electrocatalytic oxygen reduction performance of silver nanoparticle
advertisement

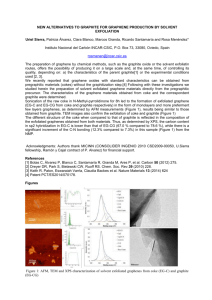
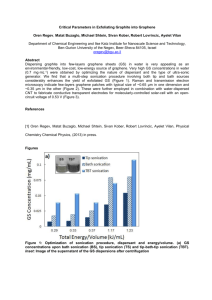
ELECTROCATALYTIC OXYGEN REDUCTION PERFORMANCE OF SILVER NANOPARTICLE DECORATED ELECTROCHEMICALLY EXFOLIATED GRAPHENE Joao Henrique Lopes, Siyu Ye, Jeff T Gostick, Jake E Barralet‡* and Geraldine Merle‡ J. H. Lopes Faculty of Dentistry, McGill University, Montreal, H3A 2B2, (Canada) E-mail: (henriquelopez@gmail.com) Dr. S. Ye Ballard Power Systems, 9000 Glenlyon Parkway, Burnaby, V5J 5J8 (Canada) E-mail: (siyu.ye@ballard.com) Prof. J. T. Gostick Department of Chemical Engineering, McGill University, Montreal, H3A 0C5, (Canada) E-mail : (jeff.gostick@mcgill.ca) 1 Prof. J. E. Barralet Faculty of Dentistry, McGill University, Montreal, H3A 2B2, (Canada) Department of Surgery, Faculty of Medicine, McGill University, Montreal, (Canada) E-mail: (jake.barralet@mcgill.ca) Dr. G. Merle Faculty of Dentistry, McGill University, Montreal, H3A 2B2, (Canada) E-mail: (geraldine.merle@mcgill.ca) SUPPORTING INFORMATION SI. I. Discussion on the TGA data The thermal stability of the as-prepared GRn was investigated by thermal gravimetric analysis (TGA) under air and compared to GRp and GO (Figure 3f). Graphite exhibited one clear step of weight loss at 750 ºC, related to the decomposition of the carbon, while Grn and GO decompose in several steps. GRn curve exhibited a four-step thermal decomposition profile: initially a very small mass loss (< 1%) started before 150 ºC attributable to the loss of residual solvent, followed by a continuous decomposition (~8%) between 150 to 450 ºC related to the pyrolysis of PSS and of the labile oxygen-containing groups. Between 450 to 750 ºC, a substantial weight loss of ~ 21% occurs due to the decomposition of more stable oxygen-containing functional groups and finally the decomposition of the carbon skeleton (> 750 ºC).1, 2, 3, 4 Although GO exhibited the same thermal decomposition profile, the recorded weight losses were considerably higher than 2 GRn (39 and 34%). The absence of an event related to the decomposition carbon skeleton for GO is in agreement with a carbonaceous matrix being throughout being oxidized.2 SI. II. Characterization of graphite and graphene – FTIR spectra Figure S1 shows FTIR absorption spectra of graphite and graphene electrochemically exfoliated. The spectrum of the graphite exhibits two absorption bands at 2928 and 2865 cm-1 representing the C-H stretch vibrations.5 The absence of –OH, C=O, C-OH or C-O bands5, 6 confirms the low oxidation degree of the graphene samples. Figure SI. FTIR spectra of graphite and graphene samples. 3 SI. III. Heterogeneous electron-transfer (HET) The heterogeneous electron-transfer (HET) rate was investigated with cyclic voltammetry in presence of 𝐾! [𝐹𝑒(𝐶𝑁)! ]/𝐾! [𝐹𝑒(𝐶𝑁)! ] (1:1 molar ratio) (Figure SII). Figure SII. Cyclic voltammograms for (a) bare (b) graphite, (c) SDS +6V, (d) PSS +6V, and (e) PSS +10V in N2-saturated 1 M KCl solution containing a total 10 mM concentration of [𝑭𝒆(𝑪𝑵)𝟔 ]𝟑!/𝟒! (1:1 molar ratio). Scans start at +0.7 V. Scan rate, 50 mVs-1. The current density generated at the GRn was greater than the graphite and bare electrodes, indicating a higher electric conductivity for the GRn electrode due to its low defects density and low oxidation degree. The redox peaks (E!/! ) were close to 0.21 V (Table 1) for each electrodes at low potential scan rate (10 mVs-1) and the I! anodic / I! cathodic ratio approaches 1, confirming the reversible redox reaction. The peak-to-peak potential separation (∆E! ) for GRn was quite low (68 mV) close to the ideal value of 59mV. In comparison the ∆E! value at the bare and graphite were 103 mV and 85 mV respectively. The ∆E! value is related to the electron 4 transfer coefficient and the low value for GRn indicates a fast electron transfer. Note that ∆E! for all electrodes were substantially higher than 60/n mV (n=1, 25 ºC), suggesting a type of quasireversible behavior.7, 8, 9 Enhanced electron-transfer kinetics (𝐾!"" ) values were determined from the anodic/cathodic peak separation and the dimensionless parameters 𝜓 and 𝐾!"" .10 We first !! assumed 𝛼 = 0.5 and used the following diffusion coefficients: 𝐹𝑒(𝐶𝑁)!! ! /𝐹𝑒(𝐶𝑁)! , 𝐷! = 7.63𝑥10!! 𝑐𝑚! ∙ 𝑠 !! , 𝐷! = 6.32𝑥10!! 𝑐𝑚! ∙ 𝑠 !! .7 The apparent electron-transfer rate constants, 𝐾!"" , are summarized in Table 1. Having the lowest ∆𝐸! of 68 mV, GRn electrode showed the fastest HET at 8.31 x 10-3 cm s-1. This value was substantially higher than GRp (∆𝐸! = 85 mV and 𝐾!"" = 2.77x 10-3 cm.s-1) and the bare GC (∆𝐸! = 103 mV and 𝐾!"" = 1.38x 10-3 cm.s-1) electrodes. References 1. Fan, Z. J.; Wang, K.; Wei, T.; Yan, J.; Song, L. P.; Shao, B. An environmentally friendly and efficient route for the reduction of graphene oxide by aluminum powder. Carbon 2010, 48 (5), 1686-1689. 2. Stankovich, S.; Dikin, D. A.; Piner, R. D.; Kohlhaas, K. A.; Kleinhammes, A.; Jia, Y.; Wu, Y.; Nguyen, S. T.; Ruoff, R. S. Synthesis of graphene-based nanosheets via chemical reduction of exfoliated graphite oxide. Carbon 2007, 45 (7), 1558-1565. 3. Wang, X. L.; Wen, X. H.; Liu, Z. P.; Tan, Y.; Yuan, Y.; Zhang, P. Rapid and efficient synthesis of soluble graphene nanosheets using N-methyl-p-aminophenol sulfate as a reducing agent. Nanotechnology 2012, 23 (48). 4. Fan, Z. J.; Kai, W.; Yan, J.; Wei, T.; Zhi, L. J.; Feng, J.; Ren, Y. M.; Song, L. P.; Wei, F. Facile Synthesis of Graphene Nanosheets via Fe Reduction of Exfoliated Graphite Oxide. Acs Nano 2011, 5 (1), 191-198. 5. Choi, E. Y.; Han, T. H.; Hong, J. H.; Kim, J. E.; Lee, S. H.; Kim, H. W.; Kim, S. O. Noncovalent functionalization of graphene with end-functional polymers. J Mater Chem 2010, 20 (10), 1907-1912. 5 6. Li, D.; Muller, M. B.; Gilje, S.; Kaner, R. B.; Wallace, G. G. Processable aqueous dispersions of graphene nanosheets. Nat Nanotechnol 2008, 3 (2), 101-105. 7. Tang, L. H.; Wang, Y.; Li, Y. M.; Feng, H. B.; Lu, J.; Li, J. H. Preparation, Structure, and Electrochemical Properties of Reduced Graphene Sheet Films. Advanced Functional Materials 2009, 19 (17), 2782-2789. 8. Ambrosi, A.; Bonanni, A.; Sofer, Z.; Cross, J. S.; Pumera, M. Electrochemistry at Chemically Modified Graphenes. Chemistry – A European Journal 2011, 17 (38), 10763-10770. 9. Lima, F.; Fortunato, G. V.; Maia, G. A remarkably simple characterization of glassy carbon-supported films of graphite, graphene oxide, and chemically converted graphene using Fe(CN)3-6/Fe(CN)4-6 and O2 as redox probes. RSC Advances 2013, 3 (24), 9550-9560. 10. Nicholson, R. S. Theory and Application of Cyclic Voltammetry for Measurement of Electrode Reaction Kinetics. Analytical Chemistry 1965, 37 (11), 1351-1355. 6