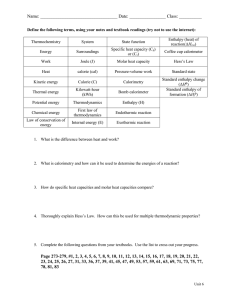
Calorimetry and Enthalpy Chapter 5.2 Heat Capacity • Specific heat capacity (c) is the quantity of thermal energy required to raise the temperature of 1g of a substance by 1⁰C • The units for specific heat capacity are J/(g•⁰C) The water has a higher specific heat capacity so it requires more thermal energy to raise its temperature The sand has a lower specific heat capacity so it requires a less thermal energy to raise its temperature Heat Capacity • Specific heat capacity values can be looked up in tables • There is one in your textbook on page 292 Calorimetry and Thermal Energy Transfer • Calorimetry is the experimental process of measuring the thermal energy change in a chemical or physical change • A calorimeter is a device that is used to measure thermal energy changes in a chemical or physical change Calorimetry Calculations • To simplify our calculations we will make three assumptions: 1. 2. 3. Any thermal energy transferred from the calorimeter to the outside environment is negligible Any thermal energy absorbed by the calorimeter itself is negligible All dilute, aqueous solutions have the same density (1.00g/mL) and specific heat capacity (4.18 J/(g•⁰C)) as water Calorimetry Calculations • We will use the following equation: q is the total amount of thermal energy absorbed or released by a chemical system in joules ∆T is the temperature change experienced by the substance as it warms or cools in ⁰C q = mc∆T m is the mass of the substance in grams note: ∆T = Tfinal - Tinitial c is the specific heat capacity of the substance in J/(g•⁰C) The value of q has two parts: q = - 237J 1. The magnitude tells us how much energy is involved 2. The sign tells us the direction of energy transfer Because of the law of conservation of energy, the total thermal energy of the system and its surroundings remains constant: qsystem + qsurroundings = 0 qsystem = - qsurroundings The chemical system undergoes either a physical change or a chemical change Energy is either absorbed from, or released to, the surroundings (the water in the calorimeter) An increase in the temperature of the water indicates an exothermic reaction, whereas a decrease in the temperature of the water indicates an endothermic reaction Practice Problem 1 • 600mL of water in an electric kettle is heated from 20⁰C to 85⁰C to make a cup of tea. How much thermal energy is absorbed by the water? Practice Problem 2 • The temperature of an aluminum fence post at 5pm is 20⁰C. The same fence post has a temperature of 6⁰C by 11pm. If the fence post releases 315kJ of thermal energy to its surroundings, what is the mass of the fence post? Practice Problem 3 • 50.00mL of aqueous copper (II) sulfate reacts with 50.00mL of aqueous sodium hydroxide in a calorimeter. The initial temperature of both solutions is 21.40⁰C and the highest temperature reached in the calorimeter is 24.60⁰C. Determine the quantity of thermal energy transferred by the reaction to the water, and state whether the reaction was endothermic or exothermic. Enthalpy Change • Enthalpy (H) is the total amount of thermal energy in a substance • Enthalpy Change (ΔH) is the energy released to or absorbed from the surroundings during a chemical or physical change Enthalpy Change Can be Measured Enthalpy change can be measured using calorimetry data As long as pressure is kept constant, the enthalpy change of a chemical system is equal to the flow of thermal energy into or out of the system ΔHsystem = |qsystem| Calculating the enthalpy change can give us information about type of reaction ΔH = Hproducts – Hreactants If ΔH > 0, the reaction is endothermic If ΔH < 0, the reaction is exothermic Molar Enthalpy Change • Molar enthlapy change (ΔHr) is the enthalpy change associated with a physical, chemical, or nuclear change involving one mole of a substance • The units for molar enthalpy change are J/mol Molar Enthalpy Change Many molar enthalpy changes have been carefully measured by scientists and the values are published in tables like the one below and can easily be looked up Using Molar Enthalpy Change • Molar enthlapy values (ΔHr) can help us calculate an enthalpy change (ΔH) • To calculate an enthalpy change (ΔH) for some amount of substance other than one mol, we need to look up the molar enthalpy value (ΔHr) and then multiply it by the number of moles (n) using the formula below ΔH = nΔHr Practice Problem 4 • A common refrigerant (Freon-12 with molar mass 120.91g/mol) is alternately vaporized in tubes inside a refrigerator, absorbing heat, and condensing in tubes outside the refrigerator, releasing heat. This results in energy being transferred from the inside to the outside of the refrigerator. The molar enthalpy of vaporization for the refrigerant is 34.99kJ/mol. If 500.0g of the refrigerant is vaporized, what is the expected enthalpy change? Representing Enthalpy Changes • There are four ways to represent enthalpy changes: 1. 2. 3. 4. Thermochemical equations with energy terms Thermochemical equations with ΔH values Molar Enthalpies Potential Energy Diagrams HOMEWORK Required Reading: p. 292-306 (remember to supplement your notes!) Questions: p. 297 #1-3 p. 301 #1-4 p. 304 #1-4 p. 306 #1-7