass & volume of each object. Record this on table 1. Use a triple beam balance or electronic scale to determine the mass in grams of each object. Record this on table 1. Fill a graduated cylinder halfway with tapwater. Measure & record the volume of the water in the graduated cylinder. READ THE MENISCUS! Place one of the objects into the graduated cylinder, and measure and record the new volume. Subtract the initial volume from the final volume. The difference is the volume of the object. Record the difference in data table 1. Repeat steps 4 - 8 for the other three objects. To determine the density of each object, divide the actual mass of each object by its volume (determined by water displacement). Record the densities in data table 1. SHS Biotechnology Revised 2015 - 2016 Page 8 Results & Data Data Table 1 Object Estimated Mass (g) SHS Biotechnology Estimated Volume (ml) Revised 2015 - 2016 Actual Mass (g) Volume of H2O in graduated cylinder (ml) Volume of H2O Object's Volume Density and object in (Subtract Column D=m/V graduated 5 from Column (Divide Column 4 cylinder 6) by Column 7) (ml) (ml) (g/ml) Page 9 Notes: Micropipettes I. II. III. IV. V. VI. Automatic pipettes are used to accurately transfer small liquid volumes. a. Glass pipettes are not highly accurate for volumes less than 1 milliliter (1 ml), but the automatic pipettes are both accurate and precise b. These are continuously adjustable digital pipettes c. Each pipette can be set to transfer any volume within its own volume range You will be able to: a. Select the proper automatic pipette to transfer a specified volume of sample. b. Set a specified volume on the pipette volume indicator using the volume adjustment knob. c. Read a digital volume setting in both micro liter (µl) and milliliter (ml) units. d. Demonstrate the correct technique to accurately transfer a sample of a stock solution to another vessel. Purpose of the Pipettor: a. Biotechnologists use a pipettor to transfer precise amounts of liquids from one container to another. b. The measurements are in microliters 1 ml=1000 µl. Care of the Pipettor - Misuse of the pipettor can result in incorrect measurements affecting your investigation. a. Do not drop the pipettor. b. Do not adjust the volume to less than 2 µl. Parts of the Pipettor (Draw in your notes). Select the Correct Pipettor: Micropipettes have a maximum volume size that they can handle. a. Volume Adjustment i. Step 1 - In the display window are three vertical numbers. 1. The top number is tens µl (10.0). 2. The middle number is ones µl (02.0). 3. The bottom number tenths µl (00.9). ii. Step 2 - Set the pipettor to the desired volume by turning the calibration knob until the volume display reads the desired numbers. 1. Attach the Disposable Tip b. Transferring Liquids i. Step 1 - Hold the pipettor so the display is facing the user and the thumb can easily depress the top pushbutton. ii. Step 2 - Lift the pipettor and attached tip away from the tip box. 1. Depress the pushbutton to the first stop before placing the pipette tip in the liquid. 2. Place the tip in the liquid. iii. Step 3 - Slowly release the pushbutton and draw the liquid into the tip. 1. NEVER LET THE PLUNGER SNAP UP! 2. Remove the tip from the liquid and visually check for liquid in the tip. 3. Remove the tip from the sample. Wipe away any droplets on the outside of the tip with a lint-free tissue. iv. Step 4 - Place the tip into the container which is to receive the liquid. 1. Slowly depress the pushbutton as far as it can go. 2. With the pushbutton fully depressed, withdraw the tip. SHS Biotechnology Revised 2015 - 2016 v. S t e p 5 1. P r e s s d o w n o n t h e t i p e j e c t o r b u t t o n Page 10 VII. VIII. to eject the tip. 2. Dispose of the tip in an appropriate container. Accuracy vs. Precision a. Accuracy means the closeness with which the dispensed volume approximates the volume set on the pipette. i. Accuracy is specified as mean error, the average deviation of replicate measurements from the expected set volume. b. Precision is the "scatter" or reproducibility of individual measurements of the same volume. c. Using the pipettes to transfer volumes which are below the recommended range will introduce larger errors. For optimal reproducibility, use the following pipetting procedures: a. Consistent SPEED and SMOOTHNESS when you press and release the PLUNGER. b. Consistent pressure on the PLUNGER at the FIRSTSTOP. c. Consistent and sufficient IMMERSION DEPTH. d. Nearly VERTICAL POSITIONING of pipette. e. AVOID ALL AIR BUBBLES: Since the plastic pipette shaft can be damaged if liquids are drawn beyond the tip into the shaft. f. NEVER lay the pipette on its SIDE nor INVERT the pipette if liquid is in the tip. SHS Biotechnology Revised 2015 - 2016 Page 11 Micropipette Lab – Pre-Lab Reading The micropipettor is used to transfer small amounts (< 1 mL) of liquids. The scales on micropipettors are in microliters (1000µl = 1 ml). The brand of micropipettors we will be using vary based on the lab station that you are working at. Some are made by Eppendorf, some by Rainin and still others are made by Carolina Biological. Regardless of who made them, they all work the same way, and they all have the same parts. They come in five sizes which are capable of pipetting three ranges of volumes: P10 = 0.5- 10 µl, P20 = 2 - 20 µl, P100 = 10-100 µl, P200 = 20-200 µl, and P1000 = 100-1000 µl. They are used in conjunction with disposable (often sterile) plastic tips; the smaller micropipettors (P10) require the smallest tips, the P20, P100, and P200 require the middle sized tips, and the P1000 uses the larger blue tips. The following is an illustration of a micropipettor: Directions for use of the micropipettor: 1. Never exceed the upper or lower limits of the pipettor. They are very expensive and delicate instruments which we cannot afford to damage. The limits are: a. P10: 0.5 to 10.0 µl b. P20: 2 to 20.0 µl c. P100: 10 to 100 µl d. P200: 20 to 200 µl e. P1000: 100 to 1000 µl 2. Set the desired volume by turning the centrally located rings clockwise to increase volume or counterclockwise to decrease volume. Some examples are provided below: 3. Place a tip on the discharge end of the micropipettor. NOTE: If sterile conditions are necessary do not allow the pipet tip to touch any object (including your hands). 4. The plunger will stop at two different positions when it is depressed. The first of these stopping points is the point of initial resistance and is the level of depression that will result in the desired volume of solution being transferred. Because this first stopping point is dependent on the volume that is being transferred, the distance you have to push the plunger to reach the point of initial resistance will change depending on the volume being pipetted. The second stopping point can be found when the plunger is depressed beyond the initial resistance until it is in contact with the body of the micropipettor. At this point the plunger cannot be further depressed. This second stopping point is used for the complete [Type text] Page 12 5. 6. 7. 8. 9. discharging of solutions from the plastic tip. You should not reach this second stop when drawing liquid into the micropipettor, only when expelling the last drop. Depress the plunger until you feel the initial resistance and insert tip into the solution, just barely below the surface of the liquid and not as deep as possible. Carefully and slowly release plunger. NOTE: If the solution you are pipetting is viscous, allow the pipet tip to fill to final volume before removing it from solution to avoid the presence of bubbles in the plastic tip which will result in an inaccurate volume. If you release the plunger too quickly, it will suck liquid up into the micropipettor body and damage it. Discharge the solution into the appropriate container by depressing plunger. This time, depress the plunger to the point of initial resistance, wait one second, and then continue pressing the plunger as far as it will go in order to discharge the entire volume of solution. Remove tip by pressing down on the tip discarder. REMEMBER TO CHANGE TIPS BETWEEN SOLUTIONS TO AVOID MIXING OR CONTAMINATING THE SOLUTIONS USED!! SHS Biotechnology Revised 2015 - 2016 Page 13 Process Lab: Micropipette Lab Purpose This laboratory activity introduces micropipetting technique. As with all fine motor skills, learning how to use a micropipet takes practice and determination. You will be rewarded with excellent DNA laboratory results in this and future labs. Directions for Using Micropipettes 1. Set pipette volume only within the range specified for that micropipette. Do not attempt to set a volume beyond the pipette’s minimum or maximum values. 2. When using a micropipette, first apply a tip. Forgetting to do this would cause liquid to be sucked into the nose cone. Since a micropipette works by air displacement, its internal mechanism must remain dry. 3. Always keep a micropipette in a vertical position when there is fluid in the tip. Do not allow liquid to accidentally run back into the nose cone. 4. Use your thumb to control the speed at which the plunger rises after taking up or ejecting fluid. Releasing the plunger too abruptly will cause liquid to pop up 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. into the nose cone and damage the piston. Hold the micropipette in one hand, almost vertical; hold the tube in your other hand. Both should be almost at eye-level. NOTE: the person holding the micropipette is also the person who should be holding the tube! Before lowering the tip into the liquid, push down the plunger to the first stop and hold in this position.(DO NOT GO PAST THE FIRST STOP OR YOU WILL GET AN INCORRECT VOLUME), then... ... dip the tip into the solution to be pipetted. Draw fluid into the tip by slowly releasing the plunger. (Cap and put down the tube with the liquid or hand it to your partner.) Hold the micropipette in one hand, almost vertical; hold the tube in your other hand. Both should be about at eye-level. Touch the micropipette tip to the inside wall of the reaction tube into which you want to expel the sample. This creates a tiny surface-tension effect which helps coax fluid out of the tip. Slowly push down the plunger of the micropipette to the first stop. Then, continue to the second stop to expel the last bit of fluid, and hold the plunger in this position. Always set the micropipet within its designated range. Always use a micropipet with a tip. Always hold a loaded micropipette in a vertical position. Always release the micropipette plunger slowly Centrifuge Instructions 1. The microfuge rotor must always be balanced. Spinning unbalanced would damage the motor of the instrument. 2. The amount of liquid in the tubes should be similar, otherwise the rotor will spin unevenly. 3. You can always prepare a "blank" tube with the appropriate volume of liquid with which to balance a single tube. 4. Samples of balanced rotor configurations SHS Biotechnology Revised 2015 - 2016 Page 14 Materials per team Solution I Solution II Solution III Solution IV Solution V Micropipette tips Supply of 1.5-mL reaction tubes Rack for reaction tubes Container for waste tips Container for liquid waste Fine-tip permanent marking pen Microcentrifuge Lab Procedure Part A: Practicing with a P-10. 1. Label two empty reaction tubes A and B with a permanent ink marker, if not already labeled. 2. Add the amounts of solutions I, II, III and V to tubes A and B as shown in the table below. 3. Spin tubes A and B in the microcentrifuge for 1-2 sec to pool the solutions. 4. Add up the total volume of liquid in Tube A. 5. AS A CHECK OF YOUR TECHNIQUE, set the micropipette to that volume and withdraw all of the liquid in tube A. The contents should just fill the tip -- no air space at the bottom of the tip; no leftover liquid in the tube. 6. Discard liquid and tip into waste container. 7. Check your technique and accuracy for tube B in the same way as you did for tube A. Solution Volume in Each Tube Reaction Tube Solution I Solution II Solution III A 4 µL 5 µL 2 µL B 6.5 µL 2.5 µL Solution IV Solution V Total in Tube 2 µL Lab Procedure Part B: Practicing with a P-100. 1. Label an empty reaction tube C, if not already labeled. 2. Add the amounts of solutions I, II and IV to tube C as shown in the table below. 3. Spin tube C for 1-2 sec. (Be sure to balance your centrifuge.) 4. Check the accuracy of your micropipeting technique with the P-100. Reaction Tube Solution I Solution II C 20 µL 40 µL Solution III Solution IV Solution V Total in Tube 40 µL Lab Procedure Part C: Practicing with a P-1000. 1. Label an empty reaction tube D if not already labeled. 2. Add the amounts of solutions III and V to tube D as shown in the table below. 3. Spin tube D for 1-2 sec. (Be sure to balance your centrifuge.) SHS Biotechnology Revised 2015 - 2016 Page 15 4. Check the accuracy of your technique with the P-1000. Reaction Tube Solution I Solution II D Solution III Solution IV 200 µL Solution V Total in Tube 550 µL Lab Procedure Part D: Practicing with all micropipettes 1. Label an empty reaction tube E, if not already labeled. 2. Fill in the blanks in the following chart. 3. Indicate on the line "P-_____" the appropriate micropipette to be used for each sample. 4. Then write in the 4 boxes below each line, the numbers that should be dialed to give you the indicated volume. 5. Add the volumes of solutions I-V indicated above into tube E, using the appropriate micropipet. 6. Check the accuracy of your technique. Reaction Tube E Solution I Solution II Solution III Solution IV Solution V Volume: 15 µL 120 µL 12 µL 38 µL 350 µL Micropipette: (P-10,P-100,P-1000) P-_____ P-_____ P-_____ P-_____ P-_____ 1 5 Micropipette Reading 0 0 SHS Biotechnology Revised 2015 - 2016 Page 16 Spectrophotometer Notes I. II. III. IV. Cuvette: Where you place samples of substances you are testing in a spectrophotometer. Purpose: Used to measure the effect of a sample of a substance on a beam of light. Uses: a. To determine: i. How much DNA is present in a cellular extract. ii. The purity of protein in enzyme preparation. iii. The activity of an enzyme. iv. To confirm the identity of an ingredient in a drug formulation. v. To solve crimes. Types of Electromagnetic Radiation a. Longer wavelengths = less energy. b. Shorter wavelengths = more energy. c. V. Transmission vs. Absorption i. Transmission = Light passes through ii. Absorption = the light is absorbed by the substance. Lambert-Beer’s Law - is an empirical relationship that relates the absorption of light to the properties of the material through which the light is travelling. a. Identifies the relationship between sample concentrations and the intensity of transmitted light. b. Transmittances and Absorbances are defined by the following equations: i. I0 = Intensity of incident light ii. It = Intensity of transmitted light iii. C = Sample’s concentration iv. 1 = Optical path length. c. Absorbance (A) is Essentially: A=abc -orabsorbance = (extinction coeff) X (cell thickness) X (concentration). d. In lay-men’s terms – the amount of light in a particular wavelength that is absorbed by the substance. SHS Biotechnology Revised 2015 - 2016 Page 17 Using the Spectrophotometer Lab – Pre-Lab Reading A spectrophotometer is a valuable instrument in biology. It allows us to measure and compare the concentration of materials in solution with relative ease. It is therefore important for you to understand how the spectrophotometer works so that you understand the data that you collect from it. WHAT DOES A SPECTROPHOTOMETER DO? A spectrophotometer is an instrument that measures either the transmittance of light through a solution or the absorbance of light by a solution. Light can either pass through an object or be stopped by an object which is in its path. We can easily see this if we try looking at a light through a piece of clear plastic — all of it Is transmitted through and none of it is absorbed. Then try to see the same light through a piece of cardboard — all of it is absorbed by the cardboard and none of it is transmitted through. In this way, you can now see that transmittance refers to the light which passes through a solution. Absorbance refers to the light which cannot pass through a solution, but instead of passing through the solution, the light is absorbed by the solution. For example, if we pass a beam of white light through a blue glass, not all the light passes through the glass to the other side — some of it is absorbed by the glass. The light which does pass through the glass is said to be transmitted and its measure is called transmittance. So, if your beam of white light is 40% absorbed by the glass, it can also be said that the light is 60% transmitted. It is now clear that the darker or denser a substance is the more light it will absorb and theses light it will transmit. Here is a diagram of the main parts of the instrument: WHAT ARE THE SCALES ON A SPECTROPHOTOMETER? Transmittance is an arithmetic scale that is divided into one hundred equal parts, so transmittance can be expressed as a percent. A value of 100% transmittance means that all the light has passed through the material in the cuvette and none was absorbed. A value of 0% transmittance means that no light passed through the sample in the cuvette. Absorbance is a logarithmic scale with unequal divisions. A value of 0.0 absorbance means that no light has been absorbed by the material and all the light has been transmitted. A reading all the way at the other end of the scale means that all the light has been absorbed by the material. WHAT IS THE “BLANK” AND WHY DO WE HAVE TO USE IT? All substances, clear or not, absorb some light. Therefore, if we wish to measure the absorbance (or the transmittance) of a material like protein in solution, we must not only consider the light absorbed by the protein, but also the light absorbed by the cuvette and the light absorbed by the solution in which the protein is dissolved. In order to measure only the absorbance of the protein in the solution, we must “subtract” out the absorbance due to the cuvette and the solvent in which the protein is dissolved. If our protein is dissolved in water, and we are trying to assess how much protein is in solution, we would ultimately want to measure how much light is absorbed by the protein alone and not the how much light is absorbed by either the water or the cuvette. In order to subtract out these factors, we [Type text] Page 18 prepare another cuvette with only water in it. This is called the “blank.” In general, the blank is a cuvette which contains everything that is in the sample (or experimental) cuvette, except the one material whose absorbance we are measuring. To use the blank, you could measure your experimental cuvette, then measure your blank cuvette and find the difference between them. However the spectrophotometer has a built in calibration system that allows you to avoid this calculation and its much like zeroing an electronic scale. Follow this procedure: 1. Before you measure a solution in an experimental cuvette, first insert the blank cuvette into the machine. The spectrophotometer will register an absorbance value for the blank. 2. Now press the “Goto WL” button and type in the wavelength that you want to use. Then press enter. 3. Next hit the “Auto Zero” button. This resets the machine to a 0% absorbance, or a 100% transmittance. 4. Take the blank out and now you are ready to read the absorbance or transmittance of the material in solution without having your data affected by the absorbance of the cuvette or the solvent. 5. DO NOT hit any additional buttons! Just put the cuvette in the holder and close the chamber door and the reading will appear on the screen. 6. Also be aware that fingerprints on the cuvettes can affect your readings, so all cuvettes need to be wiped clean with a Kimwipe before inserting them into the spectrophotometer. WHAT WAVELENGTH OF LIGHT DO I USE? Most of the material we see around due is colored due to light reflection. Why do we see a blue sky? White light hits the layers of the atmosphere and all the colors of the spectrum are absorbed, except the color blue, which is reflected back to our eyes. Why is your shirt red? Because white light hits the shirt fabric and all the colors of the spectrum are absorbed, except the color red, which is reflected back to our eyes. Everything around us absorbs certain colors (wavelengths of light) better than other colors. Since the spectrophotometer measures absorbance of light it would work the best in an experiment when we use the color light that is best absorbed by the material we are examining. In other words, the spectrophotometer shines specific wavelengths of light through the cuvette. We will get more accurate readings in our experiment, if we set the spectrophotometer to shine through the cuvette the wavelength of light that the material in the cuvette absorbs the most. How do we know which color (wavelength) of light is absorbed best by the material we are testing for in the cuvettes? To answer that question, we have to complete a test called an “Absorption Spectrum” on the material. How do we run an “Absorption Spectrum”? Just measure the absorbance of the material over a range of wavelengths. Then plot a graph of absorbance vs. wavelength of light and we are likely to get a curve which looks like this: What does this tell us? The graph shows us that the material we are studying absorbs very little light between 400- 500nm or between 700-800nm, so it would be difficult to get accurate measurements of how much material is in solution in our cuvette if the spectrophotometer was set in those wavelength ranges. But the absorbance of the material goes up very rapidly between 500-600nm and reaches a peak at about 600nm. Therefore, when we want to measure how much of this material we have in solution, it would be best if the spectrophotometer were set to this wavelength so it only shined light of wavelength 600nm through the cuvette. Luckily, absorbance spectra have been done with most material we will test for in biology, so we don’t have to develop one each time we use the spectrophotometer SHS Biotechnology Revised 2015 - 2016 Page 19 Grading Rubric – For Teacher’s Use Using the Spectrophotometer (Full-Blown Lab) I will deduct points for each of the following: Name: ___________________________ Lab Conclusion Due Date: ____/____/____ This Lab is to be written in complete sentences with correct grammar, spelling and punctuation. It is to be your own work, just because you work in groups does not mean that your answers should be even remotely close to the same wording. Question: How can different substances or concentrations of the same substance be identified using the equipment in a laboratory? Hypothesis (written in “If… then…because” format): ___________________________________________________________________ ____________________________________________________________________________ _____________________________________________________________________________ Independent Variable: _______________________________________________ Dependent Variable: ________________________________________________ After completing the lab, tell what affect the INDEPENDENT VARIABLE had on the DEPENDENT VARIABLE (describe the relationship between the two variables in a minimum of 3 sentences)? ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ Materials: Beaker of (distilled) H2O P-1000 Micropipettor and Tips Blue Dye Tube of Blue Dye Spectrophotometer Cuvettes Unknown Solution of Overall: Not written in complete sentences. Does not use correct grammar spelling & punctuation. /5 Hypothesis: Does not use proper format – If/Then/Because. Hypothesis does not completely answer lab question. Hypothesis is poorly stated. Does not clarify what the student is thinking. /10 Independent Variable: No variable identified. Incorrect Independent variable. /5 Dependent Variable: No variable identified. Incorrect Dependent variable. /5 Link between variables: Answer not 3 sentences. Question is not answered. Link between variables is not explained clearly. The link described between variables is incorrect. Description of what was done in lab is not an answer to this question. /10 Part A. Determination of the Amax of Blue Dye. [Type text] Page 20 1. Watch the demonstration on how to use the Spec in class and follow the instructions below. Pay attention to the placement of the following: a) Sample compartment door; b) Keyboard/ display unit; c) Cuvette holder; d) Detector; e) Test tube access door. 2. The spec should be on before lab begins (it takes 20 minutes to warm up). 3. Make sure there are no samples in the cuvette holder and that the sample compartment and test tube access doors are closed. 4. Press the number “1” on the key pad to select “Photometric”. 5. Press the “GOTOWL” key, then type in the desired wavelength (ie, 530) then press “Enter”. The Spectrophotometer will stall temporarily as the machine adjusts its wavelength. 6. Wipe off your cuvette containing the blank solution (a cuvette that contains 2 ml distilled water) with a KimWipe and place it in the single cuvette holder in the sample compartment. Make sure that the cuvette is aligned with the light source. Be sure to have the clear faces of the cuvette facing the light source. 7. Close the sample compartment door. 8. Press the AUTO ZERO button on the keypad. The display shows “ZEROING...” and then the reading will clear to 0.00. 9. Remove the blank. Wipe off the cuvette containing the sample (2 ml of blue dye) with a Kim Wipe, insert it into the sample compartment and close the sample compartment door, just as you did with the blank. 10. Press the F1 button to switch to %T (transmission), and record the transmission in the data table. 11. Subtract the transmission percent from 100 to get the amount of light absorbed by the sample. 12. To determine the Amax of the compound, each team will be assigned a range of 100 nm within the visible range of the spectrum (400nm to 700 nm). Read the absorbance of the sample every 10 nm within your team’s assigned range. You will then write your data on the lab white board. Next, you will compile the data from the board and graph the data for the whole class to determine the Amax for the dye. 13. Follow the procedures used above to complete your task. Remember to reinsert the blank and “zero” the reading each time you change the wavelength OR return to the machine after someone else uses it. 14. Rinse out your two cuvettes with distilled H2O and place upside down on a Kim Wipe to drain. 15. Record your numbers in the results section in your lab notebook and share your data with the class. 16. Graph the class data in this section as well. This is the wavelength you will use in Part B. SHS Biotechnology Revised 2015 - 2016 Page 21 Part B: The Effect of Concentration on Absorbance 1. Obtain 4 test tubes. Number them 1 to 4. 2. Use a P1000 to pipet 2000 µl of distilled H2O into each tube. 3. Add 2000 µl of blue dye to tube 1. This tube represents a 1/2 (or a 1:1) dilution. 4. To mix the contents of tube 1, use a vortex mixer (see demonstration in class). 5. Set the vortex speed on 4. Make sure the vortex switch is on TOUCH. 6. Place your tube in the black rubber cup of the vortex and press down to mix your sample. 7. Pipet 2000 µl of the Tube 1 contents into Tube 2. Mix using the vortexer. Tube 2 is a 1/4 (or 1:3) dilution. (You made a 1/2 dilution of a 1/2 dilution. 1/2 * 1/2 = 1/4). 8. Transfer 2000 µl of Tube 2 into Tube 3. What dilution is this? You made a 1/2 dilution of a 1:4 dilution. 9. Transfer 2000 µl of Tube 3 into Tube 4 and mix with the vortex mixer. 10. Set the spec. to the Amax wavelength that you determined in Part A. 11. Zero the spec using your blank cuvette. 12. Read the absorbance of tube 4. Record it in your results section. 13. Pour the contents of tube 4 down the sink and read the absorbance of tubes 3, 2, and 1. Record them in the results section. 14. Re-measure the absorbance of your undiluted dye. 15. Make a graph of absorbance vs. concentration of blue dye. 16. When you are finished, pour the dye dilutions into the sink, rinse out the cuvettes and test tubes with tap water followed by distilled water and place them upside down to dry. Table 1 Completed: /5 ______ Results: Make and complete the following items in your lab notebook exercise: A. TABLE 1: (Record the absorbance and percent transmission of the blue dye at 530 nm). Wavelength A of Blue Dye %T of Blue Dye 530 nm B. TABLE 2: (Record the absorbance values for your assigned wavelengths for blue dye). SHS Biotechnology Revised 2015 - 2016 Page 22 Table 2 Completed: Wavelength (nm) 400 nm 450 nm 500 nm 550 nm 600 nm 650 nm 700 nm 750 nm 800 nm 850 nm 900 nm 950 nm Absorbance _____/5 Table 3 Completed: _____/5 Hypothesis Correct? C. TABLE 3: (Record the absorbance of each of solutions in the serial dilution). Blue Dye ½ Dilution ¼ Dilution 1/8 Dilution 1/16 Dilution (100%) (50%) (25%) (12.5%) (6.25%) A of Blue Dye Dilution Based on the data collected, was your hypothesis correct? Give 2 pieces of data that clarify your decision: ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ CHOOSE 1 based on whether your hypothesis is correct: If your hypothesis was wrong, what was wrong with it (detail – minimum 2 sentences)? OR If your hypothesis was right, generate a new lab question that could be asked to create a new lab experiment to follow up on what you learned in this experiment. ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ Uses the words “Yes” or “No” to start. First sentence does not state clearly, “My hypothesis was correct/not correct.” 2 items of detailed data measured in lab (numbers, observations) were not provided to support decision. Data used is not explained. Student does not make a choice. A hypothesis cannot be both right and wrong. The decision about hypothesis correctness does not match the data collected in lab. _____/10 Hypothesis related choice 1: Wrong question answered. Does not tell what revisions to hypothesis would make it correct. Evaluates lab procedures instead of the hypothesis. Does not tell why the hypothesis was wrong. Not 2 sentences long. _____/10 Hypothesis related choice 2: Wrong question answered. Basically a repeat of the question from this lab. Not in the form of a question. Little thought or creativity shown. Not testable in lab. Question is too general. Neither question is answered. _____/10 SHS Biotechnology Revised 2015 - 2016 Page 23 CHOOSE 1: How could the experiment be repeated to make it more accurate (describe 2 ways to improve the experiment)? OR Were there any mistakes made in the process of following the procedures that affected the outcome? If so, how could these mistakes be corrected for (be detailed about the mistakes you made and how you would correct for them)? ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ How can the data we collected and the concepts we learned in this lab be applied to life? In other words, what is the practical application of the concepts from this lab (minimum 3 sentences)? ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ Choice 1: _____/10 Choice 2: SHS Biotechnology Revised 2015 - 2016 Doesn’t tell what you did wrong & how to fix it next time. Should be longer than 1 sentence. Saying, “We didn’t make any mistakes,” means you have to answer the other question. Saying, “Follow all the steps better,” does not work for an answer. This student does not understand what was happening in the lab. _____/10 Application: If a lab manager finds a series of bottles of the same chemical sitting on a shelf in a storage room, but those bottles are unmarked for their concentration, describe the process that the chemist might go through to determine the percent concentration of the chemicals in the bottles (minimum of 10 sentences in a complete paragraph). ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ Not enough detail. 2 ways not given. Answers were complaints not solutions. Saying, “Follow all instructions” is not a solution. 1 sentence is not enough. Don’t blame others, find solutions. This student does not understand what was happening in lab. Answers should be specific with at least one example/career/life experience listed. Not 3 sentences long. Student does not understand how to apply this to life or what was done in lab. Don’t give opinions (i.e this is dumb). Don’t retell what we did in lab. _____/10 Specific Question #1: _____/15 Page 24 Specific Question #2: _____/15 Total Score: ________/120 If you were asked to design an experiment where you had test various concentrations of the chemical Methylene Blue in a spectrophotometer, what wavelengths would be the best wavelength to use in the test, and the second best wavelength to use in the test. Explain why you chose these wavelengths and a least four sentences on how you would conduct the test. __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ __________________________________________________________________________________ SHS Biotechnology Revised 2015 - 2016 Page 25 Microscope Notes SHS Biotechnology Revised 2015 - 2016 Page 26 Oil Immersion Microscopy Lab – Pre-Lab Reading Types of Microscope: Light Microscope - the models found in most schools, use compound lenses to magnify objects. The lenses bend or refract light to make the object beneath them appear closer. Common magnifications: 40x, 100x, 400x Stereoscope - this microscope allows for binocular (two eyes) viewing of larger specimens. Scanning Electron Microscope - allow scientists to view a universe too small to be seen with a light microscope. SEMs do not use light waves; they use electrons (negatively charged electrical particles) to magnify objects up to two million times. Transmission Electron Microscope - also uses electrons, but instead of scanning the surface (as with SEM's) electrons are passed through very thin specimens. Magnification Your microscope has 4 magnifications: Scanning (Low), Medium, High and Oil Immersion. Each objective will have written the magnification. In addition to this, the ocular lens (eyepiece) has a magnification. The total magnification is the ocular x objective Magnification Ocular lens Total Magnification Scanning 4x 10x 40x Med Power 10x 10x 100x High Power 40x 10x 400x Oil Immersion 100X 10X 1000X Focusing Specimens 1. Always start with the scanning objective. Odds are, you will be able to see something on this setting. Use the Coarse Knob to focus, image may be small at this magnification, but you won't be able to find it on the higher powers without this first step. Do not use stage clips, try moving the slide around until you find something. 2. Once you've focused on Scanning, switch to Medium Power. Use the Coarse Knob to refocus. Again, if you haven't focused on this level, you will not be able to move to the next level. 3. Now switch to High Power. (If you have a thick slide, or a slide without a cover, do NOT use the high power objective). At this point, ONLY use the Fine Adjustment Knob to focus specimens. 4. If the specimen is too light or too dark, try adjusting the diaphragm or the light source to reduce the amount of light coming through the aperture. 5. If you see a line in your viewing field, try twisting the eyepiece, the line should move. That's because it’s a pointer, and is useful for pointing out things to your lab partner or teacher. SHS Biotechnology Revised 2015 - 2016 Page 27 Drawing Specimens 1. Use pencil - you can erase and shade areas. 2. All drawings should include clear and proper labels (and be large enough to view details). Drawings should be labeled with the specimen name and magnification. 3. Labels should be written on the outside of the circle. The circle indicates the viewing field as seen through the eyepiece, specimens should be drawn to scale - ie..if your specimen takes up the whole viewing field, make sure your drawing reflects that. Making a Wet Mount 1. Gather a thin slice/piece of whatever your specimen is. If your specimen is too thick, then the coverslip will wobble on top of the sample like a see-saw, and you will not be able to view it under High Power. 2. Place ONE drop of water directly over the specimen. If you put too much water, then the coverslip will float on top of the water, making it hard to draw the specimen, because they might actually float away. (Plus too much water is messy). 3. Place the coverslip at a 45 degree angle (approximately) with one edge touching the water drop and then gently let go. Performed correctly the coverslip will perfectly fall over the specimen. How to Stain a Slide 1. Place one drop of stain (iodine, methylene blue..there are many kinds) on the edge of the coverslip. 2. Place the flat edge of a piece of paper towel on the opposite side of the coverlip. The paper towel will draw the water out from under the coverslip, and the cohesion of water will draw the stain under the slide. 3. As soon as the stain has covered the area containing the specimen, you are finished. The stain does not need to be under the entire coverslip. If the stain does not cover as needed, get a new piece of paper towel and add more stain until it does. 4. Be sure to wipe off the excess stain with a paper towel. How to Ues the Oil Immersion Lense: 1. Once the object has been discovered on high power, turn the nosepiece so that the High and Oil objectives ore on either side of the specimen on the slide. 2. Place one drop of immersion oil onto the specimen and turn the oil objective so it touches the drop. Refocus the specimen. 3. NEVER allow the oil to touch any other objective other than the oil immersion objective. Troubleshooting Occasionally you may have trouble with working your microscope. Here are some common problems and solutions. 1. Image is too dark! - Adjust the diaphragm, make sure your light is on. 2. There's a spot in my viewing field, even when I move the slide the spot stays in the same place! - Your lens is dirty. Use lens paper, and only lens paper to carefully clean the objective and ocular lens. The ocular lens can be removed to clean the inside. 3. I can't see anything under high power! - Remember the steps, if you can't focus under scanning and then low power, you won't be able to focus anything under high power. 4. Only half of my viewing field is lit, it looks like there's a half-moon in there! - You probably don't have your objective fully clicked into place. SHS Biotechnology Revised 2015 - 2016 Page 28 Process Lab: Immersion Oil Microscopy Materials: Prepared Slides Cover Slips Microscope Newspaper Immersion Oil Hair Blank Slides Protocol Part I – Using Oil Immersion: 1. Obtain a prepared slide from the front counter. 2. Focus at low power on a region of a smeared and stained specimen which is well-spread and stained. 3. Rotate nosepiece to the medium power objective, locate desired portion of specimen in the center of the field. Refocus very carefully so that the specimen is focused as sharply as possible. 4. Rotate nosepiece to the high power objective, locate desired portion of specimen in the center of the field. 5. Partially rotate turret so that 40x and 100x objectives straddle the specimen. 6. Place a small drop of oil on the slide in the center of the lighted area. (Take care not to dribble on the stage.) 7. Rotate turret so that the 100x oil immersion objective touches the oil and clicks into place. The oil immersion lense should be touching the oil on the slide. 8. Focus only with fine focus. If you still have trouble, move the slide slightly left and right, looking for movement in the visual field, and focus on the object which moved. 9. Never go backward to the low objectives after you have applied oil to the specimen since oil can ruin the lower power objectives. 10. Draw the specimen in the results section of your notebook. 11. Repeat the above procedures for 2 other prepared slides. Protocol Part II – Making and Observing a Slide: 1. Obtain a blank slide. Make sure that it is cleaned of fingerprints. 2. Cut a word containing the letter “e” out of the print portion of a newspaper article. Place the work in the center of the blank slide, and cover with a coverslip. 3. Examine the “e” on low power and answer the questions in table #2 in your conclusions section. 4. Turn the nosepiece to the medium power. Find and focus the image, then move on to high power. 5. Focus the image, then move on to the oil-immersion by following the directions from part I. 6. Draw the “e” in the results section of your lab write up (on Oil Immersion). 7. Next, obtain another clean blank slide. [Type text] Page 29 8. Have one member of your group pull a hair from their head, eyebrow or eyelashes. Place the hair across the slide and trim it so that it does not hang off of the edges of the slide. 9. Start on low power, and proceed through the various objectives until you reach the oil immersion objective. Draw the hair on this power, in the results section of your lab write up. Results: Prepared Slide #1 Prepared Slide #2 Prepared Slide #3 Name of Specimen: (Found on Slide Name of Specimen Label) Name of Specimen Power of Magnification: (Eyepiece X Objective) Power of Magnification: SHS Biotechnology Revised 2015 - 2016 Power of Magnification: Page 30 Table #2 Part II Questions about the slide: Answer: 1. When you move the slide to the right on the stage, what direction does it appear to move in the eyepiece? 2. When the slide is moved away from you on the stage, which way does it appear to move in the eyepiece? 3. In which power (High, Med., Low) do you see more of the fieldof-View? “e” Slide Drawing 4. Why is it important to find the object you are looking for on low power before switching the microscope to a higher power? Power of Magnification: SHS Biotechnology Revised 2015 - 2016 Page 31 Bacteriology & Microbiology Unit Vocabulary Define the following terms using your textbook. There will be a quiz on these words on ______________________. Aerobe Anaerobe Antibiotic Antibiotic resistance Asexual Reproduction Bacillus Bacteriocide Colony Conjugation Death Phase E. coli Eukaryote [Type text] Page 32 Fermentation Growth Phase Insulin Lag Phase Lawn Logarithmic Phase Metabolism Microbiology Nucleus Plaque Plasmid Prokaryotes SHS Biotechnology Revised 2015 - 2016 Page 33 History of Biotechnology I. II. III. IV. V. VI. VII. Ancient Biotech - Begins with early civilization. a. Developments in ag and food production. b. Few records exist. c. Archeologists research. i. Ancient carvings and sketches sources of information. Classical Biotech - Follows ancient. a. Makes wide spread use of methods from ancient, especially fermentation. b. Methods adapted to industrial production. c. Produce large quantities of food products and other materials in short amount of time. i. Meet demands of increasing population d. Many methods developed through classical biotech are widely used today. Modern Biotech - Manipulation of genetic material within organisms. a. Based on genetics and the use of microscopy, biochemical methods, related sciences and technologies. b. Often known as genetic engineering. c. Not known when biotech began exactly d. Focused on having food and other human needs Ancient / Classical Biotech a. Useful plants brought from the wild, planted near caves where people lived. As food was available, ability to store and preserve emerged. Food preservation most likely came from unplanned events such as a fire or freeze. Domestication a. 15,000 years ago, large animals were hard to capture. People only had meat when they found a dead animal. Came up with ways of capturing fish and small animals. Food supplies often seasonal. Winter food supplies may get quite low. b. Domestication is seen by scientists as the beginning of biotech. c. Domestication = Adaptation of organisms so they can be cultured. d. Most likely began 11,000 – 12,000 years ago in the middle east i. Involved the collecting of seed from useful plants and growing crude crops from that seed. Involved the knowledge that the seed had to properly mature. e. Proper planting: i. Need for water, light etc. for plant growth. ii. Earliest plants likely grains and other food seeds. f. Raising animals in captivity began about the same time. Easier to have an animal close by that to hunt and capture one. i. Learned that animals need food and water. ii. Learned about simple breeding / How to raise young. iii. Cattle, goats and sheep were the first domesticated food animals. g. About 10,000 years ago, people had learned enough about plants and animals to grow their own food. The beginning of farming. a. Domestication resulted in food supplies being greater in certain times of the year. Products were gathered and stored. b. Some foods rotted and others changed form and continued to be good to eat. Foods stored in a cool cave did not spoil as quickly. Foods heated by fire also did not spoil as quickly. Immersing in sour liquids prevented food decay. Food preservation. a. Using processes that prevent or slow spoilage. Heating, cooling, keeps microorganisms (mo’s) from growing. Stored in bags of leather or jars of clay. Fermentation occurs if certain mo’s are present. Creates an acid condition that slows or prevents spoilage. b. Cheese - One of the first food products made through biotechnology. i. Began some 4,000 years ago. [Type text] Page 34 ii. iii. VIII. IX. X. XI. XII. XIII. Nomadic tribes in Asia - Strains of bacteria were added to milk. Caused acid to form, resulting in sour milk. Enzyme called “rennet” was added. Rennet comes from the lining of the stomachs of calves. Rennet is genetically engineered today. c. Yeast - Long used in food preparation and preservation d. Bread baking - Yeast produces a gas in the dough causing the dough to rise. e. Vinegar - Require the use of yeast in at least one stage of production. Yeast - Species of fungi. Some are useful. Some may cause diseases. a. Vinegar - Ancient product used to preserve food. Juices and extracts from fruits and grains can be fermented. b. Fermentation - Process in which yeast enzymes chemically change compounds into alcohol. In making vinegar the first product of fermentation is alcohol. Alcohol is converted to acetic acid by additional microbe activity. Acid gives vinegar a sour taste. Vinegar prevents growth of some bacteria. Keeps foods from spoiling, used in pickling. c. Biblical references to wine indicate the use of fermentation some 3,000 years ago. Fermentation control. a. Advancements occurred in the 1800’s and early 1900’s. b. Used to advance fermentation process: i. Specially designed chamber that promotes fermentation. Allowed better control, especially with vinegar. New products such as glycerol, acetone, and citric acid resulted. c. Development of yeasts that were predictable and readily available led to modern baking industry. Antibiotics - Use of fermentation hastened the development of antibiotics. A drug used to combat bacterial infections. a. Penicillin - Developed in the late1920’s, Introduced in the 1940’s. First drug produced by microbes. Many kinds available today. Limitations in their use keep disease producing organisms from developing immunity to antibiotics. Use antibiotics only when needed. Overuse may make the antibiotic ineffective when really needed later. Some disease organisms are now resistant to certain antibiotics. Modern Biotech - Deals with manipulating genetic info. a. Microscopy and advanced computer technology are used. In-depth knowledge of science. Based on genetics research from the mid 1800’s. b. Genetics - Study of heredity. Most work has focused on animal and plant genetics. c. Genes – determiners of heredity. Carry the genetic code d. Understanding genetic structure essential for genetic engineering. e. Heredity - How traits are passed from parents to offspring. Members of the same species pass the characteristics of that species. Differences exist within each species. Differences are known as variability. Use of biotech to produce new life forms. a. Emerged in mid 1900’s. Made possible by rDNA technology. rDNA = Recombinant DNA Process. Genetic material is moved from one organism to another. Materials involved are quite small. b. Challenging and often controversial. Many have opposing or negative views of biotechnology. People in Biotech: a. Zacharias Janssen - Discovered the principle of the compound microscope in 1590. Dutch eye glass maker, Anton Van Leeuwenhoek developed single lens microscope in 1670’s. First to observe tiny organisms and document observations. Anton V.L. work led to modern microscopes. Electron microscope developed in 1931 by group of German scientists. b. Gregor Mendel - Formulated basic laws of heredity during mid 1800’s. Austrian Botanist and monk. Experimented with peas. Studied inheritance of seven pairs of traits. Bred and crossbred thousands of plants. Determined that some traits were dominant and other recessive. Findings were published in 1866. Largely ignored for 34 years. c. Johan Friedrich Miescher - Swiss Biologist. Isolated nuclei of white blood cells in 1869. Led to identification of nucleic acid by Walter Flemming. d. Walter Sutton - Determined in 1903 that chromosomes carried units of heredity identified by Mendel. Named “genes” in 1909 by Wilhelm Johannsen, Danish Botanist. SHS Biotechnology Revised 2015 - 2016 Page 35 e. f. g. Thomas Hunt Morgan - Studied genetics of fruit flies. Early 1900’s. Experimented with eye color. His work contributed to the knowledge of X and Y chromosomes. Nobel Peace Prize in 1933 for research in gene theory. Ernst Ruska - Build the first electron microscope in 1932. German electrical engineer. Microscope offered 400X magnification. Alexander Fleming - Discovered penicillin in 1928. First antibiotic drug used in treating human disease. Penicillin first used in 1941. Penicillin credited with saving many lives during WWII when wounded soldiers developed infections. SHS Biotechnology Revised 2015 - 2016 Page 36 Making Yogurt Lab – Pre-Lab Reading Yogurt is a fermented milk product that contains the characteristic bacterial cultures Lactobacillus bulgaricus and Streptococcus thermophilus. All yogurt must contain at least 8.25% solids not fat. Full fat yogurt must contain not less than 3.25% milk fat, lowfat yogurt not more than 2% milk fat, and nonfat yogurt less than 0.5% milk. The full legal definitions for yogurt, lowfat yogurt and nonfat yogurt are specified in the Standards of Identity listed in the U.S. Code of Federal Regulations (CFR). The three styles of yogurt commonly found in the grocery store are Set type yogurt, Greek style yogurt and Swiss style yogurt. Set type yogurt is when the yogurt is packaged with the fruit on the bottom of the cup and the yogurt on top. Swiss style yogurt is when the fruit is blended into the yogurt prior to packaging. Greek is more solid and similar to cheese in consistency. Ingredients The main ingredient in yogurt is milk. The type of milk used depends on the type of yogurt – whole milk for full fat yogurt, lowfat milk for lowfat yogurt, and skim milk for nonfat yogurt. Other dairy ingredients are allowed in yogurt to adjust the composition, such as cream to adjust the fat content, and nonfat dry milk to adjust the solids content. The solids content of yogurt is often adjusted above the 8.25% minimum to provide a better body and texture to the finished yogurt. The CFR contains a list of the permissible dairy ingredients for yogurt. Stabilizers may also be used in yogurt to improve the body and texture by increasing firmness, preventing separation of the whey (syneresis), and helping to keep the fruit uniformly mixed in the yogurt. Stabilizers used in yogurt are alginates (carageenan), gelatins, gums (locust bean, guar), pectins, and starch. Sweeteners, flavors and fruit preparations are used in yogurt to provide variety to the consumer. A list of permissible sweeteners for yogurt is found in the CFR. Bacterial Cultures The main (starter) cultures in yogurt are Lactobacillus bulgaricus and Streptococcus thermophilus. The function of the starter cultures is to ferment lactose (milk sugar) to produce lactic acid. The increase in lactic acid decreases pH and causes the milk to clot, or form the soft gel that is characteristic of yogurt. The fermentation of lactose also produces the flavor compounds that are characteristic of yogurt. Lactobacillus bulgaricus and Streptococcus thermophilus are the only 2 cultures required by law (CFR) to be present in yogurt. Other bacterial cultures, such as Lactobacillus acidophilus, Lactobacillus subsp. casei, and Bifido-bacteria may be added to yogurt as probiotic cultures. Probiotic cultures benefit human health by improving lactose digestion, gastrointestinal function, and stimulating the immune system. General Manufacturing Procedure Steps to Making Yogurt 1. Adjust Milk Composition & Blend Ingredients: Milk composition may be adjusted to achieve the desired fat and solids content. Often dry milk is added to increase the amount of whey protein to provide a desirable texture. Ingredients such as stabilizers are added at this time. 2. Pasteurize Milk: The milk mixture is pasteurized at 185°F (85°C) for 30 minutes or at 203°F (95°C) for 10 minutes. A high heat treatment is used to denature the whey (serum) proteins. This allows the proteins to form a more stable gel, which prevents separation of the water during storage. The high heat treatment also further reduces the number of spoilage organisms in the milk to provide a better environment for the starter cultures to grow. Yogurt is pasteurized before the starter cultures are added to ensure that the cultures remain active in the yogurt after fermentation to act as probiotics; if the yogurt is pasteurized after fermentation the cultures will be inactivated. 3. Homogenize: The blend is homogenized (2000 to 2500 psi) to mix all ingredients thoroughly and improve yogurt consistency. [Type text] Page 37 4. Cool Milk: The milk is cooled to 108°F (42°C) to bring the yogurt to the ideal growth temperature for the starter culture. 5. Inoculate with Starter Cultures: The starter cultures are mixed into the cooled milk. 6. Hold : The milk is held at 108°F (42°C) until a pH 4.5 is reached. This allows the fermentation to progress to form a soft gel and the characteristic flavor of yogurt. This process can take several hours. 7. Cool : The yogurt is cooled to 7°C to stop the fermentation process. 8. Add Fruit & Flavors: Fruit and flavors are added at different steps depending on the type of yogurt. For set style yogurt the fruit is added in the bottom of the cup and then the inoculated yogurt is poured on top and the yogurt is fermented in the cup. For swiss style yogurt the fruit is blended with the fermented, cooled yogurt prior to packaging. 9. Package: The yogurt is pumped from the fermentation vat and packaged as desired. SHS Biotechnology Revised 2015 - 2016 Page 38 Grading Rubric – For Teacher’s Use Yogurt Lab (Full-Blown Lab) I will deduct points for each of the following: Name: ____________________________________________ Due Date: ____/____/____ This is to be written in complete sentences with correct grammar, spelling and punctuation. Question: Can the bacteria Lactobacillis bulgaricus and Streptococcus thermophillis be used to turn regular milk into yogurt? Hypothesis (written in “If… then…because” format): ___________________________________________________________________ ____________________________________________________________________________ _____________________________________________________________________________ Independent Variable: _______________________________________________ Dependent Variable: ________________________________________________ After completing the lab, tell what affect the INDEPENDENT VARIABLE had on the DEPENDENT VARIABLE (describe the relationship between the two variables in a minimum of 3 sentences)? ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ Materials: 100 ml of milk, thermometer, microscope, 25 grams nonfat dry milk, pH paper, prepared yogurt smears. stirring hot plate, incubator, Protocol: 1. 2. 3. Obtain 100ml of milk to a sterile 600 ml beaker. Use pH paper to take pH of the milk. Using a stirring hotplate add 25 grams of nonfat dry milk powder to milk. [Type text] stir bar, refrigerator, Overall: Not written in complete sentences. Does not use correct grammar spelling & punctuation. /5 Hypothesis: Does not use proper format – If/Then/Because. Hypothesis does not completely answer lab question. Hypothesis is poorly stated. Does not clarify what the student is thinking. /10 Independent Variable: No variable identified. Incorrect Independent variable. /5 Dependent Variable: No variable identified. Incorrect Dependent variable. /5 Link between variables: Answer not 3 sentences. Question is not answered. Link between variables is not explained clearly. The link described between variables is incorrect. Description of what was done in lab is not an answer to this question. /10 Page 39 Results pH Table Completed: 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. Kill existing bacteria (natural flora) by heating milk mixture to 85 ° C and maintain temperature for 20 minutes. While you are waiting and watching the milk so it doesn’t scorch, look at the prepared slides of the yogurt smear under the microscope. Draw what you see under HIGH power. Take pH of milk solution. Transfer to 250 ml beaker and cool on ice to 50 ° C Using a balance Inoculate milk with 5.6 grams (5 ml) of active plain yogurt (starter culture). Take pH of solution. Streptococcus thermophilus drops the pH of the solution to about 4.6 at this point some of the milk’s calcium caseinate separates. The free casein (milk protein) denatures, forming a curd. Incubate cups @ 45 degrees Celsius for 4-6 hours. Remove from incubator and refrigerate until next class period. When the solidifying liquid is refrigerated, Lactobacillus bulgaricus continue the fermentation, lowering the pH and adding to the characteristic flavor and aroma of the yogurt. At the start of the second class period, retrieve the yogurt from the refrigerator and take its pH for a last time. Taste-test the yogurt to determine if you like the flavor, and to determine if it tastes similar to commercial yogurts. Smear the yogurt on a slide and stain it as directed by the teacher. Draw your smear in the results section next to the prepared slide of yogurt. _____/10 Results: Data Answer: pH #1 pH #2 pH #3 pH #4 SHS Biotechnology Revised 2015 - 2016 Page 40 Microscope Drawings Complete: Microscope Drawings: Prepared Slide of Commercial Yogurt My Yogurt _____10 Hypothesis Correct? Power of Magnification: Power of Magnification: Based on the data collected, was your hypothesis correct? Give 2 pieces of data that clarify your decision: ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ CHOOSE 1 based on whether your hypothesis is correct: If your hypothesis was wrong, what was wrong with it (detail – minimum 2 sentences)? OR If your hypothesis was right, generate a new lab question that could be asked to create a new lab experiment to follow up on what you learned in this experiment. ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ Uses the words “Yes” or “No” to start. First sentence does not state clearly, “My hypothesis was correct/not correct.” 2 items of detailed data measured in lab (numbers, observations) were not provided to support decision. Data used is not explained. Student does not make a choice. A hypothesis cannot be both right and wrong. The decision about hypothesis correctness does not match the data collected in lab. _____/10 Hypothesis related choice 1: Wrong question answered. Does not tell what revisions to hypothesis would make it correct. Evaluates lab procedures instead of the hypothesis. Does not tell why the hypothesis was wrong. Not 2 sentences long. _____/10 Hypothesis related choice 2: Wrong question answered. Basically a repeat of the question from this lab. Not in the form of a question. Little thought or creativity shown. Not testable in lab. Question is too general. Neither question is answered. _____/10 SHS Biotechnology Revised 2015 - 2016 Page 41 Choice 1: CHOOSE 1: How could the experiment be repeated to make it more accurate (describe 2 ways to improve the experiment)? OR Were there any mistakes made in the process of following the procedures that affected the outcome? If so, how could these mistakes be corrected for (be detailed about the mistakes you made and how you would correct for them)? ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ What is the relationship between Streptococcus thermophilus and Lactobacillus bulgaricus? How do the two bacteria influence one another to make a better yogurt? Detail the substances in the milk that each bacteria uses and produces and how this influences the other bacteria. ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ Based on the information in the graph below what is the major effect of the increase in the size of the population of L. bulgaricus, which produces lactic acid as a waste product? This answer requires more than one or two sentences. _________________________________________ _________________________________________ _________________________________________ _________________________________________ _________________________________________ _________________________________________ _________________________________________ _________________________________________ SHS Biotechnology Revised 2015 - 2016 Not enough detail. 2 ways not given. Answers were complaints not solutions. Saying, “Follow all instructions” is not a solution. 1 sentence is not enough. Don’t blame others, find solutions. This student does not understand what was happening in lab. _____/10 Choice 2: Doesn’t tell what you did wrong & how to fix it next time. Should be longer than 1 sentence. Saying, “We didn’t make any mistakes,” means you have to answer the other question. Saying, “Follow all the steps better,” does not work for an answer. This student does not understand what was happening in the lab. _____/10 Application: Answers should be specific with at least one example/career/life experience listed. Not 3 sentences long. Student does not understand how to apply this to life or what was done in lab. Don’t give opinions (i.e this is dumb). Don’t retell what we did in lab. _____/10 Specific Question #1: _____/15 Specific Question #2: _____/15 Total Score: ________/125 Page 42 Dental Caries & the Microbial Flora of the Mouth Lab – Pre-Lab Reading The mouth is colonized by 200 to 300 bacterial species, but only a limited number of these species participate in dental decay or dental caries. Dental caries are caused by the irreversible solubilization of tooth mineral by acid produced by certain bacteria that adhere to the tooth surface in bacterial communities known as dental plaque. Streptococcus mutans is the main cause of dental decay. Various lactobacilli are associated with this process as well. The tooth surface normally loses some tooth mineral from the action of the acid formed by plaque bacteria after ingestion of foods containing fermentable carbohydrates. This mineral is normally replenished by the saliva between meals. However, when fermentable foods are eaten frequently, the low pH in the plaque is sustained and a net loss of mineral from the tooth occurs. This low pH selects for aciduric organisms, such as S mutans and lactobacilli, which store polysaccharide and continue to secrete acid long after the food has been swallowed. The tooth surfaces are unique in that they are the only body part not subject to metabolic turnover. Once formed, the teeth are, under the correct conditions, essentially indestructible, as witnessed by their importance in fossil records and forensic medicine. Yet in the living individual, the integrity of the teeth is assaulted by a microbial challenge so great that dental infections rank as the most universal affliction of humankind. The discomfort caused by these infections and their enormous cost (dental infections rank third in medical costs, behind heart disease and cancer, in the United States) gives dental diseases prominence despite their non-life-threatening nature. Dental Caries (Decay) Dental decay is due to the dissolution of tooth mineral (primarily hydroxyapatite, Ca10 (P04)6(0H)2) by acids derived from bacterial fermentation of sucrose and other dietary carbohydrates. These bacteria live in bacterial communities known as dental plaque which accumulates on the tooth surface. For almost a century it was believed that any bacterial community on the tooth surface could cause decay, and treatment was almost exclusively the mechanical cleaning of these surfaces by toothbrushing, using some type of mild abrasive. Such treatments based upon debridement and, in extreme cases, upon dietary carbohydrate restriction, were singularly unsuccessful in reducing dental decay. In fact, the prevalence of dental decay was so high among young men that it was the major cause of rejection from military service in World Wars I, II, and the Korean War. This staggering amount of dental morbidity led to the formation of dentistry as a separate health profession in the late 19th century; to the expectation that all people would, if they lived long enough, be edentulous (toothless); and to a dental health bill to the public of approximately 34 billion dollars per year in 1990. Things have changed. Water fluoridation has proven to be a most cost-effective way of reducing decay; fluoride dentifrices were even more effective than initially projected; and research findings indicate that most cavities actually reflect a sucrose-dependent Streptococcus mutans infection. Individuals at risk for this infection can be diagnosed and treated by frequent mechanical intervention, by intensive application of prescription levels of fluorides or other antimicrobials (such as chlorhexidine), by restriction of ingestion of sucrose between meals, or by use of products that contain sucrose substitutes (such as xylitol). The net result is that dental decay in the late 20th century is a controllable infection and should be preventable in many individuals. Almost 50% of young children are caries-free, and the level of edentia among individuals over 65, has dropped from 50% to about 20%. [Type text] Page 43 Process Lab: Dental Caries & the Microbial Flora of the Mouth MATERIALS: Liquified tube of Snyder Agar Parafilm square Incubator Procedure: 1. Obtain one Snyder agar deep tube for your group from the water bath. Label the tube so you can identify it later. 2. Chew one square of paraffin for a period of 3 minutes without swallowing the saliva. As saliva develops, collect it in the Snyder agar test tube (spit it in). 3. Mix the contents of the tube thoroughly by rolling the tube between the palms of your hands or by tapping it with your finger. If you choose, you may invert the tube several times. However, test tube lids sometimes leak when exposed to a hot liquid. 4. Rapidly cool the inoculated tube of Snyder agar in and ice-water bath. 5. Incubate the tube for 48 hours at 37 degrees C. Observe cultures and record the color of each tube, as well as the susceptibility of each student to dental caries. RESULTS: 1. Examine the Snyder test cultures daily during the 32-hour incubation period for a change in the color of the culture medium. Use an un-inoculated tube of the medium as a control. Record the color of the cultures in the chart. 1. Positive: Complete color change; green is no longer dominant. 2. Negative: No color change or a slight color change: medium retains green color throughout. Student Name SHS Biotechnology Revised 2015 - 2016 Color of Snyder Test Cultures Caries Susceptibility (Yes) Or (No) Page 44 Media Preparation & Plate Pouring Notes I. II. III. IV. V. VI. A culture is the microorganisms that grow in a culture medium. a. Culture media are solutions containing all of the nutrients and necessary physical growth parameters necessary for microbial growth. Media can be distinguished qualitatively as: a. solid vs. broth b. reducing c. selective d. differential Types of Media a. Solid – Agar Plates, Slants, Stabs b. Broth – c. Simple- contains few nutrients d. Selective- the medium contains agents which inhibit or control the growth of Bacteria or identify a characteristic of the bacteria. Ex: Antibiotics. e. Solid medium is media containing agar or some other, mostly inert solidifying agent. i. It has physical structure (broth lacks structure) and this allows bacteria to grow in physically informative or useful ways (e.g., as colonies or in streaks). ii. Solid medium is usually used as slants, stabs or petri dishes . f. Enriched Medium has added growth factors: i. Is preprepared media to which additional growth factors have been added. ii. Is used to increase the relative concentration of certain microorganisms in the culture prior to plating on solid selective medium. g. Differential media allow the growth of more than one microorganism of interest but with morphologically distinguishable colonies. i. Note that almost any medium containing a specific substrate and well tailored indicator can be used as a differential medium. Broth medium is media lacking a solidifying matrix. Media of many types can be obtained premixed, in an often dehydrated and powdered state. Such media are called preprepared media. Selective Medium is designed to suppress the growth of some microorganisms while allowing the growth of others (i.e., they select for certain microbes). a. Examples of selective media include: i. mannitol salts agar (selects against non-skin flora) [Type text] ii. M a c C o n k e y a g a r ( s e l e c t s a g a i n s t g r Page 45 VII. VIII. IX. X. am-positives) iii. eosin-methylene blue agar (selects against gram-positives) iv. phenylehyl alcohol agar (selects against gram-negatives) v. Examples of Selective media include: vi. mannitol salts agar (mannitol fermentation = yellow) vii. blood agar (various kinds of hemolysis) Preserving Cultures is important for: a. Identification, vaccine production, industrial use. b. Methods of preserving cultures include: i. Refrigeration, stabs, slants, lyophilization, freezing , Refrigeration (a.k.a.,4°C). ii. Effective short term preservation: 1. Refrigeration can be effective for short periods. Stabbing a. Cultures are stabbed deeply into agar using a inoculating needle. b. The stabs are incubated until visible cultures form, then sealed and stored at room or lower temperature. Slant Method - Cultures may be streaked onto the surface of the solid medium in a slant tube. Lyophilization is the freeze-drying of cultures. Cultures are first frozen and then dried under high vacuum. To revive cultures they are rehydrated by broth. SHS Biotechnology Revised 2015 - 2016 Page 46 Process Lab: Media Preparation and Plate Pouring Materials 10% bleach solution LB agar base 2@250 mL flask Autoclave 10 mL pipette pump Sparker safety glasses LB broth base 250 mL Media bottle 2 large Petri dishes 25 mL pipette hot plate gloves ink marker aluminum foil 6 small Petri dishes 25 mL pipette pump balance small weigh boat lab scoops hot pad 10 mL pipette Bunsen burner Part A: LB agar Preparation Procedure: 1. 125 mL of agar is the maximum amount that can be prepared in the 250 mL flask. 2. Obtain a clean 250 mL flask or beaker (beakers are easier to pour). 3. Calculate the amount of LB agar needed to make 70 mL of Agar. The recipe on a media bottle says to use 40g of media base in 1 L of dH 2O. Mass1 = Mass2 Volume1 Volume2 M1= the mass of media base to use in the original recipe’s volume (usually 1 L) V1= the volume of solvent (dH2O) in the original recipe’s volume (usually 1 L) M2= the mass of media base needed for the desired volume V2= the desired final volume of media 4. 5. 6. 7. 8. Using a small weigh boat, measure out the amount of Lb agar base required to make 70 mL of agar (30 mL /dish + 10 extra mL). Pour 70 mL of distilled or tap water (follow teacher directions) into a 250 mL beaker. Begin heating the water (about 350 degrees), stirring it as you do. Watch for steam and condensation to appear in the beaker. DO NOT wait for the water to boil. As soon as you see the first signs of steam, pour the LB agar base in to the flask and heating water. WATCH THE MIXTURE CAREFULLY!!! DO NOT let it boil over. If the liquid begins to boil, reduce temperature and remove the flask from the heat for a few seconds. 9. After a few minutes, the agar suspension should become a clear yellow color. If it is even slightly foggy, it will not set up. Keep heating until it is clear. 10. Using a hot pad, carefully remove the flask from the hot plate. Loosely cover the flask with the aluminum foil. 11. While you are waiting for the Agar cool, label two Petri dishes along the edge of the bottom of the plate with your initials, date, period and type of medium(LB). 12. Setup and light your Bunsen burner. [Type text] Page 47 13. “Flame” the flask top (pass bottle top through the hot part of the flame three times). 14. Open the Petri dish lid by tilting it upward slightly – DO NOT take the lid off completely. Pour agar into the Petri dish until it is one-half the height of the Petri plate. Watch as you pour, when you have enough agar in the bottom of the dish to cover it completely, stop pouring. 15. Continue pouring the other plates. 16. Stack the plates on top of each other AFTER the agar sets up completely. 17. Allow drying in a clean area, undisturbed for at least 24 hours before use. 18. Plates are good for about 2 weeks. Store Petri dishes in a cool, dark place. Part B: LB Broth Preparation Procedure: (50 mL of broth is an appropriate volume for a 125 mL flask) 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. Obtain a clean autoclaved 250 mL flask. Calculate the amount of LB broth needed to make 50 mL of broth. The recipe on a media bottle says to use 25g of media base in 1 L of dH2O. Measure out the amount of LB broth base required for the volume of broth desired. Record that mass in your notebook along with your calculations. Place the LB broth base into the clean 250mL flask Very slowly add 35 mL of distilled water, stirring as it is added. The water should at first make a thick paste. As you add more water and stir the broth mixture, the LB broth base will eventually become suspended. LB broth base goes into solution much more easily than agar base. Add more water until a total volume of 50 mL of suspended broth is achieved. Place the flask on a hot plate and turn the heat to about 350 degrees. WATCH THE MIXTURE CAREFULLY!!! DO NOT let it boil over. Once the liquid begins to boil, reduce temperature. The broth solution should become clear. Using a pot holder remove the flask. Loosely cover the flask with the aluminum foil. Place the flask in the autoclave, along with the rest of the class’ bottles. Your teacher will run the autoclave program. Cool the broth to room temperature before using. Results: The result for this lab is the production of the agar plates and LB Broth. SHS Biotechnology Revised 2015 - 2016 Page 48 Bacteria Culture Notes I. II. III. IV. V. VI. VII. VIII. Importance of Bacteria a. Micro-organisms colonize every environment on earth. b. >80% of life’s history was bacterial. c. You have more bacterial cells than human cells. d. Microbes play a key role in the biosphere. e. Pathogenic microbes globally are the most important cause of human disease and death. Monera: the Prokaryotic Kingdom has been split into: a. Archaebacteria b. Eubacteria Bacteria are usually classified on the basis of their shapes. Broadly, they can be divided into: a. Rod-shaped bacteria (Bacilli) b. Sphere-shaped bacteria (Cocci) c. Spiral-shaped bacteria (Spirilla) d. While many bacteria live singly, others are found in aggregates or clusters. These aggregates are named based on the arrangement of the bacterial cells of which they are composed. Using cocci as an example: i. diplococcus: are in sets of two ii. streptococcus: are in chains iii. staphylococcus: are in clusters (staphylo = a bunch of grapes). Archaebacteria: a. Extreme Thermophiles b. Methanogens c. Extreme Halophiles Extreme Thermophiles a. Hot pots, Hydrothermal vents, High temperatures, and Sulfur. i. Thermus Aquaticus Methanogens a. Marshes, lake sediments, digestive tracts of mammals (cows), sewage disposal plants, Natural Gas Extreme Halophiles a. Dead Sea, Great Salt Lake, Evaporating Ponds, Salt b. Halobacterium halobium Eubacteria Heterotrophs a. Use organic molecules as food source b. Parasites - Obtain nutrients from living organisms c. Saprophytes - Feed on dead organisms and organic wastes i. Recyclers or decomposers SHS Biotechnology Revised 2015 - 2016 Page 49 d. e. f. IX. X. XI. XII. XIII. Photosynthetic Autotrophs - Use Sunlight to make food Cyanobacteria - Blue-green, red or yellow. Chains of independent cells. Chemosynthetic Autotrophs - Make food from chemosynthesis using Sulfur, Nitrogen Metabolism. g. Obligate aerobes - Require oxygen. h. Obligate anaerobes - Killed by oxygen. i. Facultative Aerobes - Both with or without oxygen (cellular respiration vs. fermentation.) Classification on the Basis of Oxygen Requirement - This classification is based on the requirement of oxygen for the survival of the bacterium. a. Aerobic (Need Oxygen) b. Anaerobic (Do not need Oxygen) Bacterial Structure a. Very small cell b. Lack membrane bound organelles c. Lack nuclear membrane d. DNA circular – Nucleoid e. Biochemical processes in cytoplasm. BINARY FISSION - Asexual Reproduction Endospore - Survival Mechanism. Contains Bacterial DNA and a Small amount of cytoplasm. Encased by a tough outer covering that resists drying out, temperature extremes , harsh chemicals. a. Bacterium rests and doesn’t reproduce. b. Germinates when conditions are favorable The Meaning of the Names of Some Microorganisms a. Escherichia coli: Named after Theodore Escherich in 1888; found in the colon b. Entamoeba histolytica: Ent, intestinal; amoebae, shape/movement; histo, tissue; lytic, lysing or digesting tissue SHS Biotechnology Revised 2015 - 2016 Page 50 Notes Aseptic Technique I. II. III. IV. V. VI. VII. Early civilizations practiced salting, smoking, pickling, drying, and exposure of food and clothing to sunlight to control microbial growth. Use of spices in cooking was to mask taste of spoiled food. Some spices prevented spoilage. In mid 1800s Semmelweiss and Lister helped developed aseptic techniques to prevent contamination of surgical wounds. Before then: a. Nosocomial infections caused death in 10% of surgeries. b. Up to 25% mothers delivering in hospitals died due to infection. Control of Microbial Growth: a. Disinfection: Reducing the number of pathogenic microorganisms to the point where they no longer cause diseases. May use physical or chemical methods. b. Disinfectant: Applied to inanimate objects. c. Antiseptic: Applied to living tissue (antisepsis). d. Degerming: Mechanical removal of most microbes in a limited area. Example: Alcohol swab on skin. e. Sanitization: Use of chemical agent to minimize chances of disease transmission. E.g: Hot soap & water. f. Sepsis: Comes from Greek for decay or putrid. Indicates bacterial contamination. g. Asepsis: Absence of significant contamination. Aseptic techniques are used to prevent bacterial contamination. a. Bacteriostatic Agent: An agent that inhibits the growth of bacteria, but does not necessarily kill them. b. Germicide: An agent that kills certain microorganisms. c. Bactericide: An agent that kills bacteria. Most do not kill endospores. d. Viricide: An agent that inactivates viruses. e. Fungicide: An agent that kills fungi. f. Sporicide: An agent that kills bacterial endospores of fungal spores. Physical Methods of Microbial Control: a. Heat: Kills microorganisms by denaturing their enzymes and other proteins. Heat resistance varies widely. b. Moist Heat: Kills microorganisms by coagulating their proteins. In general, moist heat is much more effective than dry heat. c. Boiling: Heat to 100oC or more at sea level. Kills some forms of bacterial pathogens, almost all viruses, and fungi and their spores within 10 minutes or less. Endospores and some viruses are not destroyed this quickly. i. Reliable sterilization requires temperatures above that of boiling water. Autoclave: Chamber which is filled with hot steam under pressure. Preferred method of sterilization, unless material is damaged by heat, moisture, or high pressure. a. Temperature of steam reaches 121oC at twice atmospheric pressure. Most effective when organisms contact steam directly or are contained in a small volume of liquid. b. All organisms and endospores are killed within 15 minutes. Require more time to reach center of solid or large volumes of liquid. Pasteurization: Developed by Louis Pasteur to prevent the spoilage of beverages. Used to reduce microbes responsible for spoilage of beer, milk, wine, juices, etc. a. Classic Method of Pasteurization: Milk was exposed to 65oC for 30 minutes. SHS Biotechnology Revised 2015 - 2016 Page 51 High Temperature Short Time Pasteurization (HTST): Used today. Milk is exposed to 72oC for 15 seconds. Ultra High Temperature Pasteurization (UHT): Milk is treated at 140oC for 3 seconds and then cooled very quickly in a vacuum chamber. Advantage: Milk can be stored at room temperature for several months. Dry Heat: Kills by oxidation effects. a. Direct Flaming: Used to sterilize inoculating loops and needles. Heat metal until it has a red glow. b. Incineration: Effective way to sterilize disposable items (paper cups, dressings) and biological waste. c. Hot Air Sterilization: Place objects in an oven. Require 2 hours at 170oC for sterilization. Dry heat transfers heat less effectively to a cool body, than moist heat. d. Filtration: Removal of microbes by passage of a liquid or gas through a screen like material with small pores e. High Efficiency Particulate Air Filters (HEPA): Used in operating rooms and burn units to remove bacteria from air. Low Temperature: Effect depends on microbe and treatment applied. a. Refrigeration: Temperatures from 0 to 7oC b. Freezing: Temperatures below 0oC. c. Flash Freezing: Does not kill most microbes. Slow Freezing: More harmful because ice crystals disrupt cell structure. Osmotic Pressure: The use of high concentrations of salts and sugars in foods. a. Plasmolysis: As water leaves the cell, plasma membrane shrinks away from cell wall. Cell may not die, but usually stops growing. Radiation: Three types of radiation kill microbes: Chemical Methods of Microbial Control a. Types of Disinfectants i. Halogens: Effective alone or in compounds. ii. Alcohols: Kill bacteria, fungi, but not endospores or naked viruses. b. c. VIII. IX. X. XI. XII. SHS Biotechnology Revised 2015 - 2016 Page 52 Gram Staining Notes I. Cell membrane structure: II. Gram Staining Method Depends on whether there is a layer of lipid on the outside of the cell wall. How to gram stain: a. Heat fix the bacteria with a Bunsen burner. b. Flood the sample with crystal violet for 1 minute. i. Rinse with water until water runs clear from the slide. c. Flood the slide with Iodine for 1 minute. i. Iodine functions as a MORDENT in this step. It helps the stain to bond to the peptidoglycan. d. Rinse the sample with Ethanol. This will de-color any bacteria that have lipids on the outside of their cell wall. e. Flood the sample with Safranin for 30 seconds. i. Rinse with water until water runs clear. III. SHS Biotechnology Revised 2015 - 2016 Page 53 Gram Staining Lab – Pre-Lab Reading Gram staining, also called Gram's method, is a method of differentiating bacterial species into two large groups (gram-positive and gramnegative). The name comes from the Danish bacteriologist Hans Christian Gram who developed the technique. Gram, (1853–1938) developed the technique while working with Carl Friedländer in the morgue of the city hospital in Berlin in 1884. Gram devised his technique not for the purpose of distinguishing one type of bacterium from another but to make bacteria more visible in stained sections of lung tissue. He published his method in 1884, and included in his short report the observation that the typhus bacillus did not retain the stain. Gram staining differentiates bacteria by the chemical and physical properties of their cell walls by detecting peptidoglycan, which is present in a thick layer in gram-positive bacteria. In a Gram stain test, gram-positive bacteria retain the crystal violet dye, while a counterstain (commonly safranin) added after the crystal violet gives all Gram-negative bacteria a red or pink coloring. The Gram stain is almost always the first step in the identification of a bacterial organism. Gram staining is a valuable diagnostic tool in both clinical and research settings. But, not all bacteria can be definitively classified by this technique. This gives rise to gram-variable and gramindeterminate groups as well. Gram’s Stain is one of the most frequently used processes in identifying bacteria and is used daily in hospitals. It is a primary test which quickly and cost effectively divides bacteria into one of two types: Gram-Positive or Gram-Negative. Gram-Positive Bacteria have thicker cell walls with less fatty substances (lipids) than Gram-Negative bacteria. When washed with solvent, the cell pores close becoming less permeable and are able to retain the stain, in this case, purple. Gram-Negative Bacteria have thinner cell walls with more lipids. The solvent dissolves the lipids, which combined with thinner cell walls, washes out or decolorizes the stain. In order to see the decolorized bacteria, a counter stain (Safranine) is added to exaggerate the contrast with Gram-Negative cells. Principle of Gram's Stain The crystal violet stain is the primary stain, which stains everything in the smear purple- blue. The Gram's iodine acts as a mordant that causes the crystal violet to penetrate and adhere to the gram-positive cell. The acetone-alcohol mixture acts as the decolorizer that washes the stain away from everything in the smear except the gram-positive organisms. The safranine is the counter-stain that stains everything in the smear that has been decolorized in gram-negative organisms. There are six basic steps: 1. Apply a smear of bacteria on to a slide. Air dry and then heat fix by passing it through a flame a few times. Make sure you air dry the bacteria before heat fixing. 2. Add about 5 drops of Crystal Violet to the culture. Let stand for one minute. Wash briefly with water and shake off excess. 3. Add about 5 drops of iodine solution to the culture. Let stand for 30 seconds, wash briefly with water and shake off excess. 4. Tilt slide and decolorize with solvent (acetone-alcohol solution) until purple color stops running. Be careful not to over-decolorize. 5. Add about 5 drops of Safranine O. Let stand for one minute, wash briefly with water and shake off excess. 6. Examine under microscope at both 400x and 1,000x oil immersion. Examples: Gram-Positive Bacteria: Staphylococcus (Mostly harmless sphere-shaped, skin bacteria); Streptococcus (rod shaped bacteria: Rheumatic fever, Scarlet fever). Gram-Negative Bacteria: Escherichia, Salmonella, Shigella (Meningitis, Gonorrhea, Flu and various Respiratory Diseases) SHS Biotechnology Revised 2015 - 2016 Page 54 Gram Staining - Process Lab MATERIALS: Bacterial Cultures staining beaker, Prepared Slides glass slides, Crystal violet stain, bibulous paper, Gram's iodine, lens paper, 95% ethyl immersion oil, alcohol, microscope. safranin PROCEDURE: 1. Pick 2 prepared slides from those provided by your teacher (one gram + and one gram -). View these under oil immersion and draw them in the results section before you begin making your own slides, so you will have a basis for comparison. Obtain glass slides and prepare bacteria smears as demonstrated by your teacher. 2. Gently flood smears with crystal violet and let stand for 1 minute. 3. Gently wash with tap water. 4. Gently flood smears with the Gram's iodine mordant and let stand for 1 minute. 5. Gently wash with tap water. 6. Decolorize with 95% ethyl alcohol. Caution: Do not over-decolorize. Add reagent drop by drop until alcohol runs almost clear, showing only a blue ting. 7. Gently wash with tap water. 8. Counterstain with Safranin for 45 seconds. 9. Gently wash with tap water. 10. Blot dry with bibulous paper and examine under oil immersion. 11. Draw your Gram Stained bacteria (in color) in your results section of lab write up. Results: Bacterial Sample #1 Bacterial Sample #2 Bacterial Sample #3 Bacterial Sample #4 Power: Power: Power: Power: SHS Biotechnology Revised 2015 - 2016 Page 55 Prepared Slide #1 Prepared Slide #1 Prepared Slide #1 Prepared Slide #1 Power: Power: Power: Power: SHS Biotechnology Revised 2015 - 2016 Page 56 Aseptic Technique and Streaking for Isolation – Pre-Lab Reading Modern microbiology originated in Koch's laboratory with the development of agar-based media and the streakplate technique for obtaining pure cultures. This method consists of spreading source material over an agar surface until one microorganism at a time falls off of the loop. The medium is then incubated until colonies arise. Theoretically, each colony represents a single type of microorganism that originated from a single cell. Most source materials contain a large number of microorganisms. Pond water can be expected to contain 105 to 106 bacteria/ml. Soil can contain more than 108 bacteria per ml, and a barely visibly turbid culture of Escherichia coli contains at least 107 bacteria per ml. This means that a loop of these materials spread over the surface of an agar plate would yield 103 to 105 colonies, producing a confluent lawn of growth on the plate. Thus, simply smearing a loop of source material over a plate will not yield isolated colonies. Additional steps are needed. To streak a sample for isolation: 1. Draw a "T" on the back of the plate, dividing it into three sections. 2. Flame the inoculation loop, then allow it to cool a few seconds. Then pick up a loop of sample. 3. Turn the plate so that the top of the "T" is near the palm of your non-dominant hand. 4. Crack the lid of the plate by lifting it with your thumb and index finger. 5. Beginning at the edge of the plate above the top of the "T" and move the loop back and forth across the agar surface, pulling it towards the top of the "T" as you do so. Do this by flexing and extending the fingers holding the loop, not by moving your entire hand. Done properly, this process will result in a series of parallel lines at the top of the plate. 6. Remove the loop. Flame it again to sterilize it. Do not insert the loop back into the sample to pick up more bacteria. 7. Turn the plate 90 degrees counter-clockwise. 8. This time, streak the second third of the plate. Make sure that your loop crosses over the section that you previously streaked in order to pick up the bacteria that were deposited there and thin them out. 9. Repeat steps 6 through 8 for the last third of the plate. SHS Biotechnology Revised 2015 - 2016 Page 57 Process Lab: Aseptic Technique and Streaking for Isolation Objectives: 1. To learn the characteristics of aseptic technique, 2. To learn how to streak cells from a liquid bacterial culture onto an agar plate. In this lab, you will be given a liquid culture containing a mixture of three different bacterial species. You will attempt to identify the three bacterial species in this first week using several standard microbiology identification techniques. However, before you can attempt to identify the individual bacterial species, you will need to separate the bacteria in the mixed liquid culture from each other. The method you will use to accomplish this is a technique called streaking. The objective of this technique is to deposit single bacterial cells from the liquid culture over the surface of the agar medium in the plate. The streaking technique allows you to do this as each successive streak dilutes the number of cells from the previous streak, ideally resulting in single cells deposited within the area of the final streak. When culturing microorganisms, you must practice aseptic technique (also called sterile technique). The two purposes of aseptic technique are: to prevent bacteria in the environment from contaminating your work, to prevent bacteria in your work from contaminating you and the environment. To avoid contamination, it is essential to practice aseptic technique. When working with microorganisms, always follow these rules: 1. Sterilize the surfaces that the bacteria will contact. This kills bacteria in the environment and prevents them from contaminating your cultures. 2. Since your hands contain bacteria, you must wash your hands with soap and water, before and after you handle microorganisms. 3. Glassware and medium are sterilized by subjecting them to high temperature and pressure in an instrument called an autoclave or by UV irradiation. 4. Reduce the time that sterile medium, cultures, or bacteria are exposed to the air. 5. Work in an area with a low resident population of bacteria. Materials: (1) Bacterial medium plate (1) tube of mixed bacterial culture (1) test tube rack (1) Bunsen burner (1) Spray bottle of 10% Bleach (1) tube of uninoculated medium (1) inoculating loops (1) striker/lighter Procedures: SHS Biotechnology Revised 2015 - 2016 Page 58 1. Label the outside margin of the bottom of your agar plate with your name, lab date and time. 2. Whenever bacterial cells are transferred, strict aseptic (sterile) technique must be used. 3. The agar plate should be placed upside-down so that the lid is on the bench and the agar is facing up. 4. Flame the inoculating loop until it is red hot. This will sterilize it. 5. Remove the cap from the culture tube by grasping the cap between your baby finger and the palm of the hand that is holding the loop. Do not place the cap on the bench. This prevents the cap from becoming contaminated. 6. Flame the mouth of the tube by passing it rapidly through the Bunsen burner flame two or three times. 7. Let the loop cool for a few seconds (10 or so will do), then put the loop into the liquid inside the tube. 8. Remove the inoculum (a culture medium in which microorganisms are grown), re-flame the mouth of the tube, and replace the cap. 9. The inside of the loop contains a film of medium and bacteria cells. 10. Lift the bottom of the agar plate and hold it perpendicular to the bench top (this angle prevents air contamination). 11. Allow the loop to glide over the surface of the agar and "scribble" back and forth in the area of the plate illustrated in Step 1 of Figure 2. 12. Place the plate back into the lid. 13. Flame the loop until it is red hot. Then cool it by plunging it into the agar. 14. This flaming will kill all the remaining bacterial cells in the inoculating loop. 15. Touch the loop to the first set of streaks once and scribble the loop on the agar as illustrated in Step 2 of Figure 2. 16. The loop only touches the first streak once thereby diluting the cells in the area of the second streak. 17. Flame the loop, cool in the agar, and make a third set of streaks as illustrated in Step 3 of Figure 2, ensuring that the loop only contacts the second set of streaks once. This will further dilute the number of cells in the area of the third streak. 18. After streaking, replace the lid of the Petri plate and place it in the incubator. These plates will be incubated at 30°C for 24-36 hours to permit colony growth. 19. During the next lab period, retrieve your plates and draw what you see on your plate in the results section. [Type text] Page 59 Results: SHS Biotechnology Revised 2015 - 2016 Page 60 Notes: Colony Morphology I. III. Colony morphology gives important clues as to the identity of their constituent microorganisms. a. Size: b. Colony shape: c. Margin (edge): d. Elevation: e. Texture: f. Light Transmission: g. Color: Colony Size - Dependent on the: a. type of organism b. the growth medium c. the number of colonies present on a plate d. culture medium characteristics. e. Colony size usually stabilizes after a day or two of incubation. Colony Shape IV. Type of Margin (edge) II. SHS Biotechnology Revised 2015 - 2016 Page 61 V. Colony Elevation a. Colonies can vary in their elevations both between microorganisms and growth conditions, and within individual colonies themselves. I. Colony Texture - Possible textures include: a. shiny to dull b. smooth to wrinkled c. rough d. granular e. mucoid Colony Light Transmission a. The light transmission through colonies can range from: i. complete (transparent) ii. through intermediate (translucent) iii. through completely lacking (opaque) Colony Pigmentation a. Colonies can come in a rainbow of colors. Bacterial Cell Shapes: a. 3 Basic: i. Coccus ii. Bacillus iii. Spirillum (spirochete) b. Shape Modifiers: i. Diplo – twos or pairs ii. Strepto – Chains iii. Staphylo – Clumps iv. Tetra – groups of four II. III. IV. SHS Biotechnology Revised 2015 - 2016 Page 62 Grading Rubric – For Teacher’s Use Full Blown Lab: Bacterial Identification I will deduct points for each of the following: Name: _________________________________ Due Date: ____/____/____ This is to be written in complete sentences with correct grammar, spelling and punctuation. Question: Using the techniques learned in class, can you correctly isolate and identify the three mystery bacteria? Hypothesis (written in “If… then…because” format): _____________________________________________________________________________________ _____________________________________________________________________________________ Independent Variable: _______________________________________________ Dependent Variable: ________________________________________________ After you complete the lab, tell what affect the INDEPENDENT VARIABLE have on the DEPENDENT VARIABLE (describe the relationship between the two variables in a minimum of 3 sentences)? _______________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ Materials: PCA plate streaked last week in lab pair of forceps striker 95% ethanol immersion oil microscope grease pencil inoculating loop compound microscopes with 100X crystal violet Gram's iodine safranin glass slides Bunsen burner Overall: Not written in complete sentences. Does not use correct grammar spelling & punctuation. /5 Hypothesis: Does not use proper format – If/Then/Because. Hypothesis does not completely answer lab question. Hypothesis is poorly stated. Does not clarify what the student is thinking. /10 Independent Variable: No variable identified. Incorrect Independent variable. /5 Dependent Variable: No variable identified. Incorrect Dependent variable. /5 Link between variables: Answer not 3 sentences. Question is not answered. Link between variables is not explained clearly. The link described between variables is incorrect. Description of what was done in lab is not an answer to this question. /10 Protocol: Identification of unknown bacteria SHS Biotechnology Revised 2015 - 2016 Page 63 Part A - Colony Morphology 1. Examine the plates containing colonies of bacteria that you streaked. 2. Pick out three well-isolated colonies of different morphology. You should observe orange, cream, and white-colored colonies. 3. Using the grease pencils provided, circle and label (on the bottom of the Petri dish) a well isolated orange colony “1,” a well-isolated cream colony “2,” and a well-isolated white colony “3.” 4. Using the terms in Figure 3, describe the morphology of these colonies and enter your observations in Table 1. Part B - Gram Stain, Cell Morphology, and Cell Size 1. The cells of each of the three circled colonies will be stained using the Gram stain to determine if the cells are Gram-negative or Gram-positive. 2. Using a grease pencil, draw 3 circles on the top of a warm, clean, grease-free slide and label the circles “1,” “2,” and “3.” 3. Using an inoculating loop, transfer a loopful of water to the center of each circle on the slide. 4. Flame the loop, cool the loop by plunging it in the sterile agar, pick up a small sample of colony “1,” transfer the sample to the center of the circle labeled “1,” and spread the cells evenly while mixing them with the water. 5. Repeat this entire step for colony “2” and colony “3.” 6. Let the smears air-dry until they don’t look wet. When dry, the smear should be only faintly visible; a thick opaque film where the bacteria are piled on top of one another is useless. 7. Once the smears are dry, and holding the side of the slide with a clip, pass it through the Bunsen burner flame until the water is dry. DO NOT LET THE SLIDE GET HOT, this will destroy the bacteria and you will have to start the process over again. But, the slide does need to be worm for this step to work. 8. Cover the smears with crystal violet stain and let it stand for 1 minute. 9. Rinse the smears with drops of water until the water runs clear from the colonies. 10. Cover the smears with Gram's Iodine and let it stand for 1 minute. 11. Flood the smears on the slide with 95% ethanol until the ethanol runs clear from the smears. DO NOT let the ethanol remain in contact with the slide for any longer than 5-10 seconds, otherwise Gram-positive organisms may appear to be Gram-negative. 12. Immediately dip the slide in and out of a beaker of clean water for 2-3 seconds. 13. Cover the smears with Safranin and let it stand for 30 seconds. 14. Rinse the smears with drops of water until the water runs clear from the colonies. 15. Let the slide air-dry. 16. Focus on the cells of circle “1” of the Gram stained slide with the low power objective and then the medium, high power and oil immersion objectives. SHS Biotechnology Revised 2015 - 2016 Page 64 17. Are the cells of circle “1” Gram-positive or Gram-negative? Record the results in Table 1. 18. Describe the cell morphology of the cells in circle “1.” Record your results in Table 1. 19. Repeat this examination for bacteria 2 and 3. Part C - Additional Identification Characteristics 1. Determine whether the unknowns are obligate aerobes, obligate anaerobes, or facultative. Record this information in Table 1 under “O2 Use.” a. In order to examine the effect of oxygen, bacterial cells were stabbed into a test tube containing thioglycollate, which causes oxygen to be present only near the surface and the remainder of the tube is anaerobic. b. Obligate aerobes will grow near the surface of the agar in the tube. c. Obligate anaerobes will grow below the surface of the agar in the tube. d. Facultative organisms will grow throughout the tube. 2. Determine whether the unknown bacterial species can ferment glucose and, if so, whether gas is produced during fermentation. Record this information in Table 1 under “Glucose Use.” a. Bacteria were inoculated in a medium containing glucose as the carbon source and phenol red as a pH indicator. b. Phenol red is red at neutral pH and yellow at pH <6.8. A change in color from red to yellow indicates the formation of acid and thus fermentation of glucose. Additionally, an inverted tube is placed in the inoculated test tube; if gas is produced during fermentation, it will collect in the inverted test tube. 3. Determine whether the unknown organisms are capable of producing endospores. Record this information in Table 1 under “Endospore.” a. In order to test whether a sample of inoculum contains endospores, the sample is exposed to high heat (80°C) for 10 minutes. This treatment would kill all cells but endospores would survive and could grow when incubated at an appropriate temperature. b. If endospores are present, the resultant inoculum will give rise to a turbid culture (indicating bacterial growth); if there are no endospores present, no growth is observed. 4. Determine whether the bacteria are motile. Record this information in Table 1 under “Motile.” a. One way the motility of cells can be investigated is by injecting a stab of bacteria into a test tube containing a low concentration of agar (so the bacteria, if motile, can swim) and phenol red. Phenol red is yellow when oxidized by growing bacteria. Thus, a yellow color throughout the agar test tube indicates motility and SHS Biotechnology Revised 2015 - 2016 Page 65 a yellow line in the middle of the test tube indicates that the bacteria under investigation are non-motile. Part D - Characteristics of known bacteria A. Colony morphology 1. Examine the Known (and labeled) bacteria samples that your teacher has provided and record your observations in Table 2. Use the terms in Figure 3 to describe colony morphology. B. Gram stain, cell morphology, and cell size 2. Examine the prepared slide dos the known bacterial species provided and note the cell morphology and Gram stain reaction of each. Record your observations in Table 2. C. Additional Identification Characteristics 3. For each of the known bacteria, determine the requirements for oxygen in growth, their ability to ferment glucose, their ability to form endospores, and their motility. Record this information in Table 2. D. Identification 4. With the characteristics of the known samples identified, take a look back at your three unknowns (1, 2 & 3) and write the name of the sample in the last row of table 1. E. Cleanup 5. Discard the PCA plates with your streaked culture into the autoclave bag at the back of the lab. 6. Using a piece of lens paper, gently wipe the oil off the objective lens of the microscope and off of the prepared slides used. 7. Place glass slide with your bacterial smears into the container with the cleaning solution at the front of the lab. 8. Return the inoculating loop, the forceps, grease pencil, and Gram stain set up to the appropriate place in lab (the same place you got them from). 9. Empty and rinse the beakers of water you used for the Gram stain and return to your lab bench. SHS Biotechnology Revised 2015 - 2016 Page 66 Results: Table 1 Completed: Colony Morphology Table 1: Unknown Bacterial Samples Characteristic: Bacteria Bacteria 1 2 Gram Stain Cell Morphology Size (mm) Shape Elevation Margin Edge Color Surface Texture Oxygen Use? Glucose Use? Endospores Formed? Motile? NAME: SHS Biotechnology Revised 2015 - 2016 Bacteria 3 _____/10 Page 67 Table 2: Known Bacterial Samples Name: 1 2 Characteristic : Gram Stain Table 2 Completed: 3 4 5 _____/10 Cell Morphology Colony Morphology Size (mm) Shape Elevation Margin Edge Color Surface Texture Oxygen Use? Glucose Use? Endospores Formed? Motile? SHS Biotechnology Revised 2015 - 2016 Page 68 Hypothesis Correct? Based on the data collected, was your hypothesis correct? Give 2 pieces of data that clarify your decision: ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ CHOOSE 1 based on whether your hypothesis is correct: OR If your hypothesis was wrong, what was wrong with it (detail – minimum 2 sentences)? If your hypothesis was right, generate a new lab question that could be asked to create a new lab experiment to follow up on what you learned in this experiment. ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ CHOOSE 1: How could the experiment be repeated to make it more accurate (describe 2 ways to improve the experiment)? OR Were there any mistakes made in the process of following the procedures that affected the outcome? If so, how could these mistakes be corrected for (be detailed about the mistakes you made and how you would correct for them)? ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ How can the data we collected and the concepts we learned in this lab be applied to life? In other words, what is the practical application of the concepts from this lab (minimum 3 sentences)? ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ SHS Biotechnology Revised 2015 - 2016 Uses the words “Yes” or “No” to start. First sentence does not state clearly, “My hypothesis was correct/not correct.” 2 items of detailed data measured in lab (numbers, observations) were not provided to support decision. Data used is not explained. Student does not make a choice. A hypothesis cannot be both right and wrong. The decision about hypothesis correctness does not match the data collected in lab. _____/10 Hypothesis related choice 1: Wrong question answered. Does not tell what revisions to hypothesis would make it correct. Evaluates lab procedures instead of the hypothesis. Does not tell why the hypothesis was wrong. Not 2 sentences long. _____/10 Hypothesis related choice 2: Wrong question answered. Basically a repeat of the question from this lab. Not in the form of a question. Little thought or creativity shown. Not testable in lab. Question is too general. Neither question is answered. _____/10 Page 69 In our lab, we had you compare several known bacteria against a few unknown bacteria. Do you think this is a better way to identify bacteria in a laboratory situation, rather than just relying on a scientist’s knowledge and experience (or their memory) of the characteristics of certain species? Explain why and justify your answer. ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ Why is it important to have more than one type of characteristic to observe in order to identify a species of bacteria? In other words, why isn’t colony morphology alone enough to identify which species of bacteria you are looking at, but instead you have to use microscopic examination, staining and types or fermentation to determine the what type of bacteria you are examining? ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ Choice 1: Not enough detail. 2 ways not given. Answers were complaints not solutions. Saying, “Follow all instructions” is not a solution. 1 sentence is not enough. Don’t blame others, find solutions. This student does not understand what was happening in lab. _____/10 Choice 2: Doesn’t tell what you did wrong & how to fix it next time. Should be longer than 1 sentence. Saying, “We didn’t make any mistakes,” means you have to answer the other question. Saying, “Follow all the steps better,” does not work for an answer. This student does not understand what was happening in the lab. _____/10 Application: Answers should be specific with at least one example/career/life experience listed. Not 3 sentences long. Student does not understand how to apply this to life or what was done in lab. Don’t give opinions (i.e this is dumb). Don’t retell what we did in lab. _____/10 Specific Question #1: ______15 Specific Question #2: _____/15 Total Score: ________/125 SHS Biotechnology Revised 2015 - 2016 Page 70 Supplemental Notes: Antibiotic Resistance in Bacteria I. II. III. IV. V. VI. VII. VIII. IX. X. Fleming - discovers penicillin, the first antibiotic. a. What is an antibiotic? A natural substance secreted by one microorganism that will kill or inhibit growth and reproduction of other microorganisms. b. Antibiotics Affect s: i. Bacterial processes of growth and Reproduction. ii. Inhibit cell wall formation iii. Disrupting cell membrane function iv. Preventing DNA replication v. Inhibiting Transcription vi. Inhibiting Translation or Protein Synthesis Penicillin - Targets the cell walls of bacteria , inhibits the cross-linking, of the peptidoglycan chains. a. Halts cell wall expansion, causing cell lysis. Ampicillin - Member of penicillin family, Gram +/-. a. Inhibits cell wall synthesis. Antibiotics that Affect the Cell Wall : a. Fosfomycin, Bacitracin, Penicillin, amoxicillin, ampicillin, cephalosporin, Vancomycin Antibiotics that Affect the Plasma Membrane a. Polymyxin B Antibiotics that Affect the DNA Replication a. Sulfonamides, Prontosil Tetracyclines a. Broad spectrum b. Inhibit bacterial protein synthesis c. Blocks tRNA attachment to ribosome Chloramphenicol a. Similar to tetracycline b. Synthetic c. Inhibits protein synthesis by attaching to the ribosome d. Interferes with peptide bond formation between amino acids Antibiotics That Affect Protein Synthesis a. Mupirocin, Streptomycin, Linezolid, Tetracycline Kirby-Bauer Test - Tests the Antibiotic Susceptibility of Bacteria SHS Biotechnology Revised 2015 - 2016 Page 71 XI. a. A qualitative test of inhibition How do bacteria become resistant? a. Antibacterial lotions, Soaps and Sprays leave only resistant bacteria. b. Decreased transport of the antibiotic into the cell membrane. c. Production of enzymes that destroy the inhibitory capacity of the antibiotic d. Modification of the antibiotic binding site so that the drug no longer binds to the target. e. Production of alternate molecules that can replace those disrupted by the antibiotic. f. Plasmids i. CONJUGATION - THE PROCESS OF EXCHANGING GENETIC MATERIAL THROUGH CELL-TO-CELL CONTACT ii. TRANSFORMATION - THE PROCESS BY WHICH BACTERIA CELLS PICK UP AND INCORPORATE DNA FROM DEAD BACTERIA CELLS. THEY TAKE UP DNA FROM DEAD CELLS OF THE SAME OR A CLOSELY RELATED SPECIES iii. Transposons - Pieces of DNA That move around the chromosome. 1. Vertical transfer - Within species 2. Horizontal transfer - Between species SHS Biotechnology Revised 2015 - 2016 Page 72 Supplemental Process Lab: Antibiotic Resistance Background: The first antibiotic, penicillin was discovered by Alexander Fleming in 1929. A fungal colony had grown as a contaminant on an agar plate he streaked with the bacterium Staphylococcus aureus. Fleming noted that the bacterial colonies around the fungus were transparent, because their cells were lysing. A natural substance secreted by the Penicillium notatum killed the Staphylococcus aureus. Since then, many antibiotics have been discovered. An antibiotic is a natural substance secreted by one microorganism that will kill or inhibit growth and reproduction of other microorganisms. The antibiotics that will be tested in this experiment are ampicillin, penicillin, and tetracycline or chloramphenicol. Ampicillin is one of the most widely prescribed antibiotics. Ampicillin, a member of the penicillin family, is able to penetrate both Gram-positive and Gram-negative bacteria. It inhibits the third and final stage of bacterial cell wall synthesis, which ultimately leads to cell lysis. Ampicillin was approved by the FDA in 1963. Tetracyclines have the broadest spectrum of antimicrobial activity. Tetracyclines inhibit bacterial protein synthesis by blocking the attachment of the transfer RNA-amino acid to the ribosome. More precisely they are inhibitors of the codon/anti-codon interaction. Chloramphenicol is also a broad spectrum antibiotic that possesses activity similar to the tetracylines. It is prepared synthetically. It inhibits protein synthesis by attaching to the ribosome and interferes with the formation of peptide bonds between amino acids. A you can see, antibiotics affect bacterial processes of growth and reproduction by: inhibiting cell wall formation, disrupting cell membrane function, preventing DNA replication, inhibiting transcription, and inhibiting translation or protein synthesis. Materials: 1 pre-poured LB agar plate Cultured Bacteria (3 different species) Plating wand Your choice of 4 of the following antibiotics: Antibiotics and their Functions Antibiotic Producer Organism Activity Site or Mode of Action Streptomyces kanamyceticus Kanamycin Broad Spectrum Protein synthesis Tetracycline Streptomyces rimosus Broad spectrum Protein synthesis Triple Sulfa Synthetic Broad Spectrum Replication (Sufonamides) Nitrofurantoin Synthetic Broad Spectrum DNA Nalidixic Acid Synthetic Broad Spectrun Cell Division Streptomycin Streptomyces griseus Gram-negative bacteria Protein synthesis Streptomyces niveus Novobiocin Gram-positive (a few gram- Protein Synthesis negative) Chloramphenicol synthetic Broad spectrum Protein synthesis Erythromycin Streptomyces erythreus Gram-positive bacteria Protein synthesis [Type text] Page 73 Procedure: 1. Divide your LB plate into 4 sections on the bottom of the plate. Label the four sections with the antibiotics to be tested (for example: ampicillin, penicillin, or tetracycline), also label your name, date and period, and type of bacteria (each group will work with one of 3 different bacteria) on the bottom of the dish. 2. On the plate, spread out 100 µl of the E. coli liquid culture as demonstrated by your teacher. 3. Obtain the pretreated discs containing the four antibiotics you selected. 4. On each section of the plates put the correct antibiotic treated disc. Use flamed forceps to put the disks right on top of the spread out bacterial culture. Use slight pressure to ensure that the disc is adhered to the surface of the agar. 5. Place the plates at 37 C overnight. 6. After incubation, measure the diameter of the clear zone around the disc (including the disc itself) and record it in mm in data table #1. 7. Using the chart below, determine if the bacteria is resistant, susceptible or becoming susceptible (intermediate) to the particular antibiotic and record it in data table #1. 8. Work with several other groups and collect data for the 9 different antibiotics and the 3 different species. Antibiotic Kanamycin Tetracycline Triple Sulfa (Sufonamides) Nitrofurantoin Nalidixic Acid Streptomycin Novobiocin Chloramphenicol Erythromycin SHS Biotechnology Resistant < or = mm 13 14 10 13 14 14 14 12 13 Revised 2015 - 2016 Antibiotics and their Functions Intermediate mm 14 - 17 15 - 18 11 - 15 14 - 17 15 - 18 15 - 20 15 - 17 13 - 17 14 - 22 Susceptible = or > mm 18 19 16 18 19 21 18 18 23 Page 74 RESULTS: Table 1 Antibiotic Species #1: (name) Species #2: Width (mm) Width (mm) Susceptible? (R,I,S) Species #3: Susceptible? (R,I,S) Width (mm) Susceptible? (R,I,S) Kanamycin Tetracycline Triple Sulfa (Sufonamides) Nitrofurantoin Nalidixic Acid Streptomycin Novobiocin Chloramphenicol Erythromycin The tests we performed in lab are called Kirby-Bauer Tests. How are these tests useful to doctors and physicians who are working to help those who are suffering from illness? __________________________________________________________________________________________________________________________ __________________________________________________________________________________________________________________________ __________________________________________________________________________________________________________________________ __________________________________________________________________________________________________________________________ __________________________________________________________________________________________________________________________ __________________________________________________________________________________________________________________________ __________________________________________________________________________________________________________________________ __________________________________________________________________________________________________________________________ ___________________________________________________________________________________________________________________ SHS Biotechnology Revised 2015 - 2016 Page 75 Biochemistry Unit Vocabulary Define the following terms using your textbook. There will be a quiz on these words on ______________________. Anion Cation Lipids Molar Mass Molarity Mole Molecular Biology Pellet Protein Solute Solvent Supernatant Catalyst SHS Biotechnology Revised 2015 - 2016 Page 76 [Type text] Page 77 Important Chemical Formulas: SHS Biotechnology 78 Chemistry Review Notes I. II. Atom: Smallest particle of an element that has the characteristics of that element a. Basic Unit of matter composed of: i. Protons: + charge, 1AMU ii. Neutron: No charge, 1AMU iii. Electrons: - Charge, no mass b. Atomic Number: Number of protons in the nucleus. i. All atoms are electrically neutral therefore number of electrons equals the number of protons. c. Atomic Mass: The sum of the protons and neutrons the nucleus. i. Atomic mass = Proton + Neutrons ii. # neutrons = Atomic mass - Atomic Number Forming Ions = Loss or gain of Electrons SHS Biotechnology the in 79 Process Lab: Kool-Aid Chemistry. (Mixing Solutions) PURPOSE: 1. Calculate the amount of solute needed to make a specific concentration of solution and then make the solution. 2. Calculate the amount needed of concentrate to dilute a concentrated solution to obtain a new molarity. MATERIALS: Balance Kool-aid (Solute) (Gram formula Mass = 40) 8M concentrated juice solution paper cups PROTOCOL: Part 1: 1. 2. 3. 4. 5. 6. You will make 4 different solutions of Kool-aid. FOR ALL EQUATIONS: Molar Mass of 1 MOLE KOOL-AID = 40 GRAMS. The first solution to make is: 100 mL of a 2M Kool-aid solution. I will walk you through the creation of the first solution: a. Step 1: Calculate the amount in grams of solute needed. Example of calculations is shown below. Follow these steps for the preparation of the other solutions. i. Remember, 40 grams Kool-Aid = 1 mole. ii. Formula: Molarity = Moles / Liter. iii. First, calculate the amount (in grams) of solute (Kool-aid) needed. iv. Convert mL to L 100 mL = 0. 1 Liters. v. 2 Moles/Liter 2 Moles/0.1 Liters = 0.2 moles of Kool-aid. vi. Now convert moles to grams: Grams = Moles x Molar Mass .2 Moles X 40 (Molar Mass) = 8 grams b. Step 2: Make the solution i. Using a clean weighing boat, place it on the balance and zero the reading. ii. Slowly pour Kool-aid ( your choice of flavor) into the weighing boat until the measurement reads 8g. iii. Take a clean flask and pour the 8 g of solute (Kool-aid) into it. iv. You will now add water until the final volume of the solution (the meniscus of the solution) reaches the 100 mL mark on the flask. v. Stir the flask on the hotplate (no heat) until the solute is completely mixed in. vi. Pour into your paper cups (split with your partner). Taste the solution. vii. The manufacturer of Kool-aid recommends this concentration when mixing the product. This will serve as the control or basis for comparison. For your second solution: Make 50 mL of a 4M Kool-aid solution: a. Show all calculations needed to make the solution in Table 1 in the results section of your lab book. b. Place the amount of solute needed into a 100 mL beaker and make the solution. c. Make a taste comparison to solution #1 by determining if it is sweeter or less sweet than solution #1. Now make a 3rd solution: Make 60 mL of a 2M solution. Show all calculations. Now make a 4th solution: Make 70 mL of a I M Kool-aid solution. Show all calculations. [Type text] Page 80 7. 8. Before Tasting your solutions, load 2.5 ml of each solution into a cuvette and record the absorbance of each in Table 1 along with your taste comparisons and calculations. Part 2: DILUTIONS - There are some very concentrated solutions of juice on the front counter. You need to pick your favorite flavor. All of these solutions are 8M. a. You need to use one of these solutions to mix a solution that is 2M. b. You will need to add water to dilute the solution to obtain the correct molarity. c. Use the formula: MIVI = M2V2 d. 1. Obtain 20 ml of the 8M juice solution. e. Calculate the final volume needed to dilute the solution to a 2M solution. f. Show all equations and calculations in Table 2 in the results section. g. Using the calculations, dilute the concentrated juice. Taste, and evaluated the original concentration and the diluted concentration. h. Before Tasting your solutions, load 2.5 ml of each solution into a cuvette and record the absorbance of each in Table 2 along with your taste comparisons and calculations. RESULTS: Table 1: Solutions Calculations Solution Calculations Final Amounts Needed #1 #2 #3 #4 Table 2: Solution Dilutions Solution Calculations 8M 2M SHS Biotechnology Final Amounts Needed Flavor Comparison (Sweeter / Not as Sweet) XXXXXXXXX CONTROL XXXXXXXXXXX Flavor Comparison (Sweeter / Not as Sweet) XXXXXXXXXX CONTROL XXXXXXXXXX 81 Spectrophotometer Absorbance Reading Spectrophotometer Absorbance Reading Notes: Acids and Bases I. II. III. IV. V. VI. VII. VIII. Acids taste sour, are corrosive to metals, change litmus (a dye extracted from lichens) red, and become less acidic when mixed with bases. Bases feel slippery, change litmus blue, and become less basic when mixed with acids. ACIDS AND BASES ARE EVERYWHERE a. Every liquid you see will probably have either acidic or basic traits. One exception might be distilled water. The pH scale is used to measure how acidic or basic a liquid is. a. Although there may be many types of ions in a solution, pH focuses on concentrations of hydrogen ions (H+) and hydroxide ions (OH-). The scale goes from 0 through 14. b. Distilled water is 7 (right in the middle). c. Acids are found between 0 and 7. d. Bases are from 7 to 14. Most of the liquids you find every day have a pH near 7. They are either a little below or a little above that mark. Definitions TO KNOW a. Acid: A solution that has an excess of H+ ions. b. Base: A solution that has an excess of OH- ions. c. Aqueous: A solution that is mainly water. AQUA means water. d. Strong Acid: An acid that has a very low pH (0-3). e. Strong Base: A base that has a very high pH (11-14). f. Weak Acid: An acid that only partially ionizes (molecule breaks apart) in an aqueous solution. Usually a pH close to 7 (4-6). g. Weak Base: A base that only partially ionizes in an aqueous solution. Usually a pH close to 7 (8-10). h. Neutral: A solution that has a pH of 7. Neither acidic nor basic. WHAT REALLY HAPPENS a. What really happens in those solutions? It gets a little tricky here. We'll give you the straight answer. Acids are compounds that break into hydrogen (H+) ions in an aqueous solution. Bases are compounds that break up into hydroxide (OH-) ions in an aqueous solution. b. That pH scale is actually a measure of the number of H+ ions in a solution. If there are a lot of H+ ions, the pH is very low. If there are a lot of OH- ions, that means the number of H+ ions is very low, so the pH is high. BUFFERS AND pH OF BUFFERS a. A buffer solution is one in which the pH of the solution is "resistant" to small additions of either a strong acid or strong base. A buffer is made with a weak acid and a soluble salt containing the conjugate base of the weak acid or a weak base and a soluble salt containing the conjugate acid of the weak base. Some examples of buffer material pairs are: b. acetic acid and sodium acetate, c. phosphoric acid and potassium phosphate SHS Biotechnology 82 Process Lab: Creating a pH Scale PURPOSE: Students will be able to determine pH using various means, will have a basic understanding of the pH scale, will be able to group solutions as acids and bases, will be able determine characteristics of acids and bases and will have a basic understanding of buffers. MATERIALS: pH meter baking soda ammonia (liquefied) grapefruit juice pH 7.0 buffer solution vinegar lemon juice Mylanta tomato juice Hydrion papers distilled water cranberry juice aspirin (liquefied) Coke Universal Indicator tap water Milk of Magnesia Maalox Diet Coke orange juice bleach Dr. Pepper grape juice Sprite apple juice liquid detergent Tums pineapple juice PROTOCOL: 1. 2. 3. 4. 5. 6. 7. Your team will determine the pH of each of the samples listed above. Your data will be used to make a pH chart with the solutions arranged from lowest pH to the highest pH. Begin by calibrating your pH meter. Immerse the probe end of your meter in the pH 7.0 buffer solution and check the reading on the meter. If the reading is 7.0, then there is no need to calibrate, and you can move on to step 3 below. If it is anything other than 7.0, pleas follow the steps below starting with a. a. Using the plasic screwdriver provided, insert the flat end into the calibration screw on the back of the meter. b. Slowly turn the screw until the meter reads 7.0. c. Keep the meter immersed in the buffer solution while this process is being followed. There are 6 stations set up with samples of each of the solutions listed above. For each sample, you must measure the pH in 2 ways. a. First, dip a piece of hydrion paper into the sample and compare the color of the paper to the chart provided to determine the pH of the solution. Record this in the “pH Paper” section of Table #1. b. Second, immerse the pH meter into the sample and read the pH indicated on the meter after the read-out on the meter stabilizes. Record this in the “pH Meter” section of Table #1. Between samples, when using the pH meter, make sure to rinse the pH meter off in running water. After you have finished using the meter at the end of the lab, rinse it well and replace the protective cap. Average the meter reading and the paper reading together in Table #1. Collect the average pH readings form all of the other groups in class today and average them together in Table #2. Based on the class averages you figured create a pH scale on the blank arrow provided that reflects where each of the substances tested would sit based on its acidity or alkalinity. RESULTS: SHS Biotechnology 83 TABLE #1: Solution pH Meter pH Paper AVG. Solution pH Meter a. orange juice m. Dr. Pepper b. apple juice n. aspirin c. baking soda o. Tums d. vinegar p. Mylanta e. distilled water q. Maalox f. tap water r. grape juice g. bleach s. Pineapple Juice h. liquid detergent t. Grapefruit juice i. ammonia u. tomato juice j. lemon juice v. Coke k. cranberry juice W. Diet Coke l. Milk of Magnesia x. Sprite SHS Biotechnology 84 pH Paper AVG. TABLE #2: Solution Grp. 1 Grp. 2 Grp. 3 Grp. 4 Grp. 5 Grp. 6 Grp. 7 Grp. 8 a. orange juice b. apple juice c. baking soda d. vinegar e. distilled water f. tap water g. bleach h. liquid detergent i. ammonia j. lemon juice k. cranberry juice l. Milk of Magnesia m. Dr. Pepper n. aspirin o. Tums p. Mylanta q. Maalox r. grape juice s. Pineapple Juice SHS Biotechnology 85 Grp. 9 Grp. 10 Grp. 11 Grp. 12 Avg. t. Grapefruit juice u. tomato juice v. Coke W. Diet Coke x. Sprite SHS Biotechnology 86 Chemical Solutions – Process Lab PURPOSE: To practice preparing solutions of specific molarities and concentrations, to measure the pH of various solutions and to allow the teacher to evaluate the student’s ability to prepare solutions. PROTOCOL: 1) Part One: Measuring pH a. Use the pH paper and pH meter to measure the following solutions. When using the pH meter, rinse the electrode with distilled water between each solution. b. Record your data in Table 1. c. Average the reading for the pH meter and the pH paper, then record this in Table 1. 2) Part Two: Making a defined molar solution and adjusting pH a. EDTA - Ethylenediaminetetraacetic Acid is commonly used as a component in making gel electrophoresis buffer. b. Calculate the grams of EDTA needed to make 10 L of 0.5 M solution of EDTA. c. Once you’ve done the calculations, make the solution. d. Add calculated amount of EDTA to 10 ml of water. (EDTA will not go into solution unless pH is 8 or greater.) e. Test the pH using the meter. f. Does the pH need to be adjusted up or down? Would you add acid or base to make this adjustment? g. Before you go on check with the teacher to make sure you chose the right one. (Please keep this information quiet, we want everyone to think this out.) h. Adjust pH by slowly adding (drops) either acid or base. i. Use your pH meter to monitor the change in pH. j. Record the required information for this lab in Table 2. 3) Part Three: Calculate the amount of dextrose (C6H12O6) needed to make 500 ml of a 2 M solution. a. Where can you find the molar mass (or formula weight) of dextrose? b. Calculate the gram formula mass (formula weight) of dextrose. c. How many grams of dextrose would you need to make the 2 M solution? d. What would you use as your solvent? e. Record this information in Table 3. 4) Part Four: Describe how you would prepare 100 g of an aqueous 0.5% phenolphthalein solution. (Phenolphthalein is a solid.) a. Perform the calculation and list the amount of grams of phenolphthalein you would use. b. Once you have done the calculation, what would you actually use to make the solution? We are not actually making this solution. SHS Biotechnology 87 c. Record your answers in Table 4. 5) Part Five: Describe how you would prepare 100 ml of a solution that is 22% acetic acid by volume. a. How many milliliters of acetic acid would you need? b. In what order do you combine acid and water? c. Mix the solution and record your answers in Table 5. RESULTS: Table 1: pH Solution Name Vinegar Distilled water Baking Soda HCL NaOH Indicator Paper Results Table 2: Making a solution, Adjusting pH. Calculations: Which substance do you need to add to get to a pH of 8 or higher? pH meter results Average pH Amount of EDTA: pH Reading when EDTA dissolves: Table 3: Calculating Molar Mass to Make a Solution Question: Answer: Where can you find the molar mass (or formula weight) of dextrose? Calculate the gram formula mass (formula weight) of dextrose. How many grams of dextrose would you need to make the 2 M solution? What would you use as your solvent? SHS Biotechnology 88 Initial pH: Table 4: Calculating the Percent Concentration of Solution A Question: Answer: Perform the calculation and list the amount of grams of phenolphthalein you would use. Once you have done the calculation, what would you actually use to make the solution? Table 5: Calculating the Percent Concentration of Solution B Question: Answer: How many milliliters of acetic acid would you need? In what order do you combine acid and water? SHS Biotechnology 89 DNA Basics Unit Vocabulary Define the following terms using your textbook. There will be a quiz on these words on ______________________. Allele Autosomal Dominant Autosomal Recessive Base pair Cell Chromosome Complementary DNA (cDNA) Complementary Nucleotides Dominant Double Helix Genome Genomics Hydrogen bond Molecular Genetics Nitrogenous Base Nucleic Acid Nucleotides Polymer [Type text] Page 90 X-Ray Crystallography SHS Biotechnology 91 History of DNA Notes: I. II. III. IV. V. VI. VII. VIII. IX. X. XI. XII. XIII. XIV. XV. XVI. XVII. XVIII. 1863 – Mendel discovers that traits are transmitted from parents to progeny by discrete, independent units, later called genes. 1868 - Friedrich Meischer – this Swiss biologist carried out the first carefully thought out chemical studies on the nuclei of cells. a. Using the nuclei of pus cells obtained from discarded surgical bandages, Miescher detected a phosphoruscontaining substance that he named nuclein. b. He showed that nuclein consists of an acidic portion, which we know today as DNA, and a basic protein portion now recognized as histones, a class of proteins responsible for the packaging of DNA. 1869 - Miescher discovers DNA in the sperm of trout. 1879 - Fleming discovers chromatin, later came to be called chromosomes. 1884 – Hertwig: Nuclein is component of chromosomes, Molecular carrier of heredity information. 1912 - Physicist Sir William Henry Bragg, and his son, Sir William Lawrence Bragg, discover that they can deduce the atomic structure of crystals from their X-ray diffraction patterns. 1914 - Bacteria are used to treat sewage for the first time in Manchester, England. 1928 - Griffiths Experiment a. Was one of the first experiments suggesting that bacteria are capable of transferring genetic information through a process known as transformation. b. Also in 1928 - Fleming discovers penicillin, the first antibiotic. 1929 - Levene discovers deoxyribose. 1944 - Avery’s Experiment: a. Oswald Avery demonstrated that the substance responsible for the transformation of harmless bacteria into disease-causing monsters was DNA. 1947 - McClintock discovers transposable elements, or "jumping genes," in corn. 1950 - Erwin Chargaff Differentiates Purines: Adenine, Guanine from Pyrimidines: Thymine, Cytosine a. Also, Rosalind Franklin begins using x-ray diffraction. 1952 - The Hershey-Chase experiments: a. Were a series of experiments conducted by Alfred Hershey and Martha Chase, confirming that DNA was the genetic material. While DNA had been well known to biologists since 1869, most assumed at the time that proteins carried the information for inheritance. 1953 - Watson and Crick, Rosalind Franklin, Maurice Wilkins a. When Crick and Watson joined each other they began to make models of DNA based on previous research and they used Franklins x-ray diffraction technology. 1958 - Rosalind Franklin found out that she had ovarian cancer and continued to work. She went through three operations and experimental chemotherapy. She worked up until a few weeks before her death at the age of 37. 1958 - The Meselson - Stahl Experiment a. Revealed that DNA Replication is Semiconservative 1962 – Watson, Crick and Wilkinson get Noble Prize in Physiology for their discovery. 1968 - Chargaff’s conclusions: a. A + G = T + C; SHS Biotechnology 92 XIX. XX. XXI. XXII. XXIII. XXIV. XXV. XXVI. XXVII. XXVIII. XXIX. b. A = T; G = C 1961 - Sidney Brenner and Francis Crick establish that groups of three nucleotide bases, or codons, are used to specify individual amino acids. 1966 The genetic code is deciphered when biochemical analysis reveals which codons determine which amino acids. 1970 - Hamilton Smith, at Johns Hopkins Medical School, isolates the first restriction enzyme, an enzyme that cuts DNA at a very specific nucleotide sequence. a. Also in 1970 - Specific restriction nucleases are identified, opening the way for gene cloning. b. First complete synthesis of a gene 1972 Stanley Cohen and Herbert Boyer combine their efforts to create recombinant DNA. This technology will be the beginning of the biotechnology industry. 1973 - Stanley Cohen and Herbert Boyer perfect genetic engineering techniques to cut and paste DNA (using restriction enzymes and ligases) and reproduce the new DNA in bacteria. Somatostatin, which regulates human growth hormones, is the first human protein made using recombinant technology. 1976 Herbert Boyer cofounds Genentech, the first firm founded in the United States to apply recombinant DNA technology 1981 - Scientists at Ohio University produce the first transgenic animals by transferring genes from other animals into mice. a. Chinese scientist become the first to clone a fish - a golden carp 1983 - The Polymerase Chain Reaction (PCR) technique is conceived. PCR, which uses heat and enzymes to make unlimited copies of genes and gene fragments, later becomes a major tool in biotech research and product development worldwide. 1984 - The DNA fingerprinting technique is developed. a. The first genetically engineered vaccine is developed. 1985 - Genetic fingerprinting enters the courtroom. a. Genetically engineered plants resistant to insects, viruses and bacteria are field tested for the first time. b. The NIH approves guidelines for performing experiments in gene therapy on humans. 1997 - Scottish scientists report cloning a sheep, named Dolly, using DNA from adult sheep cells. SHS Biotechnology 93 Notes: Nucleic Acids I. II. III. DNA : Deoxyribonucleic acid a. Bacterial DNA - Single circular chromosome + Small plasmids. i. 500000 to 5000000 Base Pairs (bp) long. b. Eukaryotic DNA – Chromosomes / Chromatin. DNA Structure a. A molecule of DNA is made up of millions of tiny subunits called Nucleotides. i. Each nucleotide consists of: 1. Phosphate group. 2. Pentose sugar (deoxyribose). Carbons in this are numbered 1 – 5. 3. (1) Nitrogeneous base: Adenine, Thymine, Guanine or Cytosine b. DNA nucleotides are linked together by covalent bonds to form a single strand. c. Two nucleotides can be linked together by a covalent bond between the Deoxyribose of one molecule and the Phosphate of the next molecule. d. More nucleotides are added in the same manner until they form a chain of molecules. e. DNA consists of two nucleotide chains that are arranged into a ladder-like structure called a Double Helix. DNA double helix is formed using complementary base pairing & hydrogen bonds. a. Two strands of these nucleotide chains are bonded together into a single molecule that has a particular shape, called a DOUBLE HELIX. b. Hydrogen bonds link these two strands together, forming between the bases of each pair of nucleotides. i. Each base will only bond with one other specific base. 1. Adenine will ONLY forms hydrogen bonds with Thymine. 2. Cytosine ONLY forms hydrogen bonds with Guanine. ii. Purines: 2 of the bases are made of 2 rings. These are Adenine & Guanine. iii. Pyramidines: Composed of one ring. Cytosine & Thymine. c. Bonding: i. The Purine Adenine will ONLY bond with the Pyramidine Thyamine. Each of these require 2 hydrogen bonds to hold them together. ii. The Purine Guanine wil ONLY bond with the pyramidine Cytosine because they both require 3 hydrogen bonds to hold them together. SHS Biotechnology 94 IV. V. VI. iii. Because of this complementary base pairing, the order of the bases in one strand determines the order of the bases in the other strand. d. The phosphate and sugar form the backbone of the DNA molecule, whereas the bases form the “rungs”. The 2 strands are “Antiparallel” – One strand has a glucose whose 3’ is closest to the bottom of the chain and the 5’ carbon is closest to the top, the other strand runs in the opposite direction (5’ to 3’). DNA Structure Key Ideas. If a gene is a section of DNA that codes for a specific protein: a. Each unique gene has a unique sequence of bases. b. This unique sequence of bases will code for the production of a unique protein. c. It is these unique proteins and combination of proteins that give us a unique phenotype. Short History of DNA: (The long history will come later.) a. 1929 - Levene Discovers deoxyribose b. 1950 - Erwin Chargaff discovers the Bases. Purines: A, G. Pyrimidines: T, C. c. 1953 - Nature publishes James Watson's and Francis Crick's manuscript describing the double helical structure of DNA, which marks the beginning of the modern era of genetics. SHS Biotechnology 95 Notes: DNA Extraction I. II. III. IV. V. First, you need to find something that contains DNA. For this example, we will use Split Peas… Step 1: Blender a. Put the following into a blender: 1/2 cup of split peas (100ml), 1/8 teaspoon table salt (less than 1ml), 1 cup cold water (200ml). Blend on high for 15 seconds. b. The blender separates the pea cells from each other, so you now have a really thin pea-cell soup. c. Salty water helps the DNA precipitate (solidify and appear) when alcohol is added. d. Cooling slows down enzymatic reactions. This protects DNA from enzymes that can destroy it. i. Why would a cell contain enzymes that destroy DNA? These enzymes are present in the cell cytoplasm to destroy the DNA of viruses that may enter our cells and make us sick. A cell’s DNA is usually protected from such enzymes (called DNases) by the nuclear membrane, but adding detergent destroys that membrane. e. Pour your thin pea-cell soup through a strainer into another container (like a measuring cup). f. Add 2 tablespoons liquid detergent (about 30ml) and swirl to mix. g. Let the mixture sit for 5-10 minutes. h. Pour the mixture into test tubes or other small glass containers, each about 1/3 full. WHY DID I ADD DETERGENT TO MY PEA SOUP? a. Each cell is surrounded by a sack (the cell membrane). DNA is found inside a second sack (the nucleus) within each cell. To see the DNA, we have to break open these two sacks. We do this with detergent. b. Why detergent? How does detergent work? Think about why you use soap to wash dishes or your hands. To remove grease and dirt, right? c. Soap molecules and grease molecules are made of two parts: Both soap and grease molecules organize themselves in bubbles (spheres) with their heads outside to face the water and their tails inside to hide from the water. When soap comes close to grease, their similar structures cause them to combine, forming a greasy soapy ball. d. A cell's membranes have two layers of lipid (fat) molecules with proteins going through them. When detergent comes close to the cell, it captures the lipids and proteins. Step 2a: Heat (Optional). This step does not show up in all protocols, but it can be affective in helping to disrupt the plasma and nuclear membranes even further by denaturing the proteins found suspended in them. a. Place the tube in a hot water bath (55 – 60 degrees C) for 10 minutes. Step 3: Enzymes a. Add a pinch of enzymes to each test tube and stir gently. Be careful! If you stir too hard, you'll break up the DNA, making it harder to see. b. At this point the tube is sometimes returned to the hot water bath for another 20 minutes, but not always. c. Use meat tenderizer for enzymes. If you can't find tenderizer, try using pineapple juice or contact lens cleaning solution. d. WHY DID I ADD MEAT TENDERIZER? In this experiment, meat tenderizer acts as an enzyme to cut proteins just like a pair of scissors. i. The DNA in the nucleus of the cell is molded, folded, and protected by proteins. The meat tenderizer cuts the proteins away from the DNA. ii. What enzyme is found in meat tenderizer? The two most common enzymes used in meat tenderizer are Bromelain and Papain. These two enzymes are extracted from pineapple and SHS Biotechnology 96 VI. VII. VIII. papaya, respectively. They are both proteases, meaning they break apart proteins. Enzymatic cleaning solutions for contact lenses also contain proteases to remove protein build-up. These proteases include Subtilisin A (extracted from a bacteria) and Pancreatin (extracted from the pancreas gland of a hog). Step 3a: Centrifuge for 7 minutes. Carefully remove the tube from the centrifuge and note the two phases: upper layer: supernatant, DNA is dissolved here. This needs to be saved. Lower layer: pellet, cell debris, discard it. Step 3b: Decant the supernatant, avoiding any of the pellet. Step 4: Alcohol Separation a. Tilt your test tube and slowly pour either rubbing alcohol (70-95% isopropyl) or ethyl alcohol into the tube down the side so that it forms a layer on top of the pea mixture. Pour until you have about the same amount of alcohol in the tube as pea mixture. b. Alcohol is less dense than water, so it floats on top. Look for clumps of white stringy stuff where the water and alcohol layers meet. c. I don’t think I’m seeing DNA. What should I be looking for? Look closely. Your DNA may be lingering between the two layers. Try to help the DNA rise into the alcohol layer. Dip a wooden stick into the supernatant and slowly pull upward into the alcohol layer. Also, look very closely at the alcohol layer for tiny bubbles. Even if your yield of DNA is low, clumps of DNA may be loosely attached to the bubbles. d. DNA precipitates when in the presence of alcohol, (meaning it doesn’t dissolve in alcohol). This causes the DNA to clump together when there is a lot of it. For example, each cell in the human body contains 46 chromosomes (or 46 DNA molecules). If you lined up those DNA molecules end to end, a single cell would contain six feet of DNA! If the human body is made of about 100 trillion cells, each of which contains six feet of DNA, our bodies contain more than a billion miles of DNA! SHS Biotechnology 97 DNA Extraction from Cheek Cells – Pre-Lab Reading DNA isolation is a process of purification of DNA from sample using a combination of physical and chemical methods. The first isolation of DNA was done in 1869 by Friedrich Miescher. Currently it is a routine procedure in molecular biology or forensic analyses. Outline for a basic DNA Extraction 1. Break open (lyse) the cells or virus containing the DNA of interest- This is often done by the addition of a detergent such as SDS is often necessary to remove lipid membranes. Sometimes heat is required. 2. DNA associated proteins, as well as other cellular proteins, may be degraded with the addition of a protease. Precipitation of the protein is aided by the addition of a salt such as ammonium or sodium acetate. 3. DNA is kept in solution by the high level of salt in the solution. 4. DNA is then precipitated by mixing with cold ethanol or isopropanol and possibly centrifuging. The DNA is insoluble in the alcohol and will come out of solution, and the alcohol serves as a wash to remove the salt previously added. 5. Wash the resultant DNA pellet with cold alcohol again and centrifuge for retrieval of the pellet. 6. After pouring the alcohol off the pellet and drying, the DNA can be re-suspended in a buffer such as Tris or TE. 7. Presence of DNA can be confirmed by electrophoresing on an agarose gel containing ethidium bromide, or another fluorescent dye that reacts with the DNA, and checking under UV light. What is it used for? Extraction of DNA is often an early step in many diagnostic processes used to detect bacteria and viruses in the environment as well as diagnosing disease and genetic disorders. These techniques include but are not limited to Fluorescence In Situ Hybridization (FISH): FISH is a molecular technique that is used, among other things, to identify and enumerate specific bacterial groups. Terminal Restriction Fragment Length Polymorphism (T-RFLP): T-RFLP is used to identify, characterize, and quantify spatial and temporal patterns in marine bacterioplankton communities. Sequencing: Portions of, or whole genomes may be sequenced as well as extra chromosomal elements for comparison with existing sequence in the public data base. SHS Biotechnology 98 DNA Extraction from Cheek Cells – Process Lab PURPOSE: 1. Learn the correct basic process for extracting DNA from cells. PROTOCOL: 1) Collecting cells: Swish the saline solution around in your mouth for 30 seconds and split it into a 50 ml conical tube. 2) Get 3 microcentrifuge (1.5 mL) tubes. 3) Fill these as full as possible with liquid from your conical tube. 4) Cap the tubes and place them into the centrifuge. 5) Make sure that the centrifuge is balanced before starting the spin. Spin at 4000 rpm for 5 min. 6) After the spin, pour off the Supernatant (the saline solution) without disturbing the cell pellet. 7) Refill the microcentrifuge tubes with liquid from the conical tubes (add this on top of the pellet so the centrifuge will add more cells to the pellet. 8) Repeat these steps until the pellet at the bottom of each of these tubes is about the size of a match head. 9) Lysing the cells: Add 750 uL of lysis buffer to each of the tubes and mix by vortexing for several seconds. 10) Removing the proteins and fats from the solution: Place the tubes into the 65° C water bath for 10 minutes. 11) Add 250 uL of 5 M NaCl to the solution to each tube and mix gently by repeatedly inverting the solution for 1 minute. 12) Place the tubes in the centrifuge. Make sure to balance it before starting the spin. Spin at 4000 rpm for 10 minutes. 13) Harvesting the DNA: Carefully transfer the supernatant (the solution above the pellet) from all 3 microfuge tubes into a single glass test tube. BE CAREFUL not to bring along any of the precipitated proteins/fats to the new tube. 14) Add 1000 ul of ice cold 95% alcohol to the tube. Do not mix the alcohol and the solution in the tube. 15) Allow the alcohol to form a layer on top of the other solution. Watch for small white threads to appear in the alcohol. 16) Using a suction pipette, suction out the small threads and some of the alcohol and transfer them to a new microcentrifuge tube. 17) Label the tube with your name and leave it in the rack at the front of the lab. RESULTS: 1) Write a brief description of the DNA that you saw in your tube: ___________________________ _____________________________________________________________________________________ _____________________________________________________________________________________ ______________________________________________________ SHS Biotechnology 99 Completion Verification: 20 points Strawberry DNA Extraction – Full Blown Lab Grading Rubric – For Teacher’s Use I will deduct points for each of the following: Name: ___________________________ Due Date: ____/____/____ This is to be written in complete sentences with correct grammar, spelling and punctuation. /5 Question: Can DNA be isolated from plant cells? Hypothesis (written in “If… then…because” format): _____________________________________________________________________________________ _____________________________________________________________________________________ Independent Variable: _______________________________________________ Dependent Variable: ________________________________________________ After you complete the lab, tell what affect the INDEPENDENT VARIABLE had on the DEPENDENT VARIABLE (describe the relationship between the two variables in a minimum of 3 sentences)? ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ MATERIALS: Mild Shampoo or Dish Soap (SDS) Strawberries Microcentrifuge tubes Micropipettes Centrifuge Tris EDTA solution /10 Independent Variable: No variable identified. Incorrect Independent variable. /5 Dependent Variable: No variable identified. Incorrect Dependent variable. Link between variables: Answer not 3 sentences. Question is not answered. Link between variables is not explained clearly. The link described between variables is incorrect. Description of what was done in lab is not an answer to this question. /10 The strawberries have been pureed in a blender with 1M salt water to disrupt the cell wall of each cell. This is a mechanical breakdown of the cellular structures. SHS Biotechnology Hypothesis: Does not use proper format – If/Then/Because. Hypothesis does not completely answer lab question. Hypothesis is poorly stated. Does not clarify what the student is thinking. /5 Papain (Meat tenderizer) Ice Beaker Cheese cloth Ice cold alcohol Water bath 55oC PROTOCOL: 1. Overall: Not written in complete sentences. Does not use correct grammar spelling & punctuation. 100 2. 3. 4. 5. The puree was then strained to separate the cell walls from the released cells. Pour 15 mL of the pureed strawberries into you test tube. Add a large “pinch” of meat tenderizer into the puree mixture. Add 15 mL of the dish soap into the tube and mix well with the strawberry puree (by inverting the tube repeatedly. DO NOT SHAKE). 6. Put test tube into the hot water bath at 55oC – 60oC for 10 minutes. Heating helps to maximize the reaction to make sure that DNA is released. The temperature is critical to this step. 7. The ruptured cell mixture is now called the lysate. 8. Use the transfer pipet to drip ice cold alcohol slowly down the sides of the tube, while holding the tube at approximately an angle of 45o. Try to make a clear and undisturbed layer of alcohol to float on the lysate. The line between the two layers is called the interface. 9. At the interface, you will see the DNA precipitate out of solution and float to the top. You may spool the DNA around a glass rod, or suction it out of the tube with a dropper. 10. Collect the DNA which is in the upper layer of the alcohol with the transfer pipet. Transfer the DNA and some of the alcohol to a microcentrifuge tube. 11. Label your tube with your initials and leave it with your teacher for grading. Table 1 Completed: _____/10 Hypothesis Correct? RESULTS: Did you obtain DNA? Describe how the DNA looked: Uses the words “Yes” or “No” to start. First sentence does not state clearly, “My hypothesis was correct/not correct.” 2 items of detailed data measured in lab (numbers, observations) were not provided to support decision. Data used is not explained. Student does not make a choice. A hypothesis cannot be both right and wrong. The decision about hypothesis correctness does not match the data collected in lab. _____/10 Based on the data collected, was your hypothesis correct? Give 2 pieces of data that clarify your decision: ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ SHS Biotechnology 101 Hypothesis related choice 1: CHOOSE 1 based on whether your hypothesis is correct: If your hypothesis was wrong, what was wrong with it (detail – minimum 2 sentences)? OR If your hypothesis was right, generate a new lab question that could be asked to create a new lab experiment to follow up on what you learned in this experiment. ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ CHOOSE 1: How could the experiment be repeated to make it more accurate (describe 2 ways to improve the experiment)? OR Were there any mistakes made in the process of following the procedures that affected the outcome? If so, how could these mistakes be corrected for (be detailed about the mistakes you made and how you would correct for them)? ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ How can the data we collected and the concepts we learned in this lab be applied to life? In other words, what is the practical application of the concepts from this lab (minimum 3 sentences)? ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ Wrong question answered. Does not tell what revisions to hypothesis would make it correct. Evaluates lab procedures instead of the hypothesis. Does not tell why the hypothesis was wrong. Not 2 sentences long. _____/10 Hypothesis related choice 2: Wrong question answered. Basically a repeat of the question from this lab. Not in the form of a question. Little thought or creativity shown. Not testable in lab. Question is too general. Neither question is answered. _____/10 Choice 1: Not enough detail. 2 ways not given. Answers were complaints not solutions. Saying, “Follow all instructions” is not a solution. 1 sentence is not enough. Don’t blame others, find solutions. This student does not understand what was happening in lab. _____/10 Choice 2: Doesn’t tell what you did wrong & how to fix it next time. Should be longer than 1 sentence. Saying, “We didn’t make any mistakes,” means you have to answer the other question. Saying, “Follow all the steps better,” does not work for an answer. This student does not understand what was happening in the lab. _____/10 SHS Biotechnology 102 What is the purpose of learning how to extract DNA from a cell? Why is that always going to be a first step in any kind of genetic testing that is done on a sample? ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ Describe the functions of each of the following in the process of DNA extraction: Blender: _____________________________________________________________________ ____________________________________________________________________________ Meat Tenderizer: ______________________________________________________________ ____________________________________________________________________________ Dish Soap: __________________________________________________________________ ____________________________________________________________________________ Ice Cold Alcohol:_______________________________________________________________ ____________________________________________________________________________ Glass Rod: ___________________________________________________________________ ____________________________________________________________________________ SHS Biotechnology 103 Application: Answers should be specific with at least one example/career/life experience listed. Not 3 sentences long. Student does not understand how to apply this to life or what was done in lab. Don’t give opinions (i.e this is dumb). Don’t retell what we did in lab. _____/10 Specific Question #1: ______15 Specific Question #2: _____/20 Total Score: ________/120 DNA Profiling Unit Vocabulary Define the following terms using your textbook. There will be a quiz on these words on ______________________. Agarose Gel Electrophoresis Bioinformatics Blunt End DNA Fingerprint Electrophoresis Enzyme Ethidium Bromide Human Genome Project Lysis Mitochondria Mitochondrial DNA (mtDNA) Nuclease Palindromic Sequence Polymerase Protease Restriction Enzyme [Type text] Page 104 RFLP Sticky End SHS Biotechnology Southern Hybridization 105 DNA Profiling (Fingerprinting) Notes I. II. III. IV. What is DNA Profiling? A technique used by scientists to distinguish between individuals of the same species using only samples of their DNA. Who Invented it? The process of DNA fingerprinting was invented by Alec Jeffreys at the University of Leicester in 1985. a. He was knighted in 1994. Stages of DNA Profiling a. Stage 1: Cells are broken down to release DNA. b. Step 2: The DNA is cut into fragments using restriction enzymes. i. Each restriction enzyme cuts DNA at a specific base sequence. ii. The sections of DNA that are cut out are called restriction fragments. iii. This yields thousands of restriction fragments of all different sizes because the base sequences being cut may be far apart (long fragment) or close together (short fragment). c. Stage 3: Fragments are separated on the basis of size using a process called gel electrophoresis. i. DNA fragments are injected into wells and an electric current is applied along the gel. ii. DNA is negatively charged so it is attracted to the positive end of the gel. iii. The shorter DNA fragments move faster than the longer fragments. DNA is separated on basis of size. iv. A radioactive material is added which combines with the DNA fragments to produce a fluorescent image. v. A photographic copy of the DNA bands is obtained. d. Stage 4: The pattern of fragment distribution is then analysed. Uses of DNA Profiling: a. DNA profiling is used to solve crimes and medical problems. The DNA profile of each individual is highly specific. The chances of two people having exactly the same DNA profile is 30,000 million to 1 (except for identical twins). b. Biological materials used for DNA profiling: Blood, Hair, Saliva, Semen, Body tissue cells. c. DNA profiles can be used to determine whether a particular person is the parent of a child. SHS Biotechnology 106 V. Restriction Enzymes - are DNA-cutting enzymes found in bacteria (and harvested from them for use). Because they cut within the molecule, they are often called restriction endonucleases. a. In order to be able to sequence DNA, it is first necessary to cut it into smaller fragments. The rarer the site it recognizes, the smaller the number of pieces produced by a given restriction endonuclease. b. A restriction enzyme recognizes and cuts DNA only at a particular sequence of nucleotides. For example, HaeIII cuts DNA wherever it encounters the following sequence: 5‘ GGCC 3'. The cut is made between the adjacent G and C. c. This particular sequence occurs at 11 places in the circular DNA molecule of the virus φX174. Thus treatment of this DNA with the enzyme produces 11 fragments, each with a precise length and nucleotide sequence. d. HaeIII and AluI cut straight across the double helix producing "blunt" ends. e. However, many restriction enzymes cut in an offset fashion. The ends of the cut have an overhanging piece of single-stranded DNA. These are called "sticky ends" because they are able to form base pairs with any DNA molecule that contains the complementary sticky end. f. Mixed together, these molecules can join with each other by the base pairing between their sticky ends. The union can be made permanent by another enzyme, DNA ligase, that forms covalent bonds along the backbone of each strand. The result is a molecule of recombinant DNA (rDNA). g. Restriction Enzymes evolved by bacteria to protect against viral DNA infection. i. 3,000+ known SHS Biotechnology 107 Making Electrophoresis Gels – Process Lab PURPOSE: To learn the process of making and pouring agarose gels for use in electrophoresis and learn the basics of how gel electrophoresis works. PROTOCOL: Part 1 ABCDEFG- Most gels should be between 0.5% and 1 % agarose. One gel requires about 40 mL of solution when mixed. Determine the amount of agarose powder needed to make 2 gels. Measure out 80 mL of TAE buffer and pour it in a 250 mL Erlenmeyer flask or beaker. Begin heating the TAE on a hot plate set at 350ᴼ C. Start stirring the TAE as it heats at a rate of about 150 RPM. When steam begins to come from the mouth of the flask (not boiling), add in the correct amount of agarose powder (determined in step C). Keep stirring. H- Watch the solution constantly at this point. Continue heating the solution until it clarifies. I- Prepare the gel mold by placing the rubber dams on each end of the gel tray. J- Insert the comb into the first slot on the in the gel tray. K- Pour the liquid gel into the mold you created. If you have any bubbles in the gel, try to push them to the side with a yellow pipet tip. Let the gel harden for at least 30 minutes. SHS Biotechnology 108 Case of the Invisible Bands – Pre-Lab Reading Gel electrophoresis is used to separate macromolecules like DNA, RNA and proteins. DNA fragments are separated according to their size. Proteins can be separated according to their size and their charge (different proteins have different charges). How are DNA fragments separated using gel electrophoresis? A solution of DNA molecules is placed in a gel. Because each DNA molecule is negatively charged, it can be pulled through the gel by an electric field. Small DNA molecules move more quickly through the gel than larger DNA molecules. The result is a series of ‘bands’, with each band containing DNA molecules of a particular size. The bands furthest from the start of the gel contain the smallest fragments of DNA. The bands closest to the start of the gel contain the largest DNA fragments. When is gel electrophoresis used to separate DNA fragments? Gel electrophoresis can be used for a range of purposes, for example: To get a DNA fingerprint for forensic purposes To get a DNA fingerprint for paternity testing To get a DNA fingerprint so that you can look for evolutionary relationships among organisms To check a PCR reaction. To test for genes associated with a particular disease. When is gel electrophoresis used to separate proteins? Thanks to TV shows like CSI, many people are familiar with the use of gel electrophoresis to separate macromolecules like DNA. However, gel electrophoresis can also be used to separate out proteins. Different proteins have different sizes, mainly due to the number of amino acid building blocks in their structure. Chemical modifications attached to the protein also affect its size. Different proteins also have different charges. This can result from both the types of amino acid used to construct them, as well as the types of modifications attached to them. Different types of electrophoresis gels are used to provide different types of information. The type of gel you choose therefore depends on the type of question you are asking. Typically, gels made from polyacrylamide are used to separate proteins on the basis their different sizes. Usually, the proteins are first treated with heat and a chemical called SDS in order to unravel the protein. SDS is a detergent that gives all the proteins the same overall negative charge so that when an electric current is applied to the gel, separation is only due to the size of the protein. This technique is called SDS-PAGE (SDS-Polyacrylamide gel electrophoresis). Small protein molecules move more quickly through the gel than larger proteins, resulting in a series of ‘bands’. Each band contains a protein of a particular size. These can be compared with standards of known sizes. SHS Biotechnology 109 Process Lab: Case of the Invisible Bands Purpose: To experience what DNA Fingerprinting would look like if we used ethidium bromide to visualize DNA instead of blue positively charged stain. Materials: Standard Dye Fragments Electrophoresis Apparatus 5 Invisible samples Agarose Gel TAE Buffer UV Visualizer Protocol: 1. Place a premade agarose gel into the gel tray (mold), and place the tray into the electrophoresis chamber (wells should face the black electrode). 2. Load 20 µL of each dye sample into separate wells in the following order: a. Well #1 = Standard Dye Fragments Sample (A) b. Well #2 = Invisible Sample #1 (B) c. Well #3 = Invisible Sample #2 (C) d. Well #4 = Invisible Sample #3 (D) e. Well #5 = Invisible Sample #4 (E) f. Well #6 = Invisible Sample #5 (G) 3. After the samples are loaded, place the lid on the chamber and plug it into the power supply. Make sure to match up the black electrode to the black electrode, and red to red. 4. Set the voltage on the power supply to 100 V. Set the time to 45 minutes. 5. After electrophoresis is completed, remove the gel from the chamber and place the tray in a UV visualizing chamber or under a UV light. 6. Record the initial color of the dyes that you added to the gel in your results section by coloring that color into the wells on the diagram. 7. Record the band pattern in each lane on the diagram of the gel. 8. Determine which of the samples match by looking at the diagram you made. Results: Draw the Band patterns (in Color) that you see created on your gel. Color in what colors were placed in the wells at the start of the experiment as well: SHS Biotechnology 110 Gel Candy Electrophoresis – Pre-Lab Reading FOOD COLORING The color of food has always been an integral aspect of our culture. The early Romans believed that people not only eat with their palate, but also “eat with their eyes.” For centuries, humans have used dyes from natural ingredients to add color to food, drink, clothing and other products. For example, saffron, paprika and other spices were used to provide food yellow appearance. Companies have long been introducing color additives to a variety of products, including candies, shampoos, perfumes, drinks, etc. Color additives are used in food for several reasons. Manufacturers add colors to food to offset color loss due to product exposure to various environmental conditions, such as light, air, and moisture. Additionally, companies often add dyes to food products like beverages, jellies, pudding and condiments to make them look more attractive to consumers. Table 1 illustrates the seven commonly used food dyes in the United States. Of these dyes, Blue 1 and Red 40 are the most common blue and red dyes while Green 3 and Blue 2 are rarely used. Food containing these approved dyes are called, “For Coloring Food” and have the abbreviation “FCF” preceding their names. Companies are required to list food dyes in their list of ingredients. As the use continues to grow, concerns regarding the addition of food colors to food products also emerge. Commonly used food dyes such as yellow 5 and red 40 are believe to pose several health concerns in children, including hyperactivity and allergic reactions. The Food and Drug Administration (FDA) is responsible for regulating color additives used in a variety of products in the United States. Color additives allowed for use in foods are classified as “certifiable” or “exempt from certification”. Certifiable color additives are man-made which are tested by the manufacturers and the FDA to assures the quality and safety of the color additives. On the other hand, color additives that are “exempt from certification” are those that derived from natural sources such as vegetables, fruits, and minerals. AGAROSE GEL ELECTROPHORESIS Agarose gel electrophoresis is widely used to separate molecules based upon charge, size and shape. It is particularly useful in separating charged biomolecules such as DNA, RNA and proteins. Agarose gel electrophoresis possesses great resolving power, yet is relatively simple and straightforward to perform. The gel is made by dissolving agarose powder in the electrophoresis buffer. The solution is boiled to dissolve the agarose and then cooled to approximately 55º C and poured into a gel tray where it solidifies. The tray is submerged in a buffer-filled electrophoresis apparatus, which contains electrodes. Samples are prepared for electrophoresis by mixing them with glycerol or sucrose to give the mixture higher density. This makes the samples denser than the electrophoresis buffer. These samples can then be loaded with a micropipette or transfer pipet into wells that were created in the gel by a template during casting. The dense samples sink through the buffer and remain in the wells. A direct current power supply is connected to the electrophoresis apparatus and current is applied. Charged molecules in the sample enter the gel matrix. Molecules having SHS Biotechnology 111 a net negative charge migrate towards the positive electrode (anode) while net positively charged molecules migrate towards the negative electrode (cathode). Within a range, the higher the applied voltage, the faster the samples migrate. The buffer serves as a conductor of electricity and to control the pH. The pH is important to the charge and stability of biological molecules. Agarose is a polysaccharide derived from agar. In this experiment, UltraSpec-Agarose™, a mixture of agarose and hydrocolloids which renders the gel to be both clear and resilient, is used. At first glance, an agarose gel appears to be a solid at room temperature. However, on the molecular level, the gel contains microscopic pores which act as a molecular sieve, allowing the different molecules to pass through. Food dyes are composed of ions. When these charged ions are subjected to an electric field, the molecules will migrate toward the electrode of opposite charge. Positively charged molecules will migrate toward the negative electrode, while those with a negative charge will move toward the positive electrode. Small dye fragments move through these holes easily, but large dye fragments have a more difficult time squeezing through the tunnels. Factors such as charge, size and shape, together with buffer conditions, gel concentrations and voltage, affects the mobility of molecules in gels. Because molecules with dissimilar sizes travel at different speeds, they become separated and form discrete “bands” within the gel. After the current is stopped, the bands can be visualized. In this experiment, students will extract several different dyes from food source. The dyes will then be analyzed using agarose gel electrophoresis and their rate of migration will be observed and measured. SHS Biotechnology 112 Candy Electrophoresis – Process Lab Purpose: To practice the processes involved in gel electrophoresis and learn to analyze the results of the process. Materials: Dye Extraction Buffer Standard Dye Marker Micropipettes Beakers Electrophoresis apparatus D.C. power supply Candy (suggestions: M&M, Skittles, jelly bean, gum ball) Agarose gel 0.5 ml centrifuge tubes Distilled water Electrophoresis buffer 1.5 ml centrifuge tubes Protocol: 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. Label five microcentrifuge tubes with your initials and the colors of the M&Ms you will be investigating. Label the provided cup with your initials. ADD one M&M to the cup Add 250 μl of Dye Extraction Buffer to the beaker containing the candy. Swirl the M&M gently in the Dye Extraction Buffer to dissolve the color coating until the white layer of the candy is exposed. Remove the candy from the cup. Transfer the dissolved color solution into the appropriately labeled microcentrifuge tube. Rinse the cup. Repeat steps 2-6 with the remaining 4 candies. Place the tubes on lab bench. Place an agarose gel (on the mold tray) into electrophoresis chamber. Completely COVER the gel with 1X TAE buffer. Load 10 μl of the Standard Dye Marker into lane one of the gel. Add 20 μl of each extracted candy dye into each of the remaining lanes. Place the cover on the chamber. Check that the gel is properly oriented. Remember, the DYE samples will migrate toward the positive (red) electrode. Connect the leads to the power source. Run the electrophoresis process for 30 minutes at 150 volts. After electrophoresis is complete, turn the power off and remove the gel and casting tray from the electrophoresis chamber. Remember, as the length of a biomolecule increases, the distance to which the molecule can migrate decreases because large molecules cannot pass through the channels in the gel with ease. Therefore, the migration rate is inversely proportional to the length of the molecules— more specifically, to the log10 of molecule's size. Measure the distance traveled by each Standard Dye Molecule from the lower edge of the sample well to the lower end of each band. Record the distance in centimeters (to the nearest millimeter) in Table 1. Measure and record the migration distances of each of the fragments in the unknown samples in the same way you measured the standard bands. Plot the data from the standard molecule on the graph. After all the points have been plotted, use a ruler or a straight edge to connect the points in the plot and carry the line off of the top and side of the graph. SHS Biotechnology 113 25. Determine the length of each unknown fragment. Locate the migration distance of the unknown dye on the x-axis of your graph. Trace a vertical line extending from that point until it intersects the line of your standard curve (the one you plotted in the steps above). 26. From that point, trace a second line, this time horizontally, toward the y-axis. The value at which this line intersects the y-axis represents the approximate size of the dye in base pairs. Make note of this in Table 1. 27. Repeat for each fragment in your unknown sample. Standard Dye Colors Band Color cm bp Blue 5000 Red 3000 Purple 1000 Orange 500 Candy 1: ____________ Band Color SHS Biotechnology cm bp Candy 2: ____________ Band Color cm bp Candy 3: ____________ Band Color cm 114 bp Candy 4: ____________ Band Color cm bp Candy 5: ____________ Band Color cm bp SHS Biotechnology 115 Study Questions 1. What is the purpose of gel electrophoresis? 2. What do you think are some factors that determine the distance molecules move though the gel? Give an example. 3. If you were pouring a gel to run DNA samples, where would you place the comb? Explain. 4. Based on the results on you gel, which dye was the smallest dye molecule? Explain how you know. 5. Based on the results on you gel, which dye was the largest dye molecule? Explain how you know. 6. When determining the sizes of Dye fragments, which axis on the graph is used to plot the migration distances of the known and unknown fragments? 7. Which axis is used to plot the sizes of the known and unknown fragments? SHS Biotechnology 116 Paternity Testing Lab – Pre-Lab Reading Did I find my real dad? Is that really my son? Questions like these used to be very hard to answer. In the past, people used a blood test. This might rule out that you were the father but couldn't prove that you were. Nowadays, DNA technology is used to figure out who is the father of a child. DNA paternity testing makes it possible to determine a child's biological father to a very high degree of certainty. Everyone, except identical twins, has a unique set of DNA. When someone says that everyone's DNA is unique, what they mean is that occasionally one of these letters is different for different people. On average, two people at random have a different base every thousand bases or so. This is where the statistic that says that everyone's DNA is 99.9% the same comes from. Since you get half your DNA from your father and half from your mother, your DNA is more than 99.9% the same as your parents. Your DNA is also more similar to that of your grandparents or cousins than to that of a random stranger. Paternity tests use this greater similarity to figure out who the parents are. So how do you figure out someone's DNA is more similar to another's? There are lots of ways but we'll focus on the simplest, DNA restriction analysis or DNA fingerprinting. DNA fingerprinting uses special proteins called restriction enzymes. Restriction enzymes cut DNA but only at a certain combination of A, G, T, and C. Different restriction enzymes cut DNA at different places -- each has a unique sequence it recognizes. For example, the restriction enzyme EcoRI cuts DNA at the sequence GAATTC and will cut only at that sequence. It will not, for example, cut at GACTTC. What DNA fingerprinting does is it looks for differences in the DNA that change where these restriction enzymes can cut DNA. The pattern of DNA fragments is then compared and if the child's DNA looks like a combination of the two parents' DNA, then the child is theirs. Let's look at an example of how this might be done. Suppose we have three people: Bob, Larry, and Mary. If we take the same stretch of DNA from the three of them, small differences might mean that EcoRI will cut them differently (Figure 1). In Bob, the sequence GAATTC occurs once in this stretch of DNA. That is, in this stretch of DNA, Bob has one EcoRI site. Now suppose Mary has no EcoRI sites and Larry has two EcoRI sites in this stretch of DNA. You can see that EcoRI will cut this stretch of Bob's DNA into two fragments, Larry's into three fragments, and Mary's will not cut. When we cut the DNA with EcoRI and separate the cut fragments on an agarose gel, the gel might look something like in Figure 2. In an agarose gel, smaller fragments run faster so you get separation based on size -- the bigger fragments are near the top, the smaller are near the bottom. Now, suppose Mary has a child and she wants to determine which of two men, Bob or Larry, is the biological father of her child. She consults a paternity testing expert. The expert collects a certain stretch of DNA from Mary, Bob, Larry, and the child, and cuts the DNA with EcoRI. When the expert separates the cut DNA fragments on an agarose gel, the pattern looks like the one in Figure 3. The child's DNA must be a combination of Mary's DNA plus one of the men's DNA. The agarose gel indicates that the child's DNA is a combination of Mary's DNA (top band) plus Larry's DNA (bottom three bands). Thus, Larry is the biological father of the child. What happens if Larry and Bob have the identical sequence in this stretch of DNA? The answer is that in real life this would not happen because more than one stretch of DNA would be compared from the possible parents in the test. SHS Biotechnology 117 Process Lab: Paternity Testing Purpose: To practice identifying the parents of an individual by comparing the DNA profile of the offspring to that of the mother and possible fathers. Materials: Agarose Gel TAE Buffer DNA Stain Staining Tray DNA Samples (Mother, Child, Possible Father 1, Possible Father 2) Protocol: 1. Place the agarose gel into the tray, and place the tray into the electrophoresis chamber. Make sure that the wells are facing the black electrode. 2. Fill the electrophoresis chamber with about 350 mL of TAE buffer, it should completely cover the gel. 3. Load 10 µL of each DNA sample into its own well. The samples should added in the following order: a. Well #1 = Mother b. Well #2 = Child c. Well #3 = Alleged Father #1 d. Well #4 = Alleged Father #2 4. Place the lid on the chamber, matching black electrode to black and red to red. 5. Plug the chamber into the power supply, making sure to match the colors. 6. Set the power supply to 150 V, and run the electrophoresis for 45 minutes. 7. After electrophoresis, DNA fragments are visualized by staining the gel with a blue DNA stain. The blue dye molecules are positively charged and have a high affinity for the DNA. These blue dye molecules strongly bind to the DNA fragments and allow DNA to become visible. 8. Sketch the gel in your results section. Make sure to note what samples were loaded into which wells. Draw the band patterns as they appear on the gel. 9. Based on what you observe in your gel, which Alleged Father is the actual father of the child? Result: Draw the band pattern created on the gel below. Label what DNA was placed in each well. Based on the results obtained, who is the father of the child (who shares 50% of the child’s DNA besides the mother)? _________________________________________ ________________________________________ SHS Biotechnology 118 Restriction Digest Analysis of Lambda DNA – Pre-Lab Reading Restriction enzyme, also called restriction endonucleases, are proteins produced by bacteria that cleaves (or cut) DNA at specific sites along the molecule. In the bacterial cell, restriction enzymes cleave foreign DNA, thus eliminating infecting organisms like viruses. Restriction enzymes can be isolated from bacterial cells and used in the laboratory to manipulate fragments of DNA, such as those that contain genes; for this reason they are indispensible tools of recombinant DNA technology, genetic engineering and forensic science. A bacterium uses a restriction enzyme to defend against bacterial viruses called bacteriophages, or phages. When a phage infects a bacterium, it inserts its DNA into the bacterial cell so that it might be replicated. The restriction enzyme prevents replication of the phage DNA by cutting it into many pieces. Restriction enzymes were named for their ability to restrict, or limit, the number of strains of bacteriophage that can infect a bacterium. Each restriction enzyme recognizes a short, specific sequence of nucleotide bases. These regions are called recognition sequences and are randomly distributed throughout the DNA. Different bacterial species make restriction enzymes that recognize different nucleotide sequences. When a restriction endonuclease recognizes a sequence, it snips through the DNA molecule by splitting of a chemical bond between adjacent nucleotides. Bacteria prevent their own DNA from being degraded in this manner by disguising their recognition sequences. Enzymes called methylases add methyl groups (—CH3) to adenine or cytosine bases within the recognition sequence, which is thus modified and protected from the endonuclease. The restriction enzyme and its corresponding methylase constitute the restriction-modification system of a bacterial species. There are three classes of restriction enzymes, designated types I, II, and III. Types I and III enzymes are similar in that both restriction and methylase activities are carried out by one large enzyme complex, in contrast to the type II system, in which the restriction enzyme is independent of its methylase. Type II restriction enzymes also differ from the other two types in that they cleave DNA at specific sites within the recognition site; the others cleave DNA randomly, sometimes hundreds of bases from the recognition sequence. These are the useful enzymes in biotechnology. Restriction enzymes were discovered and characterized in the late 1960s and early 1970s by the molecular biologists Werner Arber, Hamilton O. Smith, and Daniel Nathans. The ability of these enzymes to cut DNA at precise locations enabled researchers to isolate gene-containing fragments and recombine them with other molecules of DNA—i.e., to clone genes. More than 2,500 type II restriction enzymes have been identified from a variety of bacterial species. These enzymes recognize about 200 distinct sequences, which are four to eight bases in length. The names of restriction enzymes are derived from the genus, species, and strain designations of the bacteria that produce them; for example, the enzyme EcoRI is produced by Escherichia coli strain RY13. SHS Biotechnology 119 Grading Rubric – For Teacher’s Use Restriction Digest Analysis of Lambda DNA – Full Blown Lab I will deduct points for each of the following: Name: ___________________________ Due Date: ____/____/____ This is to be written in complete sentences with correct grammar, spelling and punctuation. Question: Can DNA from the same source produce different patterns during gel electrophoresis when different restriction enzymes are used to digest the same DNA? Hypothesis (written in “If… then…because” format): _____________________________________________________________________________________ _____________________________________________________________________________________ Independent Variable: ___________________________________________________________ Dependent Variable: ____________________________________________________________ When you finish the lab, tell what affect the INDEPENDENT VARIABLE had on the DEPENDENT VARIABLE (describe the relationship between the two variables in a minimum of 3 sentences)? ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ /5 Hypothesis: Does not use proper format – If/Then/Because. Hypothesis does not completely answer lab question. Hypothesis is poorly stated. Does not clarify what the student is thinking. /10 Independent Variable: No variable identified. Incorrect Independent variable. /5 Dependent Variable: No variable identified. Incorrect Dependent variable. /5 Materials: Ice Microcentrifuge 50-100 ml beaker for ice bath Lambda HindIII DNA markers Gel staining trays Pipette tips (sterile) Overall: Not written in complete sentences. Does not use correct grammar spelling & punctuation. Distilled water TAE buffer (50x) 100 ml DNA sample loading dye Foam test tube rack Micropipettes Electrophoresis chamber Agarose DNA staining solution Restriction enzymes 8 microtubes Permanent marker Power supply Protocol: Part 1 - Restriction Digestion of Lambda DNA 1. Obtain micro test tubes that contain each of the enzyme solutions, the lambda DNA, and the restriction buffer from the front counter. Keep all the stock solutions on ice. SHS Biotechnology 120 Link between variables: Answer not 3 sentences. Question is not answered. Link between variables is not explained clearly. The link described between variables is incorrect. Description of what was done in lab is not an answer to this question. /10 2. Label four micro test tubes L, P, E, and H and place them in the micro test tube holder. a. L = Uncut lambda DNA b. P = PstI restriction digest of lambda DNA c. E = EcoRI restriction digest of lambda DNA d. H = HindIII restriction digest of lambda DNA 3. Follow the table below to determine the amounts and substances that should be added to each of the tubes: Tube Lambda Restriction PstI EcoRI HindIII DNA Buffer H 4 μl 5 μl 1 μl E 4 μl 5 μl 1 μl L 5 μl 5 μl P 4 μl 5 μl 1 μl Important Note: First add DNA, then restriction buffer, and then the enzymes to the tubes. Use a fresh pipette tip for restriction buffer and each enzyme. 4. Centrifuge all tubes in order to mix all reagents, being sure that the tubes are in a balanced arrangement in the rotor. Pulse-spin the tubes (hold the button for a few seconds). 5. Place the sample tubes in a 37°C water bath for approximately 30 minutes. Restriction enzymes work best at 37°C since they were isolated from bacteria that live inside warm-blooded animals. 6. After the incubation, place the samples in the refrigerator (4°C) until the next laboratory period. Part 2 - Agarose Gel Electrophoresis 1. Obtain your four micro test tubes L, P, E, and H and place them in the micro test tube holder at your laboratory desk. 2. Add 2.0 μl of loading dye to each of the tubes marked L, P, E, and H. Use a fresh tip with each sample to avoid contamination. 3. The DNA samples and the sample loading dye must be thoroughly mixed in each tube before placing the samples in the gel wells for electrophoresis. This is easily accomplished by buzzing the tubes again in the centrifuge. 4. Obtain an agarose gel from your teacher, or, if your teacher instructs you to do so, pour your own gel. 5. Place the casting tray, with the solidified gel in it, onto the central platform in the gel box. The wells should be at the negative (cathode) end of the box where the black electrical lead is connected. SHS Biotechnology 121 6. Pour between 250 mL – 350 mL of TAE buffer into the electrophoresis chamber. Pour in enough buffer until it just covers the wells of the gel by 1–2 mm. 7. Pipet 10 μl from each tube (M, L, P, E, and H) into separate wells in the gel chamber. Use a fresh tip for each tube. Gels are read from left to right. To keep things straight, the first sample is typically loaded in the well at the upper left-hand corner of the gel. For example: 8. Push the cover of the chamber into place, and connect electrical leads to the power supply, anode to anode (red to red) and cathode to cathode (black to black). Make sure both electrical leads are attached to the same channel of the power supply. 9. Run these at 100 V for 30–40 minutes. Shortly after the current is applied, the loading dye can be seen moving through the gel toward the positive side of the gel chamber. 10. When electrophoresis is complete, turn off the power supply, disconnect the leads from the inputs, and remove the top of gel chamber. 11. Remove the casting tray from gel chamber. The gel is very slippery. Hold the tray level. 12. Pour the excess buffer back into the original container for reuse. 13. Obtain a staining tray and mark it with a piece of tape and your initials and class period. You will stain 1 gel per tray. 14. Stain gels: Remove each gel from the gel tray and carefully slide it into the staining tray. 15. Pour enough stain into the staining tray to completely cover the gel (the gel will not be visible in the tray under the stain. 16. Stain the gels for 20 minutes. 17. Using a funnel, pour the stain into a storage bottle and save it for future use. 18. De-Staining gels: Rinse the gel and tray with warm running water. After this initial rinse, fill the tray with warm water and leave it on the top of the lab bench to de-stain until the next class period. Part 3 - Analyze Results: 19. Using a ruler, measure the distance (in mm) that each of your DNA fragments or bands traveled from the well. Measure the distance from the bottom of the well to the bottom of each DNA band and record your numbers in Table 1 in your results section. SHS Biotechnology 122 20. Since the exact size and position of the fragments in the Marker DNA are known, they can be used as standard reference points to estimate the size of unknown fragment bands. 21. Using the fragments from the HindIII lambda digest, plot the distance traveled in relationship to fragment size for each fragment in the MARKER DNA. Connect as many of the points as you can by drawing a straight line through them. This will provide a standard curve with which you will be able to determine the size of your unknown fragments from the other three samples. 22. Determine the sizes of the fragments in your uncut and digested lambda DNA samples. Your teacher will provide semi-log graph paper. Results: Table 1 Band # 1 2 3 4 5 6 Lambda/HindIII Marker HindIII Digest EcoRI Digest PstI Digest Distance (mm) Distance (mm) Distance (mm) Distance (mm) Actual Size (bp) Approx. Size (bp) Approx. Size (bp) Approx. Size (bp) 23,130 9,416 6,557 4,361 2,322 2,027 SHS Biotechnology 123 Table 1 Completed: _____/25 Semi-log Graph Completed: _____/15 Hypothesis Correct? Uses the words “Yes” or “No” to start. First sentence does not state clearly, “My hypothesis was correct/not correct.” 2 items of detailed data measured in lab (numbers, observations) were not provided to support decision. Data used is not explained. Student does not make a choice. A hypothesis cannot be both right and wrong. The decision about hypothesis correctness does not match the data collected in lab. _____/10 Based on the data collected, was your hypothesis correct? Give 2 pieces of data that clarify your decision: ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ SHS Biotechnology 124 Hypothesis related choice 1: CHOOSE 1 based on whether your hypothesis is correct: If your hypothesis was wrong, what was wrong with it (detail – minimum 2 sentences)? If your hypothesis was right, generate a new lab question that could be asked to create a new lab experiment to follow up on what you learned in this experiment. OR ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ CHOOSE: How could the experiment be repeated to make it more accurate (describe 2 ways to improve the experiment)? Were there any mistakes made in the process of following the procedures that affected the outcome? If so, how could these mistakes be corrected for (be detailed about the mistakes you made and how you would correct for them)? OR ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ How can the data we collected and the concepts we learned in this lab be applied to life? In other words, what is the practical application of the concepts from this lab (minimum 3 sentences)? ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ 1. What is the purpose of the marker DNA? Explain why it is more accurate to talk about the DNA in the gel in terms of base pairs when discussing DNA evidence in court that by simply discussing the similarity in position of DNA bands. ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ ____________________________________________________________________________________ Wrong question answered. Does not tell what revisions to hypothesis would make it correct. Evaluates lab procedures instead of the hypothesis. Does not tell why the hypothesis was wrong. Not 2 sentences long. _____/10 Hypothesis related choice 2: Wrong question answered. Basically a repeat of the question from this lab. Not in the form of a question. Little thought or creativity shown. Not testable in lab. Question is too general. Neither question is answered. _____/10 Choice 1: Not enough detail. 2 ways not given. Answers were complaints not solutions. Saying, “Follow all instructions” is not a solution. 1 sentence is not enough. Don’t blame others, find solutions. This student does not understand what was happening in lab. _____/10 Choice 2: Doesn’t tell what you did wrong & how to fix it next time. Should be longer than 1 sentence. Saying, “We didn’t make any mistakes,” means you have to answer the other question. Saying, “Follow all the steps better,” does not work for an answer. This student does not understand what was happening in the lab. _____/10 SHS Biotechnology 125 Answer each of the questions below that refer to the diagram of DNA pictured here: Application: Answers should be specific with at least one example/career/life experience listed. Not 3 sentences long. Student does not understand how to apply this to life or what was done in lab. Don’t give opinions (i.e this is dumb). Don’t retell what we did in lab. _____/10 Specific Question #1: ______15 Specific Question #2: _____/15 2a. If both enzymes were used to cut the DNA, how many fragments would be created? _____ 2b. If only enzyme A were used, how many fragments would be created? ________ 2c. If only enzyme B were used, how many fragments would be created? ________ 3. In the blank gel below, draw what the DNA profile would look like if this DNA were digested with Enzyme A, Enzyme B, and Both Enzymes together: Specific Question #3: _____/20 Total Score: ________/165 SHS Biotechnology 126 (Crime Scene) DNA Fingerprinting Using Restriction Enzymes – Pre-Lab Reading One of the most significant discoveries of molecular biology is a class of enzymes known as restriction endonucleases. These endonucleases (also known as restriction enzymes) are produced by many species of bacteria to protect themselves from invading viral DNA. Restriction enzymes act like molecular scissors, cutting double-stranded DNA at specific sequences. The utility of restriction enzymes has made molecular cloning, DNA mapping, sequencing and various genome-wide studies possible, launching the era of biotechnology. Since they were first discovered in the 1970s, over 3,000 restriction enzymes have been identified, each one given a unique acronym describing the organism from which it was first isolated. The first letter of the acronym is the first letter of the genus, the next two letters are the first two letters of the species name of the organism, and additional letters and numerals indicate specific strains and order of discovery. For example, EcoRI was the first restriction enzyme isolated from the RY13 strain of the bacterium Escherchia coli. Many restriction enzymes recognize palindromic stretches of DNA, generally 4-8 base pairs in length. The probability that a given enzyme will cut, or “digest”, a piece of DNA is directly proportional to the length of its recognition site. Statistically, an enzyme will average one cut for every 4n base pairs, where n is the length of the recognition site. For instance, an enzyme that recognizes a four base pairs long sequence (e.g., HaeIII) will cut DNA once every 256 (or 44) base pairs, while an enzyme that recognizes a six base pairs long site (e.g., EcoRI) will cut once every 4096 (or 46) base pairs. Therefore, the longer a DNA molecule is, the greater the probability is that it contains one or more restriction sites. For example, if EcoRI is used to digest human chromosomal DNA containing 3 billion base pairs and a plasmid containing 5,000 base pairs, it will cut the chromosomal DNA over 700,000 times (3 billion base pairs, cut every 4096 base pairs), but may only cut the plasmid once (5,000 base pairs, cut every 4096 base pairs). Digestion by a restriction enzyme generates DNA fragments with one of two types of DNA ends--“sticky” or “blunt”. To illustrate this, first consider the recognition site and cleavage pattern of EcoRI. EcoRI cleaves between the G and neighboring A, as indicated by the arrows in the left side of the figure. It is important to note that the positions of the cleavage are staggered, so the resulting fragments project short overhangs of single-stranded DNA with complementary sequences. Such overhangs are referred to as “sticky” ends because the single-strands can interact with—or stick to— other overhangs with a complementary sequence. Digestion of the same piece of DNA using different enzymes can produce sticky ends of different lengths and strand orientation (5’ vs. 3’). In contrast to EcoRI, HaeIII cuts both DNA strands at the same position, which generates fragments without an overhang. These so-called “blunt” ends can be joined with any other blunt end without regard for complementarity. Some restriction enzymes, such as AvaI, recognize “degenerate” sites, which contain one or more variable positions. Consequently, there are four possible sites that AvaI will recognize and cut: CCCGGG, CCCGAG, CTCGGG and CTCGAG. There are even enzymes like BglI that recognize “hyphenated” sites, which are palindromic sequences separated by a number of completely variable bases. The six G-C base pairs that BglI specifically recognizes must be separated by five base pairs of DNA; otherwise the enzyme cannot properly interact with the DNA to cleave its backbone. Because these five base pairs are not required to make up a specific sequence, BglI can recognizeand cleave up to 625 possible sequences! Depending on the distances between recognition sites, digestion of DNA by a restriction enzyme will produce DNA fragments of varying lengths. In order to analyze such a mixture of DNA fragments, scientists use a technique called agarose gel electrophoresis. AGAROSE GEL ELECTROPHORESIS SHS Biotechnology 127 Agarose gel electrophoresis separates DNA fragments according to size. First, DNA molecules are added into depressions (or “wells”) within a gel, and then an electrical current is passed through the gel. Because the sugar-phosphate backbone of DNA has a strong negative charge, the current drives the restriction fragments through the gel towards the positive electrode. At first glance, an agarose gel appears to be a solid at room temperature, but on the molecular level, the gel contains small channels through which the DNA can pass. Small DNA fragments move through these holes easily, but large DNA fragments have a more difficult time squeezing through the tunnels. Because molecules with dissimilar sizes travel at different speeds, they become separated and form discrete “bands” within the gel. After the current is stopped, the bands can be visualized using a stain that sticks to DNA. While electrophoresis is a powerful separation technique, it is not without its technical limitations. Most significantly, if two different fragments share a similar size, they will migrate together through the gel and may appear as a single band. In addition, if digestion results in a broad distribution of DNA sizes, the fragments may stain as a smear. Lastly, DNA with a streamlined secondary structure (such as supercoiled DNA) can pass through the gel more quickly than similarly sized linear DNA, which prevents an accurate comparison of size. SOUTHERN BLOT ANALYSIS RFLP analysis of genomic DNA is facilitated by Southern blot analysis. After electrophoresis, DNA fragments in the gel are denatured by soaking in an alkali solution. This causes double-stranded fragments to be converted into single-stranded form (no longer base-paired in a double helix). A replica of the electrophoretic pattern of DNA fragments in the gel is made by transferring (blotting) them to a sheet of nitrocellulose or nylon membrane. This is done by placing the membrane on the gel after electrophoresis and transferring DNA fragments to the membrane by capillary action or electrotransfer. DNA, which is not visible, becomes permanently adsorbed to the membrane, that can then be manipulated easier than gels. Analysis of the blotted DNA is done by hybridization with a labeled oligonucleotide DNA probe. The probe is a DNA fragment that contains base sequences that are complementary to the variable arrays of tandemly repeated sequences found in the human chromosomes. Probes can be labeled with reporter molecules that are used for detection. A solution containing the single-stranded probe is incubated with the membrane containing the blotted, single-stranded DNA fragments. Under the proper conditions, the probe will only base pair (hybridize) to those fragments containing the complementary sequences. The membrane is then washed to remove excess probe. Only DNA fragments that are hybridized to the probe will reveal their positions on the membrane. If the probes are isotopically labeled, the hybridized fragments will appear as discrete bands (fingerprint) on the film and are in the same relative positions as they were in the agarose gel after electrophoresis. Only specific DNA fragments of the hundreds of thousands of fragments present, will hybridize with the probe because of the selective nature of the hybridization process. In forensic analysis, DNA samples can be extracted and purified from specimens of skin, blood stains, semen, or hair roots collected at the crime scene. RFLP analyses performed on these samples is then compared to SHS Biotechnology 128 (Crime Scene) DNA Fingerprinting Using Restriction Enzymes – Process Lab Purpose: To identify the suspect present at a crime scene by digesting samples of their DNA with restriction enzymes and comparing the profiles created to the sample recovered at the crime scene, and to develop an understanding of the use of restriction enzymes as applied to RFLP-based DNA fingerprinting. Materials: Crime scene DNA sample pre-cut with Restriction Enzyme 1 (Eco RI) & a second sample pre-cut with Restriction Enzyme 2 (Hind III) Suspect #1 DNA sample Suspect #2 DNA sample Standard DNA Fragments Enzyme Reaction Buffer Restriction Enzyme 1 (Eco RI) Restriction Enzyme 2 (Hind III) Agarose Gel TAE Buffer DNA Stain Staining Tray Micropipettes Electrophoresis Apparatus Power supply Water bath (37°C or 45°C) Protocol: 1. 2. 3. 4. Label micro test tubes 1 through 4 for four restriction enzyme digestion reactions. Put your initials on the tubes. Buzz the tubes in the centrifuge to collect all the contents at the bottom of the tube. Use an automatic micropipette to dispense 10 μl of Enzyme Reaction Buffer to each of four reaction tubes labeled 1 through 4. Add DNA and enzyme to the reaction tubes as summarized in Chart 1. Chart 1: Summary of Contents in Each Tube for Restriction Digestion Reactions Sample Source Crime Scene Samples Suspect 1 Suspect 2 5. 6. 7. 8. Reaction Tube Reaction DNA 1 DNA 2 # Buffer (µL) (µL) Crime Scene DNA Sample 1 is precut with enzyme 1 (Eco RI) Crime Scene DNA Sample 2 is precut with enzyme 1 (Hind III) 1 10 µL 15 µL NONE 2 10 µL 15 µL NONE 3 10 µL NONE 15 µL 4 10 µL NONE 15 µL Enzyme 1 (Eco RI) (µL) NONE NONE 15 µL NONE 15 µL NONE Enzyme 2 (Hind III) (µL) NONE NONE NONE 15 µL NONE 15 µL Final Volume of Tube (µL) 45 µL 45 µL 40 µL 40 µL 40 µL 40 µL Use a FRESH micropipette tip for each transfer of DNA and enzyme. Cap the reaction tubes and tap gently to mix. Then buzz each tube in the centrifuge to collect contents at the bottom. Incubate reaction tubes in a 45°C waterbath for 15 minutes. After incubation, add 5 μl of 10x gel loading dye to reaction tubes 1 - 4 to stop the reactions. Cap and mix by buzzing in the centrifuge again. SHS Biotechnology 129 9. Load 40 μl of each of the DNA samples in the manner shown in the diagram: 10. Close the lid of electrophoresis chamber and connect the electrodes. Set the power source to 150 volts and run the gel for 30 min. 11. Turn off the power supply as soon as the gel has finished running. 12. Stain the gel by carefully removing the agarose gel and casting tray from the electrophoresis chamber and placing the gel in a staining tray. 13. Stain the gel. 14. Examine your gel and document the results by drawing your gel in the blank gel provided in this lab 15. Using a ruler, measure and record (in table 1) the distance from the bottom of the well to the bottom of each fragment in all lanes of your gel in mm. 1) Using table one plot the data for your markers on the semi-log graph paper provided. 2) Determine the size of the fragments from the crime scene and all of the suspects. RESULTS: Table 1 Marker Crime Scene 1 Crime Scene 1 (Eco RI) (Hind III) Suspect 1 Enzyme 1 Suspect 1 Enzyme 2 (Eco RI) Ban d# Dist. (mm) Actual Size (bp) 1 23,130 2 9,416 3 6,557 4 4,361 5 3,000 6 2,322 7 2,027 8 725 9 570 Dist. (mm) Appr. Size (bp) SHS Biotechnology Dist. (mm) Appr. Size (bp) Dist. (mm) Appr. Size (bp) Suspect 2 Enzyme 1 (Hind III) (Eco RI) Dist. (mm) Dist. (mm) 130 Appr. Size (bp) Suspect 2 Enzyme 2 (Hind III) Appr. Size (bp) Dist. (mm) Appr. Size (bp) Copy the gel band patterns the way that you see them on the gel that you created in class on the diagram below: SHS Biotechnology This is how the DNA should look if everything worked perfectly in the experiment: 131 Answer the following study questions: 1. Which suspect’s DNA matches that found at the crime scene? SHS Biotechnology 132 2. Does this automatically mean that the suspect is guilty? 3. What possible experimental problems or evidence collection problems could occur to invalidate the results? 4. If only Restriction Enzyme 1 was used, would the interpretation be the same? 5. By looking at the gel to the right, can you tell who was present at the crime scene? 6. Based on the number of millimeters that a specific fragment of DNA migrated, using the graph below, determine the size (in base pairs) of each of the DNA fragments listed below: mm 22 mm 23 mm 33 mm bp mm 40 mm 41 mm 42 mm bp mm 46 mm 48 mm 51 mm bp 7. Based on the DNA Profile to the right, what can you say definitively about who was present at the crime scene? Discuss who was present, and who contributed what type of evidence to the scene: _______________________________________________________________________ _______________________________________________________________________ ________________________________________________________ SHS Biotechnology 133 DNA Replication & PCR Unit Vocabulary Define the following terms using your textbook. There will be a quiz on these words on ______________________. Anneal Clone Cloning Cystic Fibrosis Denature Dideoxynucleotide DNA Sequencing Genetic Mapping Insertion Mutation Ligase Ligate Mutagen Mutation PCR Point Mutation Primer Reading Frame Semiconservative Replication Taq Polymerase Template [Type text] Page 134 e. Notes: Replication of DNA I. II. III. IV. V. 1953 - Watson and Crick’s paper makes some implications about how DNA copies itself, but never comes right out and says how it happens (they didn’t really know for sure HOW it replicated). But they did suggest some things that some properties of DNA that lend itself to being copied. Nature of the Genetic Material. a. Property 1 - it must contain, in a stable form, information encoding the organism’s structure, function, development and reproduction. b. Property 2 - it must replicate accurately so progeny cells have the same genetic makeup. c. Property 3 - it must be capable of some variation (mutation) to permit evolution. How is DNA replicated? a. Historical Perspective i. 1928 - Griffith discovered a “transforming principle” in heat killed bacteria (Property 1) ii. 1944 - Avery demonstrated that the transforming principle is sensitive to DNase iii. 1952 - Hershey & Chase used 32P and 35S labeling, of bacteriophage DNA and protein to show only the DNA enters the host cell and can be passed to progeny phage (consistent with Property 2) b. The exact method still had yet to be worked out. But, there were three similar but varying ideas about how this happened: i. Conservative Model ii. Semiconservative Model iii. Dispersive Model. Meselson and Stahl (1958) - get the credit for working out which model was correct. Their experiment is as follows: a. Their Experiment – DESCRIBE IN YOUR NOTES SIDEBAR b. Results = Proof for Semi-conservative model. c. Replicated DNA is passed to each new cell, each new generation. Creates complimentary strands. The old strands serve as templates. Replication of DNA involves unwinding the double helix and synthesizing two new strands. a. Deoxynucleotide Triphosphates i. Nucleotides are called DNTP’s before they are bonded together to form a DNA molecule. ii. Phosphodiester bond - Connect via Oxygen in phosphate. Forms an ester. b. Nucleotides ALWAYS connect to 3’ carbon. c. DNA Synthesis occurs 5’ to 3’ d. Leading vs Lagging Strand: i. Leading Strand – Manufactured as a single complete strand. ii. Lagging Strand – Build in segments that are connected later. SHS Biotechnology 135 R e p l i c a t i o n F o r k – T h e p o i n t w h e r e h e l icase unwinds and unzips the DNA molecule’s two backbones. Enzymes in Replication: i. Helicase: Unwinds a portion of the DNA Double Helix. ii. RNA Primase: Attaches RNA primers to the replicating strands. iii. DNA Polymerase III (delta): Binds to the 5' - 3' strand in order to bring nucleotides and create the daughter leading strand. iv. DNA Polymerase III (epsilon): Binds to the 3' - 5' strand in order to create discontinuous segments starting from different RNA primers. v. RNase H: Removes RNA primers. vi. Exonuclease (DNA Polymerase I): Finds areas where RNA Primers were removed, and replaces with DNA nucleo. vii. DNA Ligase: Adds phosphate in the remaining gaps of the phosphate - sugar backbone. viii. Nucleases: Repair nucleotide errors. Replication of DNA and Chromosomes a. Speed of DNA replication: 3,000 nucleotides/min in human 30,000 nucleotides/min in E.coli b. Accuracy of DNA replication: Very precise = 1 error/1,000,000,000 nucleotides. Where does replication start? a. Different in Prokaryotes and eukaryotes. i. Only One Replication Origin in E. coli ii. Bi-directional replication in E. coli b. Multiple Origins in Eukaryotes i. Eukaryotes have larger chromosomes than prokaryotes. ii. Replication speed 2,600 npm. Largest Drosophila (fruit fly) chromosome is 65,000,000 nucleotides long, but it can replicate in 3-4 min. iii. From a single origin, replication should take 8.5 days. iv. That means each chromosome must have some 7,000 origins of replication = Replication Bubbles f. VI. VII. SHS Biotechnology 136 c. PCR: Polymerase Chain Reaction I. II. III. IV. V. VI. VII. VIII. The Nobel Prize in Chemistry 1993 Kary B. Mullis "for contributions to the developments of methods within DNA-based chemistry" "for his invention of the polymerase chain reaction (PCR) method” Taq polymerase a. is an enzyme extracted from Thermus aquaticus, a bacterium found in the hot springs of Yellowstone National Park. b. Thermus aquaticus - The Bacterium from which Taq Polymerase is isolated.. c. Thermal Cycler – the machine that does the PCR process Materials of PCR : a. target DNA, Taq DNA polymerase, 2 Primers, ~20 nucleotides in length, Forward and reverse, four DNTP’S, AND cofactor MgCl2. How does PCR Happen? a. Locate the Target Sequence. Scientists determine which GENE they are interested in studying. b. Locate Primers Upstream and Down-stream of gene c. There are three major steps in a PCR, which are repeated for 30 or 40 cycles. This is done on an automated cycler, which can heat and cool the tubes with the reaction mixture in a very short time. Step 1 Denaturing a. 60 seconds @ 94°C b. The first part of the process separates the two DNA chains in the double helix. This is done simply by heating the vial to 90-95 degrees centigrade for 30 seconds to 1 minute. c. The heating of the sample breaks the weak hydrogen bonds between the nitrogen bases. Step 2 Annealing a. 60 seconds @ 54 - 60°C b. Forward and Reverse Primers c. Because the primers cannot bind to the DNA strands at such a high temperatures, the vial is cooled to approximately 65 degrees C . At this temperature, the primers bind or "anneal" to the ends of the DNA strands by complimentary base pairing. This takes about 30 seconds. d. The optimum temperature for annealing varies upon the number of A-T to G-C base pairing in the primer sequence. Taq Polymerase Binds a. Remember DNA Polymerases cannot replicate DNA w/o something to bond to. Step 3 Extension a. 2 minute at 72°C DNTP’s b. Since the Taq polymerase works best at around 75 degrees C (the temperature of the hot springs where the bacterium was discovered), the temperature is raised. SHS Biotechnology 137 T h e T a q p o l y m e r a s e b e g i n s a d d i n g n u c l e IX. X. otides to the primer and eventually makes a complementary copy of the template. The polymerase adds dNTP's from 5' to 3', reading the template from 3' to 5' side. d. This completes one PCR cycle. The exponential amplification of the gene in PCR. Applications of PCR a. quick, reliable method for detecting all manner of mutations associated with genetic disease from insertions, to deletions, to point mutations. i. Duchene muscular dystrophy ii. Detect unwanted Genetic material iii. Bacterial or viral infection iv. HIV infection v. Amplify degraded DNA samples vi. Egyptian mummy b. Termite in amber SHS Biotechnology 138 PCR of Chromosome 16: PV92 Gene (PTC Taste) – Pre-Lab Reading PCR (Polymerase Chain Reaction) is a revolutionary method developed by Kary Mullis in the 1980s. PCR is based on using the ability of DNA polymerase to synthesize new strand of DNA complementary to the offered template strand. Because DNA polymerase can add a nucleotide only onto a preexisting 3'-OH group, it needs a primer to which it can add the first nucleotide. This requirement makes it possible to delineate a specific region of template sequence that the researcher wants to amplify. At the end of the PCR reaction, the specific sequence will be accumulated in billions of copies (called amplicons). Components of PCR in a PCR Reaction Tube: DNA template - the sample DNA that contains the target sequence. At the beginning of the reaction, high temperature is applied to the original doublestranded DNA molecule to separate the strands from each other. DNA Taq polymerase - a type of enzyme that synthesizes new strands of DNA complementary to the target sequence. The first and most commonly used of these enzymes is Taq DNA polymerase (from Thermis aquaticus). Another commonly used PCR enzyme is Pfu DNA polymerase (from Pyrococcus furiosus) is used widely because of its higher fidelity when copying DNA. Although these enzymes are subtly different, they both have two capabilities that make them suitable for PCR: 1) they can generate new strands of DNA using a DNA template and primers, and 2) they are heat resistant. Primers - short pieces of single-stranded DNA that are complementary to the target sequence. The polymerase begins synthesizing new DNA from the end of the primer. Nucleotides (dNTPs or deoxynucleotide triphosphates) - single units of the bases A, T, G, and C, which are essentially "building blocks" for new DNA strands. How is PCR (polymerase chain reaction) done? Three major steps are involved in a PCR. These three steps are repeated for 30 or 40 cycles. The cycles are done on an automated Thermal cycler, a device which rapidly heats and cools the test tubes containing the reaction mixture. Each step -- denatauration (alteration of structure), annealing (joining), and extension -- takes place at a different temperature: Denaturation: At 94 C (201.2 F), the double-stranded DNA melts and opens into two pieces of single-stranded DNA. Annealing: At medium temperatures, around 54 C (129.2 F), the primers pair up (anneal) with the single-stranded "template" (The template is the sequence of DNA to be copied.) On the small length of double-stranded DNA (the joined primer and template), the polymerase attaches and starts copying the template. Extension: At 72 C (161.6 F), the polymerase works best, and DNA building blocks complementary to the template are coupled to the primer, making a double stranded DNA molecule. With one cycle, a single segment of double-stranded DNA template is amplified into two separate pieces of double-stranded DNA. These two pieces are then available for amplification in SHS Biotechnology 139 the next cycle. As the cycles are repeated, more and more copies are generated and the number of copies of the template is increased exponentially. .Limitations of PCR: The PCR reaction starts to generate copies of the target sequence exponentially. Only during the exponential phase of the PCR reaction is it possible to extrapolate back to determine the starting quantity of the target sequence contained in the sample. Because of inhibitors of the polymerase reaction found in the sample; reagent limitation, accumulation of pyrophosphate molecules, and self-annealing of the accumulating product, the PCR reaction eventually ceases to amplify target sequence at an exponential rate and a "plateau effect" occurs, making the end point quantification of PCR products unreliable. After the PCR Reaction: PCR amplification is only part of the identifying test, however. Once the amplification is done, the amplified segments need to be compared to other nucleotide segments from a known source (for example, a specific person, animal, or pathogenic organism). This comparison of unique segments is often done by placing PCR-generated nucleotide sequences next to known nucleotide sequences from humans, pathogens, or other sources in a separating gel. Electrical current is run through the gel and the various nucleotide sequences form bands that resemble a "ladder" according to their electrical charge and molecular size (gel electrophoresis). Bands or "ladder" like steps that migrate to the same levels in the gel show identity of nucleotide sequences. This method is one of the most popular ways PCR tests are completed. SHS Biotechnology 140 Grading Rubric – For Teacher’s Use PCR of Chromosome 16: PV92 Gene (PTC Taste) – Full Blown Lab I will deduct points for each of the following: Name: ___________________________ Due Date: ____/____/____ This is to be written in complete sentences with correct grammar, spelling and punctuation. Question: Is it possible to detect the presence of a specific gene or set of alleles in an individual’s genome by using the process of PCR? Hypothesis (written in “If… then…because” format): ______________________________________________________________________________ ______________________________________________________________________________ Independent Variable: ___________________________________________________________ Dependent Variable: ____________________________________________________________ After completing the lab, tell what affect did the INDEPENDENT VARIABLE had on the DEPENDENT VARIABLE (describe the relationship between the two variables in a minimum of 3 sentences)? ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ MATERIALS: InstaGene™ matrix Lab marker Mastermix Loading dye 1x TAE electrophoresis buffer PV92 homozygous (+/+) microtube rack 2 Water baths (56 and 100°C) Ice Gel box and power supply DNA stain PV92 homozygous (-/-) micropipettes pipette tips Centrifuge PCR tubes ThermoCycler Agarose gel Gel staining tray MMR-DNA standard PV92 heterozygous (+/-) PROTOCOL: /5 Hypothesis: Does not use proper format – If/Then/Because. Hypothesis does not completely answer lab question. Hypothesis is poorly stated. Does not clarify what the student is thinking. /10 Independent Variable: No variable identified. Incorrect Independent variable. /5 Dependent Variable: No variable identified. Incorrect Dependent variable. /5 Link between variables: Answer not 3 sentences. Question is not answered. Link between variables is not explained clearly. The link described between variables is incorrect. Description of what was done in lab is not an answer to this question. /10 PART 1: 1. Each member of your team should have two screw-cap tubes, each containing 200 μl of InstaGene matrix. Label the tube on the cap and on the side with your initials. SHS Biotechnology Overall: Not written in complete sentences. Does not use correct grammar spelling & punctuation. 141 2. Label the tubes as "tube 1" and "tube 2". 3. Using a sterile 20–200 μl pipet tip, gently scrape the inside of both cheeks 10 times each with the tip. This is most easily done by pinching and extending the corner of your mouth with one hand, and scraping the cheek with the tip in the other hand. Use firm, but gentle pressure. The goal is to remove the surface layer of epithelial cells from your cheek lining. 4. Place the tip that contains your cheek cells into the screw-cap tube labeled as "tube 1". 5. Using a second sterile 20–200 μl filter pipet tip, gently scrape the inside of both cheeks 10 times each with the tip. Place the tip that contains your cheek cells into your screwcap tube labeled as "tube 2". 6. Place each of the tips on the end of a P-200 micropipette that is set on a 100 μl setting. Pipet up and down 5 times into the InstaGene matrix – the action of pipetting up and down mixes and transfers your cheek cells into the InstaGene matrix. 7. Screw the lid tightly on your tubes and place in the foam micro-tube holder. When all members of your team have collected their samples, float the rack and tubes in a 56°C water bath for 10 min. 8. At the halfway point (5 minutes), remix the contents of the tubes by shaking or vortexing several times and place back in the water bath for the remaining 5 minutes. 9. Remove the tubes from the water bath and remix by vortexing the tubes. 10. Now float the rack of tubes in a 100°C water bath for 6 minutes. 11. After 6 minutes, remove the tubes from the 100°C water bath and vortex several times to resuspend the sample. 12. Place the eight tubes in a balanced arrangement in the centrifuge. Pellet the matrix by spinning for 5 minutes at 6,000 x g (or 10 minutes at 2,000 x g) in the centrifuge. SHS Biotechnology 142 13. Using a 200 μl pipet tip, remove 170 μl of the supernatant from one of your screwcap tubes and transfer the supernatant into the other. You will now be left with one screwcap tube that contains your isolated genomic DNA. 14. Give your tubes to your teacher for storage until the next lab class. PART 2: 1. 2. 3. 4. 5. 6. After obtaining your tubes again, centrifuge them for 2 minutes at 6,000 x g or for 5 minutes at 2,000 x g in a centrifuge. Each member of the team should obtain a PCR tube and capless micro-tube adaptor. Label each PCR tube with your initials and place it into the capless micro-tube (which acts as a convenient holder for the small PCR tube). Place the PCR tube in its holder in the foam rack. Transfer 20 μl of your DNA template from your screwcap tube into the bottom of the PCR tube. Do not transfer any of your matrix beads into the PCR reaction—the reaction will be inhibited. Locate the tube of yellow PCR master mix (labeled "Master" or “MM”), which will be on ice on the front counter. Transfer 20 μl of the master mix into your PCR tube. Mix by pipetting up and down 2–3 times. Remove your PCR tube from its holder and place the tube in the Thermal Cycler. When all of the PCR samples are in the Thermal Cycler, the teacher will begin the PCR reaction. The reaction will undergo 40 cycles of amplification, which will take approximately 3 to 4 hours. SHS Biotechnology 143 PART 3: 1. 2. 3. 4. Remove your PCR samples from the freezer and place in the micro-tube rack. If a centrifuge is available, place the PCR tubes in the adaptor and pulse spin the tubes (~3 seconds at 2,000 x g) to bring the condensation that formed on the lids to the bottom of the tubes. Add 10 μl of 5x loading dye to each of your PCR tubes. Obtain a pre-poured agarose gel. Place the casting tray with the solidified gel in it, into the platform in the gel box. The wells should be at the (-) cathode end of the box, where the black lead is connected. Pour ~275 ml of electrophoresis buffer into the electrophoresis chamber. Pour buffer into the gel box until it just covers the wells. Using a separate tip for each sample, load the samples into the first seven wells of the gel in the following order: Lane Lane 1: Lane 2: Lane 3: Lane 4: Lane 5: Lane 6: Lane 7: Lane 8: (If Needed) 5. 6. 7. Sample MMR-DNA Standard Homozygous (+/+) control Homozygous (-/-) control Heterozygous (+/-) control Student 1 Student 2 Student 3 Student 4 Amount 10 μl 20 μl 20 μl 20 μl 20 μl 20 μl 20 μl 20 μl Secure the lid on the gel box. The lid will attach to the base in only one orientation: red to red and black to black. Connect electrical leads to the power supply. Turn on the power supply. Set it to 100 V and electrophorese the samples for 45 minutes. DNA fragments are visualized by staining the gel with a blue DNA stain. The blue dye molecules are positively charged and have a high affinity for the DNA. These blue dye molecules strongly bind to the DNA fragments and allow DNA to become visible. SHS Biotechnology 144 8. This protocol allows quick visualization of DNA bands in agarose gels within 15 minutes. For quick staining, 120 ml of 100x Fast Blast DNA stain can stain two 7 x 7 cm or 7 x 10 cm agarose gels. Table 1 Completed: _____/20 RESULTS: Table 2 Completed: 1. Based on the band patterns that you see in your gel, record the genotype of the individuals who were tested in the lab in table 1: Table 1 Individual Name Genotype (+/+, -/-, +/-) _____/20 2. Collect some class data by filling out the Table 2: Table 2 Genotype This Class All Classes Expected % Class % Data Data +/+ 25 % +/50% -/25% Total 100% SHS Biotechnology All Classes % 145 Draw the gel that you created in lab in the diagram below: Gel Drawing Completed: _____/25 Hypothesis Correct? Uses the words “Yes” or “No” to start. First sentence does not state clearly, “My hypothesis was correct/not correct.” 2 items of detailed data measured in lab (numbers, observations) were not provided to support decision. Data used is not explained. Student does not make a choice. A hypothesis cannot be both right and wrong. The decision about hypothesis correctness does not match the data collected in lab. _____/10 Hypothesis related choice 1: Based on the data collected, was your hypothesis correct? Give 2 pieces of data that clarify your decision: ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ CHOOSE 1 based on whether your hypothesis is correct: If your hypothesis was wrong, what was wrong with it (detail – minimum 2 sentences)? If your hypothesis was right, generate a new lab question that could be asked to create a new lab experiment to follow up on what you learned in this experiment. OR ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ SHS Biotechnology 146 Wrong question answered. Does not tell what revisions to hypothesis would make it correct. Evaluates lab procedures instead of the hypothesis. Does not tell why the hypothesis was wrong. Not 2 sentences long. _____/10 Hypothesis related choice 2: Wrong question answered. Basically a repeat of the question from this lab. Not in the form of a question. Little thought or creativity shown. Not testable in lab. Question is too general. Neither question is answered. _____/10 Choice 1: CHOOSE 1: How could the experiment be repeated to make it more accurate (describe 2 ways to improve the experiment)? Were there any mistakes made in the process of following the procedures that affected the outcome? If so, how could these mistakes be corrected for (be detailed about the mistakes you made and how you would correct for them)? OR ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ How can the data we collected and the concepts we learned in this lab be applied to life? In other words, what is the practical application of the concepts from this lab (minimum 3 sentences)? ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ Using the picture of the gel below, and what you learned about PCR in the lab, determine how many people have each of the genotypes tested (lane 2 is the control for +/+, lane 3 is the control for -/-, and lane 5 is the control for +/-). Lanes 8 & 15 are empty on purpose. Not enough detail. 2 ways not given. Answers were complaints not solutions. Saying, “Follow all instructions” is not a solution. 1 sentence is not enough. Don’t blame others, find solutions. This student does not understand what was happening in lab. _____/10 Choice 2: Doesn’t tell what you did wrong & how to fix it next time. Should be longer than 1 sentence. Saying, “We didn’t make any mistakes,” means you have to answer the other question. Saying, “Follow all the steps better,” does not work for an answer. This student does not understand what was happening in the lab. _____/10 Application: Answers should be specific with at least one example/career/life experience listed. Not 3 sentences long. Student does not understand how to apply this to life or what was done in lab. Don’t give opinions (i.e this is dumb). Don’t retell what we did in lab. +/+ = _______ _____/10 -/- = ________ Specific Question #1: +/- = ________ SHS Biotechnology 147 ______/10 Describe what the Thermal Cycler does in each of the three steps of the process of PCR (Denature, Anneal, Extension): ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ Specific Question #2: _____/15 Specific Question #2: _____/20 Specific Question #3: What substances are needed in the PCR tube before it can be placed into the thermal cycler, and what are their purposes? _____________________________________________________________________________________ _____________________________________________________________________________________ _____________________________________________________________________________________ _____________________________________________________________________________________ Using the picture of the gel to the right, explain why we are able to use gel electrophoresis to separate the two versions of this gene (+ and -) by size: _____________________ _________________ _________________ _________________ _________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ SHS Biotechnology 148 _____/20 Total Score: ________/205 DNA Sequencing Pre-Lab Reading DNA SEQUENCING DNA sequencing was developed during the 1970’s from research groups in the United States and England. Since its early days, these methods have been refined and automated. There are two basic approaches to DNA sequence analysis. One involves a set of organic chemical reactions while the other uses an enzymatic process. The chemical method is tedious and labor-intensive, whereas the enzymatic approach, which is often called the dideoxy method or Sanger sequencing, is quite fast. The Sanger sequencing method is based on the selective incorporation of chain-terminating dideoxynucleotides by DNA polymerase during PCR. For sequence analysis, four separate enzymatic reactions are performed, one for each nucleotide. Each reaction contains a DNA polymerase, a DNA template to which a primer has already been annealed, all four deoxyribonucleotide triphosphates (dATP, dGTP, dCTP, dTTP), and the appropriate buffer for in vitro DNA synthesis. The “G” reaction contains dideoxyGTP, the “C” reaction DideoxyCTP, the “A” reaction DideoxyATP, and the “T” reaction DideoxyTTP. Once a Dideoxynucleotide is incorporated into a single strand, DNA synthesis is completely terminated. The site of the Dideoxynucleotide incorporation allows one to determine the position of that base. The Dideoxynucleotide lacks a 3’-OH group on the deoxyribose ring and it is impossible to add another nucleotide to the growing strand since a 3’-OH group is absolutely required. Non-isotopic methods using fluorescent dyes and automated DNA sequencing machines have replaced the traditional isotopic methods. In spite of the detection differences, the basic biochemistry of the dideoxy sequencing method is essentially the same. Since a particular reaction will contain millions of growing DNA strands, a “nested set” of fragments is obtained with each fragment is terminated at a different position corresponding to the random incorporation of the Dideoxynucleotide. Figure 4 shows the “nested set” of fragments procuded for a hypothetical sequence in the “G” reaction. The “G” reaction contains dATP, dCTP, dGTP, dTTP, the Klenow fragment of DNA polymerase, an appropriate solution for DNA synthesis, dATP and a small amount of dideoxyGTP. As can be seen, ddGTP (dideoxyGTP) incorporation randomly and infrequently will produce a “nested set” of fragments which terminate with a ddGTP. The “nested set” is complimentary to the region being sequenced. Similar “nested sets” are produced in the separate “A”, “T”, and “C” reactions. For example, the “A” “nested set” would terminate with a ddATP. It should be readily apparent that together the “G, A, T, C” “nested sets” contain radioactive 32P-la-labeled fragments ranging in size for the hypothetical sequence. SHS Biotechnology 149 DNA Sequencing Lab – Process Lab PURPOSE: To run the results of a sequencing PCR experiment on a gel and interpret the results MATERIALS: DNA Sequenced "A" TAE Buffer (50x) Power supply DNA Sequenced "C" Loading Dye DNA Sequenced "G" DNA Stain DNA Sequenced "T" Micropipettes PROTOCOL: 1. Place gel (on the tray) into electrophoresis chamber. 2. Cover the gel with TAE buffer. The gel should be completely submerged. 3. Load the entire sample into the well (35-38 μL) in the order indicated by Table 1. Gel Loading Instructions: Lane Tube Label on Strip Nucleotide Sequences in Tube: 1 A or E Adenine (A) 2 B or F Cytosine (C) 3 C or G Guanine (G) 4 D or H Thymine (T) 4. Place cover on the electrophoresis chamber and check that the gel is properly oriented. 5. Connect the leads to the power source and perform the electrophoresis for 30 min. at 150 volts. 6. After electrophoresis is complete, remove the gel and casting tray from the electrophoresis chamber and stain the gel according to the instructions given by your teacher. 7. After staining, draw your gel in the diagram provided on this page. 8. Based on your drawing and the example picture provided, what is the sequence of bases in the piece of DNA we sequenced in lab? ____________________ _____________________________________________________________ SHS Biotechnology 150 Agarose gel Alectrophoresis Apparatus Questions 1. How is sequenced DNA read on a DNA sequencing gel? 2. What is a dideoxynucleotide? 3. How are these used in DNA sequencing? 4. If the concentration of a dideoxynucleotide is too low when mixed with the other four deoxynucleotides, how would that affect the sequencing results? 5. What would happen if the concentration of a dideoxynucleotide is too high? EDVO-Kit 120 DNA FINGERPRINTING BY PCR AMPLIFICATION SHS Biotechnology 151 Proteins & Genetic Engineering Unit Vocabulary Define the following terms using your textbook. There will be a quiz on these words on ______________________. Amino Acid Ampicillin Amplify Anticodon Central Dogma Codon Coenzyme Competency Electroporation Exon Gene Cloning Gene Splicing Gene Therapy Genetic Engineering Intron Marker Gene Origin of Replication Polypeptide Promoter Recombinant DNA (rDNA) Transcription Transduction Transfection Transformation Transformation Efficiency Translation Transposon SHS Biotechnology 152 RNA and Protein Synthesis: I. II. III. IV. V. DNA: Permanent Molecule. RNA: Temporary Molecule. a. Compare the structure of DNA & RNA: b. RNA stands for Ribo Nucleic Acid – Named after the sugar in its backbone (Ribose). c. Thymine is not present. It has been replaced by Uracil. Types of RNA: a. Messenger (mRNA) is a copy of the information carried by a gene on the DNA. The role of mRNA is to move the information contained in DNA to the translation machinery. b. Transfer (tRNA) is the information adapter molecule. It is the direct interface between aminoacid sequence of a protein and the information in mRNA. c. Ribosomal RNA (rRNA) is a component of the ribosomes, the protein synthetic factories in the cell. Historical Background: a. 1909 - Archibald Garrod i. He suggested genes control enzymes that catalyze chemical processes in cells. ii. Inherited Diseases - “inborn errors of metabolism” where a person can’t make an enzyme. iii. Example: Alkaptonuria – Where urine turns black after exposure to air. Lacks - an enzyme to metabolize alkapton. b. George Beadle and Edward Tatum. Worked with Neurospora and proved the link between genes and enzymes. i. Their Experiment: 1. Grew Neurospora on agar. Varied the nutrients in the agar. 2. Looked for mutants that failed to grow on minimum agar. ii. Experimental Results: 1. Three classes of mutants for Arginine Synthesis. 2. Each mutant had a different block in the Arginine Synthesis pathway. 3. Mutations were abnormal genes. c. One Gene - One Enzyme Hypothesis - One gene controls the synthesis of one protein. Levels of Protein Structure a. Primary- Sequence of amino acids. b. Secondary – curling of chain into helices and sheets. c. Tertiary – Overall bending and shape of the entire amino acid chain. d. Quarternarty – Combination of multiple polypeptide chains into a final protein. Based on the modern understanding that proteins are made from more than 1 polypeptide. SHS Biotechnology 153 a. b. c. The hypothesis has been modified today to be more accurate: One Gene - One Polypeptide Hypothesis. Genetic Code: i. Sequence of DNA bases that describe which Amino Acid to place in what order in a polypeptide. ii. The genetic code gives the primary protein structure. iii. Code Is based on triplets of bases. iv. Has redundancy; some AA's have more than 1 code. Codon - A 3-nucleotide “word” in the Genetic Code. 64 possible codons known. SHS Biotechnology 154 Protein Synthesis Notes: I. II. III. IV. V. VI. Overview a. Transcription: synthesis of RNA under the direction of DNA (mRNA) b. Translation: actual synthesis of a polypeptide under the direction of mRNA Transcription - Process of making RNA from a DNA template. a. Transcription Steps: 1) RNA Polymerase Binding. 2) Initiation. 3) Elongation. 4) Termination. b. RNA Polymerase: Enzyme for building RNA from RNA nucleotides. c. RNA Polymerase Binding: Requires that the enzyme find the “proper” place on the DNA to attach and start transcription. d. RNA Polymerase Binding Needs: Promoter Regions on the DNA. Transcription Factors. e. Promoters: Regions of DNA where RNA Polymerases can bind. i. About 100 nucleotides long. Include initiation site and recognition areas for RNA Polymerase. ii. TATA Box - Short segment of T,A,T,A. Located 25 nucleotides upstream for the initiation site. 1. Recognition site for transcription factors to bind to the DNA. iii. Transcription Factors - Proteins that bind to DNA before RNA Polymerase. 1. Recognizes TATA box, attaches, and “flags” the spot for RNA Polymerase. iv. Transcription Initiation Complex - The complete assembly of transcription factors and RNA Polymerase bound to the promoter area of the DNA to be transcribed. Initiation: Actual unwinding of DNA to start RNA transcription. a. Requires Initiation Factors. b. Getting Transcription started is complicated. i. Gives many ways to control which genes are decoded and which proteins are synthesized. Elongation a. RNA Polymerase untwists DNA 1 turn at a time. b. Exposes 10 DNA bases for pairing with RNA nucleotides. c. Enzyme moves 5’ 3’. Rate is about 60 nucleotides per second. Termination a. DNA sequence that tells RNA Polymerase to stop. b. RNA Polymerase detaches from DNA after closing the helix. c. Final Product = Pre-mRNA. This is a “raw” RNA that will need processing. Modifications of RNA: 5’ Cap, Poly-A Tail, & Splicing a. 5' Cap - Modified Guanine nucleotide added to the 5' end. i. Protects mRNA from digestive enzymes. ii. Recognition sign for ribosome attachment. SHS Biotechnology 155 b. VII. Poly-A Tail - 150-200 A nucleotides added to the 3' tail i. Protects mRNA from digestive enzymes. ii. Aids in mRNA transport from nucleus. c. RNA Splicing - Removal of non-coding regions of RNA. Coding regions are then spliced back together. i. Introns - Intervening sequences “Garbage”. Removed from RNA. ii. Exons - Expressed sequences of RNA. Translated into AAs. Translation - Process by which a cell interprets a genetic message and builds a polypeptide. a. Materials Required: tRNA, Ribosomes, & mRNA b. Transfer RNA = tRNA - About 80 nucleotides long. i. Carries AA for polypeptide synthesis. ii. Has double stranded regions and 3 loops. iii. AA attachment site at the 3' end. iv. 1 loop serves as the Anticodon. 1. Anticodon - Region of tRNA that base pairs to mRNA codon. Usually is a compliment to the mRNA bases, so reads the same as the DNA codon. c. Ribosomes - Two subunits made in the nucleolus. Made of rRNA (60%)and protein (40%). i. Large Subunit has 3 sites for tRNA. 1. P site: Peptidyl-tRNA site - carries the growing polypeptide chain. 2. A site: Aminoacyl-tRNA site -holds the tRNA carrying the next AA to be added. 3. E site: Exit site d. Translation Steps: 1) Initiation. 2) Elongation. 3) Termination i. Initiation - Brings together: mRNA, a tRNA carrying the 1st AA, the 2 subunits of the ribosome. ii. Initiation Steps: 1. Small subunit binds to the mRNA. 2. Initiator tRNA (Met, AUG) binds to mRNA. 3. Large subunit binds to mRNA. Initiator tRNA is in the P-site iii. Initiation requires other proteins called "Initiation Factors”. e. Elongation Steps: 1) Codon Recognition, 2) Peptide Bond Formation, 3) Translocation. SHS Biotechnology 156 DNA Mutations Notes I. II. III. IV. V. Changes to DNA are called mutations a. A change to the DNA will… change the mRNA, which… may change protein, which… may change a physical trait. The genetic code is degenerate - There are many back-up systems in place in your DNA: a. A mutation may have no effect on the phenotype if it happens in an intron. b. Many amino acids have multiple codons that code for them. c. Changes in the third base of a codon often have no effect. Types of mutations a. Changes to the letters (A,C,T,G bases) in the DNA. b. Point mutation - change to ONE letter (base) in the DNA may cause change to protein, may not. c. Frameshift mutation - addition of a new letter (base) in the DNA sequence deletion of a letter (base) in the DNA. i. These shift the DNA so it changes how the codons are read big changes to protein! Point Mutations - One base change can change the meaning of the whole protein. a. Missense mutation = changes amino acid. b. Silent mutation = no change to protein. c. Nonsense mutation = change to STOP. Frameshift Mutations - Add or delete one or more bases, changes the meaning of the whole protein. a. Addition = add one or more bases. b. Deletion = lose one or more bases. SHS Biotechnology 157 Size Exclusion Chromatography Notes I. II. III. IV. V. VI. Proteins - 75% of dry matter in living things is protein. a. Biologist must purify protein from other proteins in the cell. b. What are characteristics of Proteins we could use to separate them? i. Hydrophilic / Hydrophobic ii. Positive / Negative iii. Size iv. Active site v. Amino Acids Chromatography - Allows the separations of individual component from complex mixtures a. Biology = Separation of chlorophyll b. Biotechnology = Purify biological molecules c. Two phases: i. Mobile phase – solvent, molecule to be separated. ii. Stationary phase – Paper, glass beads, resin. 1. Molecules travel through the stationary phase at different rates because of their chemistry. d. Protein Purification i. Affinity Chromatography - Ion Exchange, Anion exchange, Cation exchange. ii. Gel Filtration Chromatography - size exclusion chromatography. Affinity Chromatography – Uses the molecules Biological function or Individual chemical structure a. Loading affinity column. b. Proteins sieve through matrix of affinity beads. c. Proteins interact with affinity ligand with some binding loosely and others tightly. d. Wash off proteins that do not bind. e. Wash off proteins that bind loosely. f. Elute proteins that bind tightly to ligand and collect purified protein of interest. Ion Exchange Chromatography - separates biomolecules based on differences in their (anionic / cationic) charge characteristics. Gel Filtration Chromatography – Same process as Affinity. But the function of the gel is different. Hemoglobin is brown in color, 65,000 Daltons in size, made of 4 sub units a. Vitamin B12 is pink in color, 1350 Daltons in size SHS Biotechnology 158 Size Exclusion Chromatography – Process Lab PURPOSE: To learn how the process of size exclusion / affinity chromatography works in order to use it in an upcoming lab. MATERIALS: (12) Collection tubes (1) Column end-caps 1 (1) Lab marker 1 (1 drop) Protein mixture 1 (1) Chromatography column (1) Pipette (1) Test tube rack (4 mL) Column buffer PROTOCOL: 1. Place the 12 collection tubes in your test tube rack. 2. Label 10 collection tubes sequentially from 1 to 10. Label the last two tubes “waste” and “column buffer”. 3. Pipette 4 ml of Column Buffer into the tube labeled column buffer. 4. Remove the caps on both ends of the Poly-Prep sizing column. Drain all of the buffer into the “waste” collection tube. 5. Gently place the column onto collection tube 1 (Do not jam the column tightly into the collection tubesthe column will not flow). 6. Observe the top of the column bed; all of the buffer should have drained from the column. This is best observed by looking directly over the column—the “grainy” appearance of the column beads should be visible. Carefully load one drop of protein mix onto the top of the column bed. The pipette should be inserted into the column and the drop should be loaded just above the top of the column so that it minimally disturbs the column bed. 7. Allow the protein mix to enter the column bed. This is best observed by looking directly over the column. 8. Carefully add 250 μl of column buffer to the top of the column. This is best done by inserting the pipet tip into the column so that it rests just above the column bed. Carefully let the buffer run down the side of the tube and onto the top of the bed. Begin to collect drops into tube 1. 9. When all of the liquid has drained from the column, add another 250 μl of column buffer to the top of the column. 10. When all of the liquid has drained from the column, add 3 ml of column buffer to the top of the column. This can be done by adding 1 ml from the pipette three times. Transfer the column to tube 2 and begin to count the drops that enter into each tube. Collect 5 drops of buffer into tube 2. SHS Biotechnology 159 11. When 5 drops have been collected into tube 2, transfer the column onto tube 3. Collect 5 drops of buffer into each collection tube. When 5 drops have been collected into a tube, lift it off and transfer it to the next tube. 12. Continue collecting 5 drops into each tube. When you reach tube 10, collect a final 10 drops. Cap the column when finished collecting drops. 13. Store your samples and column according to your teachers instructions. RESULTS: Record the color and substance found in each of the tubes in the table below: Tube 1 Tube 2 Tube 3 Tube 4 Tube 5 Tube 6 Tube 7 Tube 8 Tube 9 Tube 10 Color Description Color (with pencils) Substance in Tube Draw an arrow under the table at the tube(s) that would have the most pure samples of vitamin B12. Draw a star under the table at the tube(s) that would have the most pure sample of hemoglobin. SHS Biotechnology 160 Genetic Engineering Notes I. II. III. IV. V. VI. VII. What is Genetic Engineering? - Involves the manipulation of genes (DNA sequence) within a cell or organism to produce a desired result. a. A change in the genetic makeup of an organism through: i. deliberate addition (insertion) ii. Removal (deletion) iii. intentional mutation of DNA b. Most commonly it involves the transfer of a gene from one organism to another. Selective Breeding - Breed only those plants or animals with desirable traits. People have been using selective breeding for 1000’s of years with farm crops and domesticated animals. Recombinant DNA - The ability to combine the DNA of one organism with the DNA of another organism. a. Recombinant DNA technology was first used in the 1970’s with bacteria. b. Recombinant Bacteria i. Remove bacterial DNA (plasmid). ii. Cut the Bacterial DNA with restriction enzyme. iii. Cut the DNA from another organism with same restriction enzyme. iv. Combine the cut pieces of DNA together with another enzyme and insert them into bacteria. v. Reproduce the recombinant bacteria. vi. The foreign genes will be expressed in the bacteria. Benefits of Recombinant Bacteria a. Bacteria can make human insulin or human growth hormone. b. Bacteria can be engineered to “eat” oil spills. How to Create a Genetically Modified Plant a. Create recombinant bacteria with desired gene. b. Allow the bacteria to “infect" the plant cells. c. Desired gene is inserted into plant chromosomes. Uses in Medicine a. Gene therapy - Transforming sick person’s cells with healthy copies of defective genes. b. 1982 - Humulin® is approved for the treatment of diabetes. Basic Steps of Genetic Engineering a. Step 1 Isolation of Foreign DNA - involves finding the gene you want to sequence and cutting it out of the chromosome (DNA) with restriction enzymes SHS Biotechnology 161 i. R e s t r i c t i o n E n z y m e s E v o l v e d b y b acteria to protect against viral DNA infection b. Step 2 Insertion of DNA into Bacterial Plasmid - scientists cut plasmid (DNA) with restriction enzyme. They insert gene (DNA) of interest forming recombinant DNA i. Recombinant DNA - The manipulation and combination of DNA from two sources. ii. Recombination - Insert a foreign gene into a host (Plasmid for example) into the bacterial cell. 1. Goal – To produce many copies ( clones) of a particular gene 2. Reporter gene – tags gene of interest – to identify the presence of a gene c. Vectors – carry the New gene to its final destination i. Characteristics of a Vector 1. Can replicate independently in the host cell. 2. Has restriction sites in the vector. 3. Has a reporter gene that will announce its presence in the host cell. 4. Is a small size. d. Step 3 Transformation - Insert recombinant plasmid into bacteria. Bacteria produced with the recombinant DNA expresses the gene of interest. e. TRANSFORMATION - THE PROCESS BY WHICH BACTERIA CELLS PICK UP AND INCORPORATE DNA FROM DEAD BACTERIA CELLS. SHS Biotechnology 162 Rainbow Transformation Pre-Lab reading DNA CAN BE TRANSFERRED BETWEEN BACTERIA In nature, DNA is transferred between bacteria using two main methods— transformation and conjugation. In transformation, a bacterium takes up exogenous DNA from the surrounding environment (Figure 1). In contrast, conjugation relies upon direct contact between two bacterial cells. A piece of DNA is copied in one cell (the donor) and then is transferred into the other (recipient) cell. In both cases, the bacteria have acquired new genetic information that is both stable and heritable. Frederick Griffith first discovered transformation in 1928 when he observed that living cultures of a normally non-pathogenic strain of Streptococcus pneumonia were able to kill mice, but only after being mixed with a heat-killed pathogenic strain. Because the non-pathogenic strain had been “transformed” into a pathogenic strain, he named this transfer of virulence “transformation”. In 1944, Oswald Avery and his colleagues purified DNA, RNA and protein from a virulent strain of S. pneumonia to determine which was responsible for transformation. Each component was mixed each with a non-pathogenic strain of bacteria. Only those recipient cells exposed to DNA became pathogenic. These transformation experiments not only revealed how this virulence is transferred but also led to the recognition of DNA as the genetic material. The exact mode of transformation can differ between bacteria species. For example, Haemophilus influenzae uses membrane-bound vesicles to capture doublestranded DNA from the environment. In contrast, S. pneumoniae expresses competency factors that allow the cells to take in single-stranded DNA molecules. In the laboratory, scientists can induce cells—even those that are not naturally competent—to take up DNA and become transformed. To accomplish this, DNA is added to the cells in the presence of specific chemicals (like calcium, rubidium, or magnesium chloride), and the suspension is “heat shocked”—moved quickly between widely different temperatures. It is believed that a combination of chemical ions and the rapid change in temperature alters the permeability of the cell wall and membrane, allowing the DNA molecules to enter the cell. Today, many molecular biologists use transformation of Escherichia coli in their experiments, even though it is not normally capable of transforming in nature. GENETIC ENGINEERING USING RECOMBINANT DNA TECHNOLOGY Many bacteria possess extra, non-essential genes on small circular pieces of double-stranded DNA in addition to their chromosomal DNA. These pieces of DNA, called plasmids, allow bacteria to exchange beneficial genes. For example, the gene that codes for ß-lactamase, an enzyme that provides antibiotic resistance, can be carried between bacteria on plasmids. Transformed cells secrete ß-lactamase into the surrounding medium, where it degrades the antibiotic ampicillin, which inhibits cell growth by interfering with cell wall synthesis. Thus, bacteria expressing this gene can grow in the presence of ampicillin. Furthermore, small “satellite” colonies of untransformed cells may also grow around transformed colonies because they are indirectly protected byß-lactamase activity. Bacterial Transformation Recombinant DNA technology has allowed scientists to link genes from different sources to bacterial plasmids (Figure 2). These specialized plasmids, called vectors, contain the following features: 1. Origin of Replication: a DNA sequence from which bacteria can initiate the copying of the plasmid. 2. Multiple Cloning Site: a short DNA sequence that contains many unique restriction enzyme sites and allows scientists to control the introduction of specific genes into the plasmid. 3. Promoter: a DNA sequence that is typically located just before (“upstream” of) the coding sequence of a gene. The promoter recruits RNA polymerase to the beginning of the gene sequence, where it can begin transcription. SHS Biotechnology 163 4. Selectable marker: a gene that codes for resistance to a specifi c antibiotic (usually ampicillin, kanamycin or tetracycline). When using selective media, only cells containing the marker should grow into colonies, which allows researchers to easily identify cells that have been successfully transformed. TRANSFORMATION EFFICIENCY In practice, transformation is highly inefficient—only one in every 10,000 cells successfully incorporates the plasmid DNA. However, because so many cells are used in a transformation experiment (about 1 x 109 cells), only a small number of cells must be transformed to achieve a positive outcome. If bacteria are transformed with a plasmid containing a selectable marker and plated on both selective and nonselective agar medium, we will observe very different results. Nonselective agar plates will allow both transformed and untransformed bacteria to grow, forming a bacterial “lawn”. In contrast, on the selective agar plate, only transformed cells expressing the marker will grow, resulting in recovery of isolated colonies. Because each colony originates from a single transformed cell, we can calculate the transformation efficiency, or the number of cells transformed per microgram (μg) of plasmid DNA (outlined in Figure 3). For example, if 10 nanograms (0.01 μg) of plasmid were used to transform one milliliter (ml) of cells, and plating 0.1 ml of this mixture (100 microliters, or 100 μl) gives rise to 100 colonies, then there must have been 1,000 bacteria in the 1 ml mixture. Dividing 1,000 transformants by 0.01 μg DNA means that the transformation efficiency would be 1 X 105 cells transformed per μg plasmid DNA. Transformation efficiency generally ranges from 1 x 105 to 1 x 108 cells transformed per μg plasmid. CONTROL OF GENE EXPRESSION Scientists can regulate the expression of recombinant proteins using a genetic “on/off” switch called an inducible promoter (Figure 4). These sequences allow precise control because expression of the gene will only “turn on” in the presence of a small molecule like arabinose, tetracycline, or IPTG (isopropyl-ß-D-thiogalactopyranoside). In this experiment, the plasmids that we will be using to transform our E. coli have been engineered to contain the DNA sequence of blue, pink, or purple chromogenic proteins (pChromoBlue, pChromoPink, pChromoPurple). Expression of these chromogenic proteins is under the control of an inducible promoter. The host bacteria have been genetically engineered to contain the gene for a special RNA polymerase (T7), which is controlled by the lac promoter. Under normal circumstances, the bacteria make a protein called lac repressor, which binds to this promoter and blocks expression of the T7 polymerase. Without T7 polymerase, the chromogenic protein cannot be expressed, and cells will not produce pigment. However, when IPTG is added, lac repressor is inactivated, and T7 polymerase is expressed. This polymerase specifically recognizes the promoter on the chromogenic protein-containing plasmid and transcribes large quantities of mRNA. Finally, the mRNA is translated to produce blue, pink or purple proteins, causing the cells to be pigmented. EXPERIMENT OVERVIEW: In this experiment, chemically competent E. coli will be transformed with a mixture of plasmids that contain genes for ampicillin and a chromogenic protein (pink, purple or blue). Transformants will be selected for the presence of plasmid using LB-ampicillin plates, and the transformation efficiency will be calculated. In addition, some cells will be exposed to IPTG, whereas others will not be exposed to IPTG. Because blue, pink, and purple chromogenic proteins will only be expressed in the presence of the small molecule IPTG, this experiment will demonstrate differential gene expression. At the end of the activity, students will have experience observing and analyzing acquired traits (ampicillin resistance and pigmentation) as exhibited by transformed bacterial cells. Students should also possess an enhanced understanding of the abstract concepts of transformation and gene expression. SHS Biotechnology 164 Rainbow Transformation – Process Lab PURPOSE: - To complete a bacterial transformation and calculate the transformations efficiency for the resulting transformants. MATERIALS: pChromoBlue™ Plasmid DNA, Ampicillin, Petri plates (small), Toothpicks (sterile), Two Water baths (37°C and 42°C), Ice, pChromoPink™ Plasmid DNA, IPTG, Petri plates (large), Inoculating loops (sterile), Thermometer, Starter Plates pChromoPurple™ Plasmid DNA, CaCl2, micropipettes, Microcentrifuge tubes, Incubation Oven (37°C), PROTOCOL: 1. Label one microcentrifuge tube with “+DNA” and a second microcentrifuge tube with “-DNA”. 2. Transfer 500 μL ice-cold CaCl2 solution into the ”– DNA” tube using a sterile 1 ml pipet. 3. Using an inoculation loop, Transfer approx. 10 well-isolated colonies (each colony should be approx. 1-1.5 mm in size) from the E. coli source plate to the “-DNA” tube. 4. Twist the inoculation loop between your fingers to free the cells. 5. Resuspend the bacterial cells in the CaCl2 solution by vortexing vigorously until no clumps of cells are visible and the cell suspension looks cloudy. 6. Transfer 250 μl of the cell suspension to the tube labeled “+ DNA”. PLACE tubes on ice. 7. Add 10 μl of the rainbow transformation mixture “RTM” to the tube labeled “+ DNA”. 8. Gently Mix the sample by twisting the inoculation loop between your fingers (as you did above) while it is in the tube. 9. DO NOT add the plasmid to the “-DNA” tube. 10. Chill the tubes on ice for 10 minutes. 11. Place the transformation tubes in a 42° C water bath for 90 seconds. 12. Immediately return the tubes to the ice and chill for two minutes. 13. Transfer 250 μL of Recovery Broth to each tube. 14. Mix by vortexing the tube. 15. Incubate the cells for 30 minutes in a 37° C water bath. This is considered a recovery period. 16. While the cells are recovering, label the bottom of four agar plates as indicated below: Plate Label LB LB / Amp LB / Amp LB / Amp / IPTG Name & Period / -DNA Name & Period / - DNA Name & Period /+ DNA Name & Period /+ DNA SHS Biotechnology 165 17. After the recovery period, Remove the tubes from the water bath and place them on the lab bench. 18. Transfer 250 μL recovered cells from the tube labeled “ –DNA “ and dispense it directly on the middle of the agar in each of the plates you labeled -DNA. 19. Using a new sterile tip, Transfer 250 μL recovered cells from the tube labeled “ +DNA “ and dispense it directly on the middle of the agar in each of the plates you labeled +DNA. 20. Spread the cells over the entire plate using an inoculating bar as demonstrated by your teacher. Use one sterile bar to spread both -DNA samples. Change to a fresh bar before spreading the +DNA samples. Make sure the cells have been spread over the entire surface of the plates. 21. Cover and Stack the plates on top of one another and TAPE them together. 22. Place the plates in the inverted position (agar side on top) in a 37° C incubator for 24 to 48 hours. 23. If colors are faint, incubate the plates at 4° C for the second 24 of the 48 hours. 24. After incubation, Record the following in Table 1: a. The number of colonies on the plate. b. The colors of bacteria visible on the plates. Plate LB ~ (- DNA) Number of Colonies Colors Observed LB / Amp ~ (- DNA) LB / Amp ~ (+ DNA) LB / Amp / IPTG ~ (+ DNA) SHS Biotechnology 166 Calculate Transformation Efficiency - Your next task in this investigation will be to learn how to determine the extent to which you genetically transformed E. coli cells. This quantitative number is referred to as the transformation efficiency. In many experiments, it is important to genetically transform as many cells as possible. Determine TE for only the Rainbow Colony plate. 1) Total number of cells on plate 2) Determine the amount of DNA spread on platea) Total amount of DNA (µg) used in the “+” tube Total = concentration x volume = ___________ = ___________ b) Fraction of DNA spread on the plate = ___________ c) µg of DNA spread on plate = ___________ d) TE = ___________ SHS Biotechnology 167 Transformation with Blue and Green Fluorescent Proteins Pre-lab Reading The plasmid that we will be using to transform our E. coli has been engineered to contain the DNA sequence the Green Fluorescent Protein (GFP) and the Blue Fluorescent Protein (BFP). GFP and BFP are small proteins, approximately 27 kilodaltons in size. GFP possesses the ability to absorb blue light and emit green light in response, while BFP absorbs violet light and emit blue light in response. This activity, known as fluorescence, does not require any additional special substrates, gene products or cofactors to produce visible light. GFP was first isolated from the jellyfish Aequorea victoria in the1970’s. Once scientists identified its DNA sequence, they were able to use genetic engineering to introduce fluorescent proteins into other organisms, such as E. coli and the nematode Caenorhabditis elegans. Scientists also identified particular amino acid substitutions in GFP that altered the behavior of its ‘chromophore’, a special structure within the protein that is responsible for light production. Different changes bring about different patterns of light absorption and emission, allowing scientists to develop a rainbow of fluorescent proteins. For example, GFP can be converted to BFP by making two amino acid substitutions, one of which is in the chromophore (His-Tyr). For their discovery and development of GFP and other fluorescent proteins, Osamu Shimomura, Martin Chalfie and Roger Tsien were awarded the Nobel Prize in Chemistry in 2008. Fluorescent proteins have become an essential tool in cell and molecular biology. Using DNA cloning strategies, proteins can be “tagged” with fluorescent proteins and then expressed in cells. These tags simplify purification because fluorescently labeled proteins can be tracked using UV light. The most useful application of fluorescent proteins are as a visualization tool during fluorescent microscopy studies. By tagging other proteins with fluorescent proteins, researchers can determine where those proteins are normally found in the cell. Similarly, using a fluorescent protein as a reporter, scientists can observe biological processes as they occur within living cells. For example, in the model organism zebrafish (Daniorerio), scientists use GFP to fluorescently label blood vessel proteins so they can track blood vessel growth patterns and networks. Scientists also tag regulatory DNA sequences with the GFP coding sequence so they can observe patterns of when and where the gene is expressed. In this way, GFP can reveal the role these regulatory sequences might normally play in a cell. In summary, fluorescent proteins including GFP and BFP and fluorescent microscopy have enhanced our understanding of many biological processes by allowing scientists to watch biological processes in real-time. CONTROL OF GENE EXPRESSION Scientists can regulate the expression of recombinant proteins using a genetic “on/off” switch called an inducible promoter. These sequences allow precise control because expression SHS Biotechnology 168 of the gene will only “turn on” in the presence of a small molecule like arabinose, tetracycline, or IPTG (isopropyl-ß-D-thiogalactopyranoside). In this experiment, we will use an inducible promoter to regulate the expression of GFP and BFP. The host bacteria have been genetically engineered to contain the gene for a special RNA polymerase (T7), which is controlled by the lac promoter. Under normal circumstances, the bacteria make a protein called lac repressor, which binds to this promoter and blocks expression of the T7 polymerase. Without T7 polymerase, the fluorescent protein cannot be expressed, and cells will not fluoresce. However, when IPTG is added, lac repressor is inactivated, and T7 polymerase is expressed. This polymerase specifically recognizes the promoter on the fluorescent protein-containing plasmid and transcribes large quantities of mRNA. Finally, the mRNA is translated to produce GFP or BFP protein, causing the cells to fluoresce. In this experiment, chemically competent E. coli will be transformed with pFluoroGreen and/or pFluoroBlue, plasmids that contain genes for ampicillin and a fluorescent protein (GFP and BFP, respectively). Transformants will be selected for the presence of plasmid using LB-ampicillin plates, and the transformation efficiency will be calculated. In addition, some cells will be exposed to IPTG, whereas others will not be exposed to IPTG. Because GFP and BFP proteins will only be expressed in the presence of the small molecule IPTG, this experiment will demonstrate differential gene expression. At the end of the activity, students will have experience observing and analyzing acquired traits (ampicillin resistance and fluorescence) as exhibited by transformed bacterial cells. Students should also possess an enhanced understanding of the abstract concepts of transformation and gene expression. SHS Biotechnology 169 Grading Rubric – For Teacher’s Use Transformation with Blue and Green Fluorescent Proteins – Full Blown Lab I will deduct points for each of the following: Overall: Not written in complete sentences. Does not use correct grammar spelling & punctuation. Name: __________________________________________ /5 This Lab is to be written in complete sentences with correct grammar, spelling and punctuation. QUESTION: How can an organism that has been genetically transformed be identified as transformed? Hypothesis (written in “If… then…because” format): ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ ____________________________________________________________________________ Independent Variable: _____________________________________________________ Dependent Variable: ______________________________________________________ ______________________________________________________________________ ______________________________________________________________________ ______________________________________________________________________ ______________________________________________________________________ MATERIALS: SHS Biotechnology /10 Independent Variable: No variable identified. Incorrect Independent variable. /5 Dependent Variable: No variable identified. Incorrect Dependent variable. After completing the lab, tell what affect the INDEPENDENT VARIABLE have on the DEPENDENT VARIABLE (describe the relationship between the two variables in a minimum of 3 sentences)? E. coli starter plate pFluoroGreen™ plasmid Ampicillin IPTG Luria Broth Agar, sterile Luria “Recovery” Broth Water baths (37°C and 42°C) Incubation Oven (37°C) Ice 4 Poured agar plates (1 LB, 2 LB/amp, 1 LB/amp/IPTG) Hypothesis: Does not use proper format – If/Then/Because. Hypothesis does not completely answer lab question. Hypothesis is poorly stated. Does not clarify what the student is thinking. pFluoroBlue™ plasmid CaCl2 Inoculating loops Microcentrifuge tubes Two Thermometer Long wave U.V. light /5 Link between variables: Answer not 3 sentences. Question is not answered. Link between variables is not explained clearly. The link described between variables is incorrect. Description of what was done in lab is not an answer to this question. /10 170 PROTOCOL: 1. Label one microcentrifuge tube with “+DNA” and a second microcentrifuge tube with “DNA”. 2. Transfer 500 μL ice-cold CaCl2 solution into the ”– DNA” tube using a sterile micropipette tip. 3. Using a sterile inoculation loop, transfer approx. 10 well-isolated colonies (each colony should be approx. 1-1.5 mm in size) from the E. coli source plate to the “-DNA” tube. 4. Twist the inoculation loop between your fingers to free the cells. 5. Resuspend the bacterial cells in the CaCl2 solution by vortexing vigorously until no clumps of cells are visible and the cell suspension looks cloudy. 6. TRANSFER 250 μl of the cell suspension to the tube labeled “+ DNA”. 7. Place tubes on ice. 8. Add the following plasmids to the tube labeled “+ DNA”. DO NOT add the plasmid to the “-DNA” tube. a. 5 μl of each pFluoroGreen™ (GFP) & 5 μl of pFluoroBlue™ (BFP) for a total volume of 10 μl. 9. Incubate the tubes on ice for 10 minutes. 10. Place the transformation tubes in a 42° C water bath for 90 seconds. 11. Immediately return the tubes to the ice bucket and incubate them for two minutes. 12. Add 250 μL of Recovery Broth to each tube using a sterile micropipette tip. 13. Gently mix by flicking the tube. 14. Incubate the cells for 30 minutes in a 37° C water bath. 15. While the cells are recovering, LABEL the bottom of four agar plates with your initials and period as well as with the information indicated below: a. –DNA (LB plate) b. –DNA (LB/Amp plate #1) c. +DNA (LB/Amp plate #2) d. +DNA (LB/Amp/IPGT plate) 16. After the recovery period, remove the tubes from the water bath and place them on the lab bench. 17. Using a sterile micropipette tip, transfer 250 μL recovered cells from the tube labeled “DNA “ to the middle of each plate that you labeled -DNA. 18. Using a new sterile micropipette tip, transfer 250 μL recovered cells from the tube labeled “ +DNA “ to the middle each of the plates that you labeled +DNA. 19. Spread the cells over the entire plate using the method demonstrated by your teacher. 20. Cover the plates and wait five minutes for the cell suspension to be absorbed by the agar. 21. Stack the plates on top of one another and tape them together. 22. Place the plates in the inverted position (agar side on top) in a 37° C bacterial incubation oven for overnight incubation (16-18 hours). SHS Biotechnology 171 23. After incubation, Record the following in Table 1: a. The number of colonies on the plate. b. The colors of bacteria visible on the plates. c. The colors of bacteria visible under UV light on the plate. Table 1 Plate Number Colors of Observed Colonies Table 1 Completed: _____/20 Hypothesis Correct? Colors Under UV LB ~ (- DNA) LB / Amp ~ (- DNA) LB / Amp ~ (+ DNA) _____/10 LB / Amp / IPTG ~ (+ DNA) Hypothesis related choice 1: Based on the data collected, was your hypothesis correct? Give 2 pieces of data that clarify your decision: ______________________________________________________________________ ______________________________________________________________________ ______________________________________________________________________ ______________________________________________________________________ ________________________ CHOOSE 1 based on whether your hypothesis is correct: If your hypothesis was wrong, what was wrong with it (detail – minimum 2 sentences)? If your hypothesis was right, generate a new lab question that could be asked to create a new lab experiment to follow up on what you learned in this experiment. OR ______________________________________________________________________ ______________________________________________________________________ ______________________________________________________________________ ______________________________________________________________________ ________________________ SHS Biotechnology Uses the words “Yes” or “No” to start. First sentence does not state clearly, “My hypothesis was correct/not correct.” 2 items of detailed data measured in lab (numbers, observations) were not provided to support decision. Data used is not explained. Student does not make a choice. A hypothesis cannot be both right and wrong. The decision about hypothesis correctness does not match the data collected in lab. 172 Wrong question answered. Does not tell what revisions to hypothesis would make it correct. Evaluates lab procedures instead of the hypothesis. Does not tell why the hypothesis was wrong. Not 2 sentences long. _____/10 Hypothesis related choice 2: Wrong question answered. Basically a repeat of the question from this lab. Not in the form of a question. Little thought or creativity shown. Not testable in lab. Question is too general. Neither question is answered. _____/10 Choice 1: CHOOSE 1: How could the experiment be repeated to make it more accurate (describe 2 ways to improve the experiment)? OR Were there any mistakes made in the process of following the procedures that affected the outcome? If so, how could these mistakes be corrected for (be detailed about the mistakes you made and how you would correct for them)? ______________________________________________________________________ ______________________________________________________________________ ______________________________________________________________________ ______________________________________________________________________ ______________________________________________________________________ Calculate Transformation Efficiency - Determine TE for GFP/BFP Plate. _____/10 Choice 2: 1) Total number of cells on plate 2) Determine the amount of DNA spread on plate: Not enough detail. 2 ways not given. Answers were complaints not solutions. Saying, “Follow all instructions” is not a solution. 1 sentence is not enough. Don’t blame others, find solutions. This student does not understand what was happening in lab. Doesn’t tell what you did wrong & how to fix it next time. Should be longer than 1 sentence. Saying, “We didn’t make any mistakes,” means you have to answer the other question. Saying, “Follow all the steps better,” does not work for an answer. This student does not understand what was happening in the lab. _____/10 = ___________ T.E. Calculation: ______/20 a) Total amount of DNA (µg) used in the “+” tube = ___________ b) Fraction of DNA spread on the plate =___________ c) µg of DNA spread on plate = _________ d) TE = ___________ SHS Biotechnology 173 How can the data we collected and the concepts we learned in this lab be applied to life? In other words, what is the practical application of the concepts from this lab (minimum 3 sentences)? ______________________________________________________________________ ______________________________________________________________________ ______________________________________________________________________ ______________________________________________________________________ ______________________________________________________________________ ______________________________________________________________________ What was the purpose of the LB plate in the lab? What is it meant to demonstrate to someone looking at the data? ______________________________________________________________________________ ______________________________________________________________________________ ______________________________________________________________________________ Pictured here is a map of the GFP plasmid. In order for the bacteria to glow, they had to be growing on an agar that provided them with the substance arabinose. That is because of the araC operon on the plasmid. Describe how araC controls the transcription of the GFP gene and why arabinose is necessary to make this happen. __________________________________ __________________________________ __________________________________ __________________________________ __________________________________ __________________________________ __________________________________ __________________________________ __________________________________ __________________________________ __________________________________ __________________________________ __________________________________ __________________________________ SHS Biotechnology 174 Application: Answers should be specific with at least one example/career/life experience listed. Not 3 sentences long. Student does not understand how to apply this to life or what was done in lab. Don’t give opinions (i.e this is dumb). Don’t retell what we did in lab. _____/10 Specific Question #1: ________/5 Specific Question #2: ________/15 Describe what a reporter gene is and what we used as a reporter gene in this lab. ______________________________________________________________________ ______________________________________________________________________ ______________________________________________________________________ ______________________________________ What is a vector, and what did we use as our vector in this lab? _____________________________________________________________________ _____________________________________________________________ Exogenous DNA does not passively enter E. coli cells that are not competent. Describe the heat shock we used to make the cells competent. _____________________________________________________________ _____________________________________________________________ _____________________________________________________________ _____________________________________________________________ What is the difference in the amino acid structure of the green and blue fluorescent proteins? _____________________________________________________________________________ _____________________________________________________________________________ _____________________________________________________________________________ SHS Biotechnology 175 Specific Question #3: ________/15 Specific Question #4: ________/15 Specific Question #5: ________/15 Specific Question #6: ________/15 Total Score: ________/195 PURIFICATION & SIZE DETERMINATION OF GREEN & BLUE FLUORESCENT PROTEINS Pre-Lab Reading Bioluminescence from marine microorganisms has been observed by many summer visitors at various beaches around the world. It always fascinates the observer by the repeated parade of both color and light on the sand during the ebb and flow. This observation takes second place to the light produced by the bioluminescent jelly fish, Aquorea victoria. A bright bursting energy of light is observed when energy is transferred to the green fluorescent protein (gfp) which is located in a specialized photogenic cell located in the base of the jellyfish umbrella. There are several variants to gfp protein that have been genetically engineered and which dramatically enhance classroom laboratory experiments. An excellent companion to gfp is the blue fluorescent protein (bfp) which is cloned and well characterized. This family of proteins had been known for some time and significant research in this area had been published. The fluorescent proteins have been cloned and expressed. These proteins do not require substrates, other gene products, or cofactors. When exposed to long or short U.V. light, they will emit a bright green or blue light that is clearly visible in bacteria that are transformed by plasmids that contain genes for the gfp or bfp. Likewise, purification of gfp or bfp is simplified by their detection based on fluorescence. There are many examples of chimeric proteins that are fusion products using the gfp or bfp fluorescent proteins as biological tags. Such fusions are at either the Nor C- terminal and often result in no loss in the fluorescence or biological activity of the chimeric protein. These new biotechnology tools have made it possible to conduct studies that deal with protein localization and trafficking within cells. The green fluorescent protein (gfp) has 238 amino acid residues and has a molecular weight of approximately 40,000 daltons. It appears that most of the intact protein is required for maintaining fluorescence and only small deletions of a few amino acids are allowed without compromising the integrity of the protein structure. Interestingly, the chromophore responsible for light emission is within the primary structure of the protein and resides in a tripeptide at positions 65 to 67 which is cyclic and is composed of the amino acids Ser-Tyr-Gly. The importance of protein folding is clearly demonstrated with gfp where the protein is fluorescent only upon proper conformational folding. The blue fluorescent protein (bfp) is a derivative variant of the gfp. It has a His-66 substitution at the Tyr66 position and a second substitution from Tyr-145 to Phe-145. The initial bfp known as P-4 had only the His-66 substitution and was not as bright as the double mutant. With the crystal structure of gfp determined, several other variations of the gfp were genetically engineered using site directed mutagenesis. In such procedures, specific mutations are introduced in a protein to determine the impact of the mutation on structure and function of the protein. The set of gfp and bfp proteins can be used as a dramatic tool to visually demonstrate the effect of pivotal amino acid changes on the structure and function of proteins. Amino acid substitution can also be used to demonstrate the effects of accumulated mutations on aging and various cancers. In this experiment, bacterial extracts containing gfp or bfp will be fractionated by chromatography using a molecular sieve matrix. Factors that affect the separation include size, shape and associated non-protein biologicals such as carbohydrate residues. The fluorescent proteins will be detected on the column and subsequently in the test tubes by examination under long U.V. light. For most proteins, such columns can also be used to determine apparent molecular weights. Accurate estimation of protein polypeptide composition and size(s) are performed by analyzing the fractions that contain the protein of interest by denaturing polyacrylamide gel electrophoresis SHS Biotechnology 176 Purification & Size Determination of GFP & BFP – Process Lab PURPOSE: In this experiment, students will learn methods and procedures to partially purify the FluoroGreen™ (gfp) and FluoroBlue™ (bfp) fl uorescent proteins. The molecular weights of the proteins will be determined using denatured SDS polyacrylamide gel electrophoresis. MATERIALS: Cell Extract containing green protein (gfp) Chromatography Columns Micropipettes Cell Extract containing blue protein (bfp) Long U.V. lamps Distilled water Column Elution Buffer Ring stands and column clamps PROTOCOL: Part I - Setting up the Chromatography Column: 1. 2. 3. 4. 5. 6. 7. 8. 9. Vertically mount the column on a ring stand or other type of apparatus that will hold the column above the collection tube. Make sure it is straight. Slide the cap onto the spout at the bottom of the column. Fill about one-third of the column with the elution buffer. Mix the slurry (molecular sieve) thoroughly by swirling or gently stirring. Carefully pipet the mixed slurry into the column by letting it stream down the inside walls of the column. If the flow is stopped by an air pocket, stop adding the slurry and firmly tap the column until the air is removed and the slurry continues to flow down the side of the column. Place an empty beaker under the column to collect wash buffer. Remove the cap from the bottom of the column and allow the matrix to pack into the column. Wash the packed column with 5ml of 1x elution buffer. Do not allow the column to dry. Slide the cap onto the spout and make sure it does not drip. Part 2 – Collecting Column fractions of GFP Protein: 1. 2. 3. 4. 5. 6. 7. 8. Label the first set of eight microtest tubes #1-8. Slowly load the column with 200 µL of the GFP extract. Allow the extract to completely enter the column. Elute the column with 1x elution buffer. Add buffer slowly (several drops at a time) to avoid diluting the protein sample. Using the graduated marks on the sides of the tubes, collect 0.5ml fractions in the labeled microcentrifuge tubes. Store fractions on ice immediately upon collection. Do not allow the column to dry. Check all fractions by using long wave U.V. light to identify tubes that contain the fluorescent GFP or BFP proteins. Part 3 – Collecting Column fractions of BFP Protein: 1. 2. Wash the column with 10ml of 1x Elution buffer. Label a second set of eight microtest tubes #9-16. Repeat steps 2-5 (from above) except with 200 µL of the (bfp) extract. Store the 0.5ml fractions on ice immediately upon collection. Do not allow the column to dry. Questions: 1. 2. 3. 4. What is the anticipated difference in apparent molecular weight between pFluoro-Green™ (gfp) and pFluoroBlue™ (bfp)? Why is the molecular sieving matrix swelled prior to packing the column? What is the basis of molecular sieve chromatography? Can molecular sieve chromatography columns be used to separate DNA fragmen SHS Biotechnology 177 BIOTECHNOLOGY Performance Skills Evaluation Score Sheet 2015 - 2016 Performance rating scale: A minimum score of 3 for each of the following skills must be achieved to meet state skill certification requirements. 4 = highly skilled Successfully demonstrated without supervision 3 = moderately skilled Successfully demonstrated with limited supervision 2 = limited skill Demonstrated with close supervision 1 = not skilled Demonstration requires direct instruction and supervision Skill Term 4 1.2 Research and present biotechnology concepts using effective communication skills. 2.1 Demonstrate appropriate use of personal protective devices 2 2.2 Demonstrate proper aseptic/sterilizing techniques. 2 2.4 Demonstrate proper use and handling of micropipettes. 2 3.2 Maintain accurate records and documentation according to minimum good documentation practices (GDP). 4.2 Prepare solutions of defined concentrations and pH. 1 6.1 Prepare bacterial growth media. 1 6.2 Demonstrate the ability to culture and maintain microorganisms. 1 7.1 Perform a restriction digest and analyze the results with gel electrophoresis. 3 7.2 Demonstrate the ability to use PCR technology. 3 7.3 Demonstrate the ability to use proper separation techniques to differentiate between proteins based on size and structure (chromatography and SDS-PAGE). 8.1 Perform a bacterial transformation and analyze results. SHS Biotechnology 178 4 3 4 4 3 2 1