ASCO Update 6-5-13 - DelMar Pharmaceuticals
advertisement
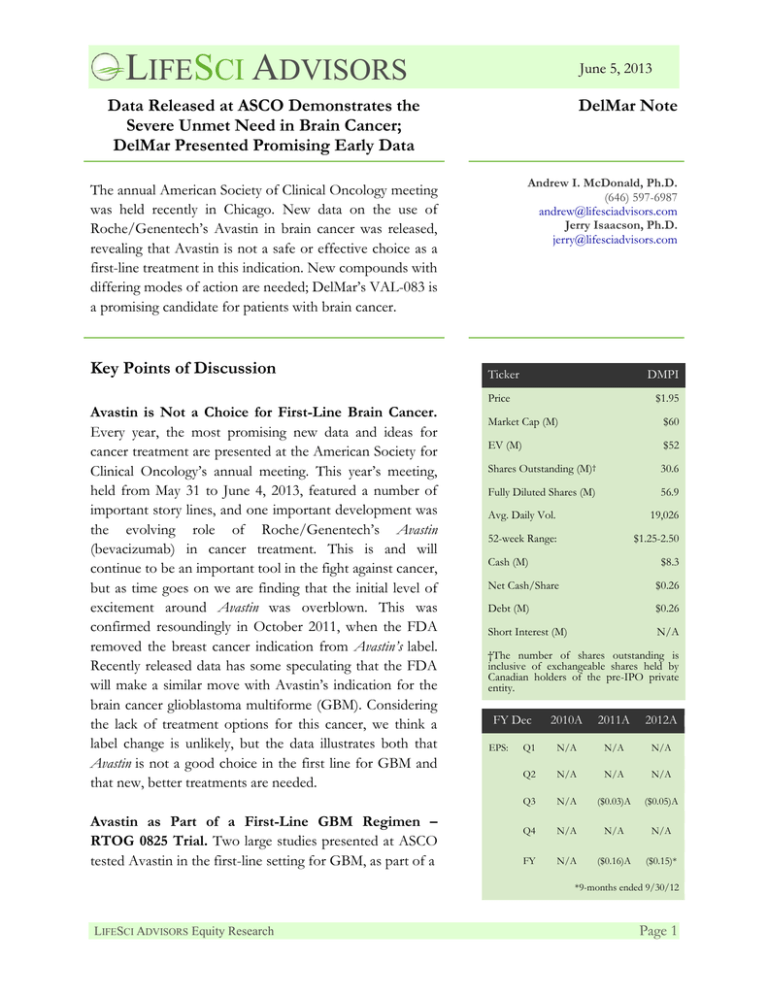
LIFESCI ADVISORS June 5, 2013 Data Released at ASCO Demonstrates the Severe Unmet Need in Brain Cancer; DelMar Presented Promising Early Data DelMar Note Andrew I. McDonald, Ph.D. (646) 597-6987 andrew@lifesciadvisors.com Jerry Isaacson, Ph.D. jerry@lifesciadvisors.com The annual American Society of Clinical Oncology meeting was held recently in Chicago. New data on the use of Roche/Genentech’s Avastin in brain cancer was released, revealing that Avastin is not a safe or effective choice as a first-line treatment in this indication. New compounds with differing modes of action are needed; DelMar’s VAL-083 is a promising candidate for patients with brain cancer. Key Points of Discussion Avastin is Not a Choice for First-Line Brain Cancer. Every year, the most promising new data and ideas for cancer treatment are presented at the American Society for Clinical Oncology’s annual meeting. This year’s meeting, held from May 31 to June 4, 2013, featured a number of important story lines, and one important development was the evolving role of Roche/Genentech’s Avastin (bevacizumab) in cancer treatment. This is and will continue to be an important tool in the fight against cancer, but as time goes on we are finding that the initial level of excitement around Avastin was overblown. This was confirmed resoundingly in October 2011, when the FDA removed the breast cancer indication from Avastin’s label. Recently released data has some speculating that the FDA will make a similar move with Avastin’s indication for the brain cancer glioblastoma multiforme (GBM). Considering the lack of treatment options for this cancer, we think a label change is unlikely, but the data illustrates both that Avastin is not a good choice in the first line for GBM and that new, better treatments are needed. Avastin as Part of a First-Line GBM Regimen – RTOG 0825 Trial. Two large studies presented at ASCO tested Avastin in the first-line setting for GBM, as part of a Ticker DMPI Price $1.95 Market Cap (M) $60 EV (M) $52 Shares Outstanding (M)† 30.6 Fully Diluted Shares (M) 56.9 Avg. Daily Vol. 19,026 52-week Range: $1.25-2.50 Cash (M) $8.3 Net Cash/Share $0.26 Debt (M) $0.26 Short Interest (M) N/A †The number of shares outstanding is inclusive of exchangeable shares held by Canadian holders of the pre-IPO private entity. FY Dec EPS: 2010A 2011A 2012A Q1 N/A N/A N/A Q2 N/A N/A N/A Q3 N/A ($0.03)A ($0.05)A Q4 N/A N/A N/A FY N/A ($0.16)A ($0.15)* *9-months ended 9/30/12 LIFESCI ADVISORS Equity Research Page 1 LIFESCI ADVISORS June 5, 2013 combination that also includes Temodar (temozolomide) and radiation. One of the trials, known as RTOG 0825, was sponsored by the National Cancer Institute and randomized 637 newly diagnosed GBM patients to treatment Temodar and radiation plus either Avastin or placebo.1 The co-primary endpoints for the trial were overall survival (OS) and progression-free survival (PFS). An important secondary endpoint was the impact of O6-methylguanine-DNA methyltransferase (MGMT) methylation on the results.2 The results of this trial did not support the use of Avastin in this patient population. Regarding OS, there was no statistical difference between the control and Avastin-added arms, at 16.1 months versus 15.7 months (p=0.11), and actually numerically favored the placebo arm. The addition of Avastin did have a significant impact on PFS, at 10.7 months versus 7.3 months for placebo (p=0.004) although these results did not meet a pre-determined PFS endpoint. There was an increase in grade 3 or higher adverse events (AEs) for the Avastin arm, including neutropenia, hypertension, and deep vein thrombosis/pulmonary embolism. MGMT status was not predictive of the results, however patients with MGMT methylation trended towards lower OS in the Avastin arm when compared to placebo. Roche AVAglio Study of Avastin in Combination with First-Line Therapy. The second study relating to Avastin in brain cancer was conducted by Roche and was known as AVAglio. 3 The treatment protocol for this trial was very similar to RTOG 0825, with either Avastin or placebo being added to standard of care, which included Temodar and radiation. The co-primary endpoints of PFS and OS were also the same. This trial enrolled 921 patients, and there was a significant increase in PFS of 10.6 months for the Avastin arm versus 6.2 months for the placebo arm (p=0.001)4. These results should be viewed in light of multiple factors. First, it is clear from these data that Avastin is not a good choice for the first-line treatment of GBM. This program has been seen as the most promising development for GBM for many years now, so these disappointing results further highlight the need for better treatments. Second, we see that Avastin has an impact on PFS but no impact on OS. So, as has happened in other indications, the drug is clearly affecting the tumor but this does not translate to a survival benefit. One hypothesized reason for this is that while Avastin affects the tumor for some time, eventually it comes back more aggressively. This was also observed in breast cancer with Avastin. It has also been hypothesized that Avastin, an antibody which can’t enter the brain, may therefore not be able to directly affect the tumor so that any effect it is having is systemic and therefore not as potent. We conclude from this data that a valuable market opportunity and an important unmet medical need remain in GBM. Multiple companies are exploring small molecule as well as antibody 1 http://clinicaltrials.gov/show/NCT00884741 http://meetinglibrary.asco.org/content/111571-132 3 http://clinicaltrials.gov/show/NCT00943826 4 http://meetinglibrary.asco.org/content/114612-132 2 LIFESCI ADVISORS Equity Research Page 2 LIFESCI ADVISORS June 5, 2013 approaches to treating this deadly cancer, and immunotherapies are also being tested. Hopefully more than one of these will emerge as good treatment options for patients with GBM. As we are seeing in prostate cancer, these new therapies should and will also be tested in various combinations. It is possible that a combination of an immunotherapy and a traditional drug such as VAL-083 may prove to be very useful in this indication, but only time will tell. Importance of MGMT Status in Treating GBM. As mentioned above, the RTOG 0825 trial for Avastin included effect of MGMT methylation as a secondary endpoint. MGMT is a DNA repair enzyme that can inhibit the ability of the front-line treatment Temodar to induce cell death in tumor cells of patients with GBM. DelMar’s VAL-083 induces cell death in tumors via a mechanism that is independent of MGMT expression, making VAL-083 a potential treatment for Temodar resistant tumors that express MGMT. Pre-clinical research presented at the 2012 American Association for Cancer Research,5 and also at ASCO, directly addressed the effect of VAL-083 on cells expressing MGMT. The study demonstrated that both temozolomide and VAL-083 can induce cell death in cells that do not express MGMT, but only VAL-083 induces death in MGMT-expressing cells. Therefore, VAL-083 may be an effective therapy for patients who express MGMT but do not respond to Temodar treatment. This raises the possibility of using the MGMT status of patients in the selection criteria for future trials, and also of using VAL-083 in combination with Temodar. DelMar Presented VAL-083 Phase I/II Dose Escalating Study at ASCO. DelMar initiated a Phase I/II clinical trial of VAL-083 in November 2011. The study is an open label, single-arm, safety, tolerability and pharmacokinetic study of VAL-083 for adult patients with recurrent malignant glioma or secondary brain tumors that have failed both Temodar and Avastin, unless contraindicated. The trial protocol calls for administration of VAL-083 on the first 3 out of every 21 days. The primary endpoint for the Phase I/II trial is to establish the maximum tolerated dose (MTD) of VAL-083. Secondary endpoints include monitoring antitumor activity, progression free survival (PFS), and pharmacokinetics of the treatment. Once the MTD is established, additional patients will be enrolled to begin the Phase II portion of the study; radiographic response (the ability of the drug to shrink the tumor as shown brain scans) and PFS will be monitored as the primary endpoints. These endpoints have been used by the FDA in the past to approve products such as Temodar and Avastin for refractory GBM. At the outset of this trial, patients with malignant glioma or secondary brain tumors were treated together. However, DelMar recently announced that the trial would be broken into two arms so these different patient groups may be evaluated separately. The concern is that patients with secondary brain tumors may Hu, K., et al., 2012. VAL083, a novel N7 alkylating agent, surpasses temozolomide activity and inhibits cancer stem cells providing a new potential treatment option for glioblastoma multiforme. American Association for Cancer Research, 2012 Annual Meeting. Abstract #811. 5 LIFESCI ADVISORS Equity Research Page 3 LIFESCI ADVISORS June 5, 2013 be heavily pre-treated and therefore may experience myelosuppression and a smaller MTD than GBM patients. Updated data presented at ASCO revealed that 2/8 patients with GBM experienced a response and 1/6 patients with secondary brain tumors experienced a response. Patients characteristic and responses are summarized in the table below. Figure 1: VAL-083 Patient and Response Summary Source: DelMar ASCO Presentation6 We expect that DelMar will present interim data from this trial at upcoming conferences including, possibly, the Society for Neuro-Oncology (SNO) conference in November. If patient enrollment continues at the current pace, we would not be surprised to see DelMar find the MTD for VAL-083 in time for the SNO meeting. Based on historical precedent, we also anticipate that clinical response rates above 20% may be sufficient for approval of new therapies in refractory GBM. Notably, DelMar’s data achieve this level or response. Upcoming Expected Milestones 2H 2013 – Expected completion of Phase I of the Phase I/II trial of VAL-083 in GBM. 1H 2014 – Potential expansion of VAL-083 Phase II trial to a registration study. About DelMar: More information on DelMar Pharmaceuticals can be found in our Initiation of Coverage Report at http://lifesciadvisors.com/clients/delmar/. 6 http://www.delmarpharma.com/DelMarASCO2013-11x17.pdf LIFESCI ADVISORS Equity Research Page 4 LIFESCI ADVISORS June 5, 2013 DISCLOSURES The material presented in this report is provided for information purposes only and is not to be used or considered as a recommendation to buy, hold or sell any securities or other financial instruments. Information contained herein has been compiled by LifeSci Advisors and prepared from various public and industry sources that we believe to be reliable, but no representation or warranty, expressed or implied is made by LifeSci Advisors, its affiliates or any other person as to the accuracy or completeness of the information. Such information is provided with the expectation that it will be read as part of a broader analysis and should not be relied upon on a stand-alone basis. Past performance should not be taken as an indication or guarantee of future performance, and we make no representation or warranty regarding future performance. The opinions expressed in this report reflect the judgment of LifeSci Advisors as of the date of this report and are subject to change without notice. This report is not an offer to sell or a solicitation of an offer to buy any securities. The offer and sale of securities are regulated generally in various jurisdictions, particularly the manner in which securities may be offered and sold to residents of a particular country or jurisdiction. Securities discussed in this report may not be eligible for sale in some jurisdictions. To the full extent provided by law, neither LifeSci Advisors nor any of its affiliates, nor any other person accepts any liability whatsoever for any direct or consequential loss arising from any use of this report or the information contained herein. No LifeSci Advisors directors, officers or employees are on the Board of Directors of a covered company and no one at a covered company is on the Board of Directors of LifeSci Advisors. Neither the analyst who authored this report nor any of LifeSci Advisors’ directors, officers, employees invest in the securities of the company that is the subject of this report. LifeSci Advisors has been compensated by the company that is the subject of this report for this and future research reports, investor relations services, and general consulting services. LIFESCI ADVISORS Equity Research Page 5



