1 Unit 3 Section A: Properties of Petroleum Lab
advertisement

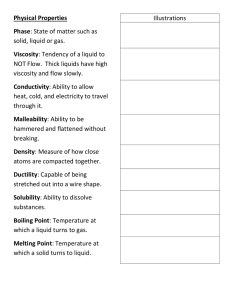
Name: Partner: Unit 3 Section A: Properties of Petroleum Lab Introduction In this investigation, you will explore some of the physical properties of several petroleum-based materials. In particular, you will measure the densities and relative viscosities of several materials separated from petroleum, as well as the same properties of water. Viscosity is the term for resistance to flow. A material with high viscosity flows slowly and with difficulty, like honey. A material with low viscosity flows readily, like water. You will determine relative viscosities, which means ranking materials on a scale from most viscous to least viscous. You will determine if there is a relationship between the number of carbons in a petroleum-based material and its density and viscosity. Procedure Appearance and State of M atter 1. Obtain test tubes containing water, mineral oil, kerosene, motor oil, and household lubricating oil, and a piece of paraffin wax and a piece of asphalt. 2. Record the physical state of matter of each sample in Data Table 1. 3. Describe the appearance of each sample in Data Table 1. Relative Viscosity 1. With your thumb on the cap, hold the capped tube containing water upside down until the bead inside is in the cap of the tube. 2. Gently turn the tube horizontally (the bead will stay at one end). 3. Quickly turn the tube upright so that the bead is at the top and start the timer or stopwatch. 4. Stop the timer when the bead reaches the bottom of the tube. 5. Record the time needed for the bead to reach the bottom of the tube in Data Table 2. 6. Repeat Steps 1 – 5 two more times for water. 7. Repeat Steps 1–6 for each liquid, petroleum-based sample. Density Liquids: 1. Obtain the mass of each of the capped test tubes containing liquid and a bead. Record each mass in Data Table 3. Solids: 1. Determine the mass of each piece of solid sample and record the mass in Data Table 3. 2. Determine the volume of each piece of solid sample by placing the solid in a graduated cylinder partially filled with water. Record the initial volume and the final volume of water in Data Table 3. Questions 1. In Data Table 2, rank your samples in order of relative viscosity, assigning number 1 to the least-viscous material and number 5 to the most-viscous material. 2. Rank the materials (including water) from least to most viscous. 3. Propose a rule, based on your observations in this investigation, about the relationship between number of carbon atoms in a substance and the viscosity of that substance. Cont. on pg. 2 1 4. Calculate the density of each of the liquid and solid samples in Data Table 3. Show your work for all calculations. 5. Does the number of carbons in a hydrocarbon affect the density of that material? Explain your response to question 5, using data collected in this lab: 6. In oil spills, the oil’s density plays a major role. Explain. 7. You should never use water to extinguish a gasoline or oil fire. Why not? Data Table 1 – Appearance and State of M atter M aterial Mineral Oil Carbon Atoms per M olecule 12-20 Asphalt More than 34 Kerosene 12-16 Paraffin Wax More than 19 Motor Oil 15-18 Household Lubricating Oil 14-18 Physical State at Room Temperature Appearance 2 Data Table 2 – Viscosity M easurements M aterial Average Time (in sec) for bead to fall Relative Viscosity W ater Trial 1: _________________ Trial 2: _________________ Trial 3: _________________ Average: _________________ M ineral Oil Trial 1: _________________ Trial 2: _________________ Trial 3: _________________ Average: _________________ Kerosene Trial 1: _________________ Trial 2: _________________ Trial 3: _________________ Average: _________________ M otor Oil Trial 1: _________________ Trial 2: _________________ Trial 3: _________________ Average: _________________ Household Lubricating Oil Trial 1: _________________ Trial 2: _________________ Trial 3: _________________ Average: _________________ 3 Data Table 3 -- Density M easurements Average mass of capped tube + bead: _______________________________ Average volume of liquid: _______________________________ Liquid M ass of Capped Tube, Bead, and Liquid (g) Calculated M ass of Liquid (g) (show work) Calculated Density of M aterial (g/mL) (show work) M ass of Sample (g) Volume of Sample (mL) Calculated Density of M aterial (g/mL) (show work) Water Mineral Oil Kerosene Motor Oil Household Oil Solid Paraffin Final Volume (mL): ____________ Initial Volume (mL): ___________ Vol. of Sample (mL): ___________ Asphalt Final Volume (mL): ____________ Initial Volume (mL): ___________ Vol. of Sample (mL): ___________ 4