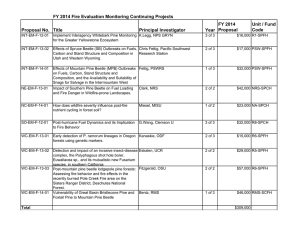
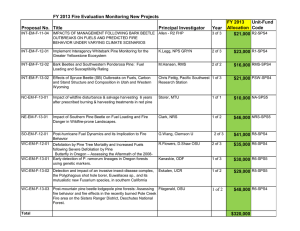
Changing temperatures influence suitability for modeled mountain
advertisement

Click Here JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 111, G02019, doi:10.1029/2005JG000101, 2006 for Full Article Changing temperatures influence suitability for modeled mountain pine beetle (Dendroctonus ponderosae) outbreaks in the western United States Jeffrey A. Hicke,1 Jesse A. Logan,2 James Powell,3 and Dennis S. Ojima1 Received 8 September 2005; revised 17 January 2006; accepted 13 February 2006; published 16 June 2006. [1] Insect outbreaks are significant disturbances in forests of the western United States, with infestation comparable in area to fire. Outbreaks of mountain pine beetle (Dendroctonus ponderosae Hopkins) require life cycles of one year with synchronous emergence of adults from host trees at an appropriate time of year (termed ‘‘adaptive seasonality’’) to overwhelm tree defenses. The annual course of temperature plays a major role in governing life stage development and imposing synchrony on mountain pine beetle populations. Here we apply a process-based model of adaptive seasonality across the western United States using gridded daily temperatures from the Vegetation/Ecosystem Modeling and Analysis Project (VEMAP) over the period 1895–2100. Historical locations of modeled adaptive seasonality overlay much of the distribution of lodgepole pine (Pinus contorta Douglas), a favored host, indicating that suitable temperatures for outbreak occurred in areas of host availability. A range of suitable temperatures, both in the mean and over an annual cycle, resulted in adaptive seasonality. Adaptive seasonality typically occurred when mean annual temperatures were 3°–6°C, but also included locations where mean temperatures were as low as 1°C or as high as 11°C, primarily as a result of variability in winter temperatures. For most locations, years of adaptive seasonality were uncommon during 1895–1993. We analyzed historical temperatures and adaptive seasonality in more detail in three northern forest ecoprovinces. In the Northern and Middle Rockies, areas of adaptive seasonality decreased at lower elevations and increased at higher elevations during warmer periods, resulting in a movement upward in elevation of adaptive seasonality. In contrast, the Cascade Mountains exhibited overall declines in adaptive seasonality with higher temperatures regardless of elevation. Projections of future warming (5°C in the western United States) resulted in substantial reductions in the overall area of adaptive seasonality. At the highest elevations, predicted warmer conditions will result in increases in the area of adaptive seasonality. Our findings suggest that future climate change may alter forest ecosystems indirectly through alteration of these important disturbances. Citation: Hicke, J. A., J. A. Logan, J. Powell, and D. S. Ojima (2006), Changing temperatures influence suitability for modeled mountain pine beetle (Dendroctonus ponderosae) outbreaks in the western United States, J. Geophys. Res., 111, G02019, doi:10.1029/2005JG000101. 1. Introduction [2] Insect outbreaks are important processes in forest ecosystems, affecting succession, biogeochemical cycling, and forest stand characteristics [e.g., Fleming et al., 2002; McCullough et al., 1998]. Insect and pathogen infestations influence an average annual area in the United States of 20.4 million ha, resulting in losses of $1.5 billion [Dale et 1 Natural Resource Ecology Laboratory, Colorado State University, Fort Collins, Colorado, USA. 2 U.S. Department of Agriculture Forest Service, Logan, Utah, USA. 3 Department of Mathematics and Statistics, Utah State University, Logan, Utah, USA. Copyright 2006 by the American Geophysical Union. 0148-0227/06/2005JG000101$09.00 al., 2001]. The United States Department of Agriculture Forest Service reported 1 – 5 million ha of insect- and disease-caused tree mortality during 1997 – 2003 [USDA Forest Service, 2004]. Forest area affected by insect outbreaks is comparable to that burned by fire. In the conterminous United States, fire has burned about 0.4– 2 million ha in contemporary times [Houghton et al., 2000; Leenhouts, 1998]. In Canada, areas of outbreak and fire were similar in magnitude over the period 1920 – 1989 [Kurz and Apps, 1999]. In terms of volume, annual losses during 1982– 1987 resulting from insect-caused tree mortality and growth loss in Canada averaged 52 million m3, 50% more than caused by fire [Hall and Moody, 1994]. [3] Recent studies have implicated climate, specifically increasing temperature, as a major factor in insect outbreaks G02019 1 of 12 G02019 HICKE ET AL.: CLIMATE CHANGE AND INSECT OUTBREAKS throughout western North America [Carroll et al., 2004; Fastie et al., 1995; Logan and Powell, 2001]. Surface temperatures are projected to warm in coming decades [Intergovernmental Panel on Climate Change (IPCC), 2001]. These climate changes will likely influence the distribution and outbreak frequency of many insect populations [Fleming, 2000; Logan et al., 2003; Williams and Liebhold, 2002]. [4] Bark beetles (members of the family Scolytinae) constitute a class of insects distributed throughout North American forests that invade host tree species to feed and reproduce, typically in dead or dying host trees. Most of these beetles colonize twigs and branches without appreciable harm to the living tree, or they attack dead or dying trees. However, a few species (such as mountain pine beetle, Dendroctonus ponderosae Hopkins), termed primary or aggressive bark beetles, attack the main stem of living trees, killing the host tree in the process. [5] Vast areas of bark beetle infestation in western North America have occurred recently or are ongoing. In recent decades, extensive outbreaks have occurred on the Kenai Peninsula, Alaska (500,000 ha [Holsten et al., 2001]), in the southeastern United States (3 million ha [USDA Forest Service, 2004]), and across the western United States (4.3 million ha [USDA Forest Service, 2004]). Projections of outbreaks forecast large areas of damage. The Western Bark Beetle Report [USDA Forest Service, 2002] states that ‘‘over the next 15 years, 8.5 million ha of western forests are at high risk of experiencing significant tree mortality caused by bark beetles.’’ [6] Mountain pine beetle affects the most area of any bark beetle in western North America. The area attacked by mountain pine beetle in Canada has risen to 7 million ha in 2004 [Ministry of Forests, 2005]. Forest area infested by mountain pine beetle in the United States exceeded 1.5 million ha in the early 1980s. After a low of 150,000 ha in the late 1990s, the area infested in the United States has risen in recent years and is currently at almost 1 million ha [USDA Forest Service, 2004]. [7] The majority of the mountain pine beetle life cycle is spent as larvae feeding in the phloem tissue of host pine trees [Amman et al., 1990]. This feeding activity eventually girdles and kills successfully attacked trees (i.e., trees that cannot resist infestation) [Amman and Cole, 1983; Furniss, 1997]. Most western pines are suitable hosts for this insect; ponderosa pine, Pinus ponderosa Lawson, and lodgepole pine, Pinus contorta Douglas, are among the major host species [Amman et al., 1990]. After completing multiple life stages over the course of 1 or 2 years, adult beetles emerge from the host tree to attack new hosts [Amman et al., 1990]. [8] Because host tree species have evolved chemical and physical defenses, mountain pine beetles mass attack trees to overcome these defenses [Amman et al., 1990; Raffa and Berryman, 1987]. Synchronous emergence of adult beetles from hosts is therefore required to maintain the outbreak [Amman, 1985]. Furthermore, emergence should occur at an appropriate time of year that avoids mortality from early season freezing yet allows full ovipositional development before lethal fall or winter temperatures [Amman, 1973]. Finally, outbreaks occur when the beetle life cycle completes in 1 year (termed ‘‘univoltine’’). This combination of G02019 univoltine conditions with synchronous emergence at an appropriate time of season is termed ‘‘adaptive seasonality’’ [Logan and Powell, 2001]. [9] In contrast to many insect species, the mountain pine beetle lacks an obligatory physiological state for synchronizing development (e.g., diapause) [Logan and Bentz, 1999; Powell and Logan, 2005]. Instead, life stage development is under direct temperature control. A processbased model of mountain pine beetle phenology has been developed that incorporates the essential details of the mountain pine beetle life cycle [Bentz et al., 1991; Logan et al., 1995; Logan and Powell, 2001; Powell et al., 2000]. For a given annual cycle of daily temperatures, this model estimates whether climate conditions result in adaptive seasonality. The current model was parameterized using developmental data from beetles collected from lodgepole pine in central Idaho. The model, therefore, characterizes the seasonality of populations found in the northern US Rocky Mountains; implications of this assumption are discussed in section 4. The model has successfully predicted outbreaks in lodgepole pine in central Idaho [Powell and Logan, 2005]. [10] The objective of this study was to apply the adaptive seasonality model to broad regions across the western United States over long time periods. With a data set of historical and projected temperatures developed for the conterminous United States, we evaluated the spatial patterns of adaptive seasonality. In addition, we analyzed how future climate change will affect adaptive seasonality and therefore forest ecosystems. 2. Methods 2.1. Adaptive Seasonality Model [11] Adaptive seasonality modeling simulates the mountain pine beetle life cycle. The model has been discussed in detail in previous studies [Bentz et al., 1991; Logan and Amman, 1986; Logan et al., 1995; Logan and Powell, 2001]; here we briefly describe the method used. The basic modeling framework considers that the developmental rate, r(T), is the inverse of the time required to complete a particular life stage at temperature T. Life stage developmental rates (rj) were determined from laboratory measurements for Idaho beetle populations in lodgepole pine bolts. The functional forms and parameters for developmental rates of all eight life stages of the mountain pine beetle are given by Logan and Amman [1986] and Bentz et al. [1991]. The developmental index, aj, in stage j is the fraction of the jth life stage completed at any particular time by the median individual in the population, and is related to the developmental rate by a differential equation: d aj ðt Þ ¼ rj ½T ðt Þ: dt ð1Þ Life stage j begins at time tj1, which is the time of completion of the previous life stage, and defines the initial condition of equation (2), 2 of 12 aj t ¼ tj1 ¼ 0: ð2Þ HICKE ET AL.: CLIMATE CHANGE AND INSECT OUTBREAKS G02019 By integrating equation (1) from this initial condition, we find the developmental index, aj, at time t, aj ðt Þ ¼ Zt 0 0 rj ½T ðt Þdt : ð3Þ tj1 Life stage j finishes at time tj, at which aj = 1, aj t ¼ tj ¼ 1: ð4Þ By evaluating equation (3) numerically and finding the earliest t for which the cumulative development is 1, it is possible to determine the developmental time spent in stage j in a variable temperature environment. Logan and Powell [2001] illustrate these concepts graphically and with additional detail. [12] Beginning with an egg laid on an arbitrary start date and progressing though the other life stages, the adaptive seasonality model numerically integrates equation (3) using annual temperature cycles, thereby computing the time required to complete each life stage. Each initial oviposition date is thus associated with a particular emergence date; the output of the model is the total number of days required to complete all life stages, starting on a given oviposition date. The model is iterated from generation to generation, resulting in a sequence of oviposition dates. As discussed by Powell and Logan [2005], a natural consequence of this model structure is that every dynamic sequence of ovipositional dates is attracted to the same life cycle regardless of initial oviposition date. If this cycle is exactly 1 year long (and therefore consists of a single, fixed point), the population is univoltine, and the univoltine fixed point attracts and therefore synchronizes the emergence of the entire beetle population. Multiple generations of beetles are modeled for each annual cycle of temperature to evaluate the convergence of the results. [13] Three conditions must be met for adaptive seasonality: Development must complete in a single year, populations must be able to synchronize their emergence, and emergence must occur at a reasonable time of year. The first two conditions are satisfied only if a univoltine fixed point exists, as described above. The third condition ensures that eggs are laid before vulnerability to freezing becomes an issue, and guarantees that cold-hardened larval instars occur during the coldest season. This condition is checked by determining if the univoltine fixed point appears in an appropriate seasonal window (July through August). It is important to recognize that temperatures may be too high to support adaptive seasonality as well as too low [Logan and Powell, 2001]; as shown later, this turns out to be an issue with projected climate change. [14] Logan and Powell [2004] and Powell and Logan [2005] used this model to explore climatic control of an ongoing outbreak of mountain pine beetle in Stanley, Idaho. Modeling results since the summer of 1995– 1996 have predicted univoltine, synchronous life cycles for mountain pine beetles (i.e., adaptive seasonality). These predictions have been accompanied by an rapid increase in outbreak area as revealed by aerial detection surveys [Logan and Powell, 2004]. The last mountain pine beetle outbreak in the G02019 Stanley, ID area occurred in the late 1920s to early 1930, also an unusually warm period [Logan and Powell, 2001]. 2.2. VEMAP Temperatures [15] The adaptive seasonality model is driven by hourly temperatures through the course of a year. Daily minimum and maximum temperatures are sufficient for deriving a diurnal temperature cycle, which is then used in the model. Few spatially extensive records of daily temperatures are available at sufficiently fine temporal resolution to drive the model. In this study, we used temperatures from the Vegetation/Ecosystem Modeling and Analysis Project (VEMAP [Kittel et al., 2004]). [16] VEMAP temperatures are based on measurements for the period 1895 – 1993; 1993 was the last year for which observations were available for the development of the data set. Observational input data sets to VEMAP include the United States Historical Climate Network (USHCN), cooperative networks, and the SNOTEL (snowpack telemetry) data set. The USHCN data were selected for their high quality (i.e., minimal biases). However, this data set is limited in spatial extent, and as a result the cooperative network and SNOTEL data sets were utilized to fill in spatial and temporal gaps in the VEMAP data set. The SNOTEL data in particular were useful for specifying higher elevation behavior. VEMAP temperatures were generated with the goal of completeness in space and time and as a result may be limited in their ability to detect long-term trends [Kittel et al., 2004]. [17] General circulation model climate scenarios were downscaled to the VEMAP grid for the period 1994 – 2100 [VEMAP Members, 1995]. These scenarios resulted from runs using the Canadian Climate Center (CGCM1; 3.75° by 3.75°, 19 vertical levels) and the United Kingdom Hadley Centre (HADCM2; 2.5° by 3.75°, 10 vertical levels) models. Both models included increasing atmospheric CO2 and sulfate concentrations. Measured concentrations were used until 1993; afterward, both atmospheric constituents increased at an idealized rate of 1% per year. This scenario lies near the middle of the range of emission scenarios used in the 2001 Intergovernmental Panel on Climate Change (IPCC) assessment in terms of CO2 concentrations in 2100 [IPCC, 2001]. Model output was interpolated to the VEMAP grid and adjusted for high-resolution topography. For this study, we report results using the CCC model. Temperatures in 2100 predicted by the HADCM2 in the western United States were 1.5°C cooler than those from the CCC model, but similar qualitative results of adaptive seasonality were obtained with the HADCM2 model. Both models tended to predict a global mean temperature increase slightly larger than the mean of a variety of models used in the most recent IPCC assessment [IPCC, 2001]. [18] The spatial resolution of VEMAP data sets is 0.5°. Although relatively coarse, this resolution accounts for some topographic changes that are important when assessing temperature variability across the mountainous western United States. Topographic effects on temperature were accounted for through the use of a model that includes elevation for interpolation of climate variables (ParameterRegression on Independent Slopes [Daly et al., 1997]). [19] Hourly temperatures are required by the adaptive seasonality model. Temporal downscaling of monthly 3 of 12 G02019 HICKE ET AL.: CLIMATE CHANGE AND INSECT OUTBREAKS G02019 mountain pine beetle outbreaks [USDA Forest Service, 2004]: the Northern Rockies (total area is 10 million ha), the Middle Rockies (21 million ha), and the Cascades (14 million ha). For each year, annual temperatures from all VEMAP grid cells within each ecoprovince were used to compute the mean annual temperature. Although daily temperatures drove the adaptive seasonality model, annual temperatures were plotted to convey long-term variability and trends. Area of adaptive seasonality within the ecoprovince was calculated as the sum of the area of VEMAP grid cells with adaptive seasonality for the given year. [22] Grid cells within each ecoprovince were divided into low and high elevation bins to allow temporal analysis of differences in temperature and adaptive seasonality behavior across an elevation gradient. We selected median elevations within each ecoprovince to separate low and high bins; these elevations were 1300 m (Northern Rockies), 2000 m (Middle Rockies), and 1000 m (Cascade). 3. Results Figure 1. Locations (in black) that had at least 1 year of adaptive seasonality from 1895– 1993. White lines indicate distribution of lodgepole pine, a favored host of mountain pine beetle; note the generally good agreement with areas of adaptive seasonality. Thick white circles at 47.25°N, 121.75°W (Washington), 38.75°N, 105.25°W (Colorado), and 47.75°N, 116.25°W (Idaho) indicate locations shown in Figure 4. Three of Bailey’s ecoprovinces (Cascade, Northern Rockies, and Middle Rockies) displayed in Figure 5 are indicated with hatching. Also, 43.9°N, 114.8°W indicates location of mountain pine beetle population that was used to parameterize the model. Spatial resolution of grid cells is 0.5°. State names are indicated by abbreviations. means to hourly values was accomplished in two steps. First, the VEMAP project utilized a modified version of WGEN to generate daily minimum and maximum values [Kittel et al., 2004]. WGEN parameterizes the means and variances of values using daily and monthly observations [Parlange and Katz, 2000]; VEMAP used 526 high-quality stations during the period 1930 – 1989 to develop these parameterizations [Kittel et al., 2004]. [20] Hourly temperatures for use in the adaptive seasonality model were calculated from VEMAP daily minimum and maximum temperature using a sawtooth pattern. The sawtooth pattern was found to predict observed voltinism better than a variety of other methods, including sinusoidal, sinusoidal during the day with an exponential lag at night, square, and Newton’s Law of Cooling with a radiant flux term (p < 0.01 using a chi-squared analysis) [Hanks et al., 2001]. Minimum temperatures were assumed to occur at sunrise, and maximum temperatures were assumed to occur 3 hours before sunset. Temperatures were linearly interpolated between these times and values. 2.3. Ecoprovince Results [21] Average temperatures and area of adaptive seasonality were computed for three of Bailey’s ecoprovinces [Bailey and Hogg, 1986] that have experienced extensive 3.1. Presence of Adaptive Seasonality [23] To indicate locations across the western United States that had adaptive seasonality, we mapped cells for which we estimated at least one year of adaptive seasonality during the historical time period (1895 –1993) (Figure 1). Thermally suitable locations for outbreak occurred across the northern and central mountain ranges of the western United States, including the Cascades, the Sierra Nevada, and the Rocky Mountains. We also plotted the distribution of a favored host, lodgepole pine [Little, 1971]. The modeled adaptive seasonality occurred in most regions where lodgepole pine is found, even in the very localized region around the Black Hills of South Dakota (44°N, 103°W). In addition, the modeled locations of climate suitability in the United States was in good agreement with a map of the probable distribution of mountain pine beetle [Amman et al., 1990]. [24] Notable differences compared with the lodgepole pine distribution occurred in the northwest corner of Wyoming (44°N, 104°W) and in the Southern Rockies of Colorado (40°N, 107°W), where modeled adaptive seasonality occurred at lower elevations, but not at the elevations of lodgepole pine. Outbreaks have occurred or are ongoing in lodgepole pine in both locations, however [Despain, 1990; USDA Forest Service, 2003]. Another known outbreak area where the model did not predict adaptive seasonality is in ponderosa pine in the mountains of Arizona (35°N, 111°W) [Furniss and Carolin, 1977]. The model predicted adaptive seasonality in regions where lodgepole pine is not found, most significantly in southern Utah (38°N, 112°N), where ponderosa pine is found, and northeastern Nevada (41°N, 116°W). Modeled adaptive seasonality differed in some details from the map of potential mountain pine beetle range as defined by Amman et al. [1990], most notably in Arizona and New Mexico. [25] The total number of years of adaptive seasonality during 1895– 1993 varied considerably, ranging from 1 to 80. No spatial pattern was apparent across the western United States (Figure 2a) since both elevation and latitude/longitude played roles in determining adaptive seasonality. The cumulative distribution of years of adaptive 4 of 12 G02019 HICKE ET AL.: CLIMATE CHANGE AND INSECT OUTBREAKS G02019 Figure 2. (a) Number of years of adaptive seasonality during the 99 years between 1895 and 1993. (b) Cumulative distribution of number of years of adaptive seasonality. A large number of locations, scattered throughout the region, had only a few years of adaptive seasonality, implying that the temperatures conditions were usually unfavorable for outbreak. seasonality revealed that the majority of years within most grid cells were climatically unsuitable for mountain pine beetle outbreak. For example, 50% of the grid cells had <20 years when temperatures produced adaptive seasonality, and 80% of the grid cells had <55 years, just over half of the total number of years between 1895 and 1993. 3.2. Variability in Temperatures Resulting in Adaptive Seasonality [26] A range of annual cycles of temperature resulted in adaptive seasonality. Plotting the distribution of mean annual temperatures of years and locations revealed that 5 of 12 G02019 HICKE ET AL.: CLIMATE CHANGE AND INSECT OUTBREAKS Figure 3. Frequency distribution of mean annual temperatures for years and grid cells with adaptive seasonality. Although the peak is around 5°C, the range extends from 1° – 11°C. most cases of adaptive seasonality occurred when mean temperatures were 4° – 5°C (Figure 3). However, years when mean temperatures were as low as 1°C or as high as 11°C also sometimes resulted in adaptive seasonality. The slight increase in the distribution at 9° – 10°C was associated with locations in northwestern Washington, where warmer winter conditions resulted in adaptive seasonality (see Figure 4). [27] The mean annual cycle of temperatures at three locations that experienced adaptive seasonality highlights this variability (Figure 4). Washington temperatures were relatively high in winter. In contrast, Colorado winter temperatures were lower by 9°C, though summer temperatures were similar. Idaho temperatures were intermediate in winter but higher in summer by 2°C. Substantial variability in the annual cycle of temperatures that resulted in adaptive seasonality also occurred within years at one location (not shown). 3.3. Historical Behavior for Three Ecoprovinces [28] Adaptive seasonality during 1895 – 1993 occurred in many locations within the three selected ecoprovinces (Cascades, Northern Rockies, Middle Rockies) (Figure 1). Mean temperatures exhibited similar behavior across the ecoregions and elevations (Figure 5). Warm periods occurred between the late 1930s and the early 1940s, with recent years showing large increases in temperature (except for higher elevation Middle Rockies). Although longer-term temperature patterns in the VEMAP data set may be contaminated by the inclusion of lower quality stations, comparisons with high quality USHCN stations revealed similar patterns (http://www.nrel.colostate.edu/jhicke/ climate_data). The area of adaptive seasonality was negatively correlated with temperature at lower elevations in the Rockies ecoprovinces (Figures 5a, 5c, 6a, and 6c), but positively correlated at higher elevations (Figures 5b, 5d, 6b, and 6d). In the Cascades ecoprovince, area was negatively correlated with temperature at both lower and higher elevations (Figures 5e, 5f, 6e, and 6f). G02019 3.4. Effects of Future Warming [29] The scenario of future climate change by the CCC general circulation model and downscaled to the VEMAP grid predicts warming in the western United States of 5°C by 2100 (Figure 7, bottom). We found that these increases will reduce the overall area of adaptive seasonality as temperatures become too high in many areas to support adaptive seasonality. By the end of the time period (2100), we estimated that only 1 million ha in the western United States will have thermally suitable conditions for outbreak, compared to 20– 30 million ha historically. [30] Binning temperatures and area by elevation revealed different patterns. Over the western United States, reductions in area of adaptive seasonality at the lowest elevations (0– 1500 m; Figure 7) occurred after 1980 and will continue into the future. Between 1500 and 2500 m, we forecast that the decline will begin about 2025. For the highest two elevation bins, increases will occur initially (1994 forward) as temperatures increase. However, the area of adaptive seasonality will then decrease after 2040 in the 2500- to 3000-m bin, and after 2060 for the highest elevation bin. Our results indicated that the declines in area for the upper three elevation bins will occur when mean annual temperatures were 5° – 6°C. The mean annual temperature of locations of adaptive seasonality within the lowest elevation bin was historically about 7°C, and therefore projected temperature increases after 1994 (in fact, slightly before) will drive these areas out of adaptive seasonality immediately. 4. Discussion 4.1. Spatiotemporal Patterns of Adaptive Seasonality [31] The overlapping adaptive seasonality and lodgepole pine distributions imply that mountain pine beetle and Figure 4. Mean annual cycle of temperature for the three locations, Washington (WA; elevation is 515 m; solid curve), Idaho (ID; 921 m; dashed-dotted curve), and Colorado (CO; 2662 m; dashed curve), circled in Figure 1. Only years of adaptive seasonality were included. Note the wide range of annual cycles, primarily resulting from variability in winter temperatures, associated with adaptive seasonality. 6 of 12 G02019 HICKE ET AL.: CLIMATE CHANGE AND INSECT OUTBREAKS G02019 Figure 5. Anomalies of temperature (black) and area of adaptive seasonality (gray) relative to the 1895 –1993 means for three ecoprovinces. (left) Lower elevations for each ecoprovince. (right) Higher elevations. Thick lines represent time series smoothed with a 20-point Lowess smoother. lodgepole pine occupy the same bioclimatic envelope. Persistence of lodgepole pine across a landscape requires periodic disturbance [Burns and Honkala, 1990; Clements, 1910; Despain, 1990; Pfister and Cole, 1985]. Bark beetles act as disturbances to these forests, and at various times following outbreak, can enhance the potential for fire [Fleming et al., 2002; Stuart et al., 1989; Turner et al., 1999], another forest disturbance. Outbreak and fire create conditions favorable for lodgepole pine regeneration instead of other species [Burns and Honkala, 1990]. [32] The variability in mean annual temperatures and mean annual cycles of temperature that led to adaptive seasonality primarily resulted from a range of winter temperatures. During these cold conditions, life stage development is slow, and thus sensitivity to temperature variability is reduced. From late spring throughfall, temperatures were more similar among the sites analyzed (Figure 4). The range of mean conditions resulting in adaptive seasonality demonstrates the necessity of process-based models of insect phenology for assessing climate effects on outbreaks. A simple analysis of mean annual temperature, while instructive (as in some of the figures in this paper), may miss some important dynamics. [33] Whether adaptive seasonality increased or decreased in response to interannual variability in warming depended on the current thermal regime. At warmer locations (lower elevations or coastal mountains), we found that contemporary temperatures were suitable for adaptive seasonality, whereas higher elevations or locations in the interior of the continent were too cold. Thus the higher mean temperatures of the Cascades supported adaptive seasonality at all elevations, and increases in temperature reduced the area of adaptive seasonality. In contrast, in the colder Rockies, the locations of adaptive seasonality moved upslope in response to warming. [34] Uncertainty about future temperature projections may lead to increased or decreased warming relative to the CGCM1 results we used here. The response of adaptive seasonality to this range can be gauged from our findings. Additional warming beyond that predicted by the CGCM1 model will result in reductions to the area of adaptive seasonality at the highest elevations, where we predict suitable conditions will still occur in 2100. Decreased future warming will lead to greater area of adaptive seasonality (i.e., lower reductions in area). For instance, the temperature increase predicted by HADCM2 of 3.5°C instead of 5°C would result in about 3 million ha of adaptive seasonality in 2100, corresponding to the year 2075 in the CGCM1 runs. [35] Our predicted sensitivity of outbreaks to climate change are in agreement with a large body of existing 7 of 12 G02019 HICKE ET AL.: CLIMATE CHANGE AND INSECT OUTBREAKS G02019 Figure 6. Anomalies of temperature and area of adaptive seasonality relative to the 1895 – 1993 means for three ecoprovinces (same as plotted in Figure 5). (left) Lower elevations for each ecoprovince. (right) Higher elevations. In the lower elevation Rocky Mountain ecoprovinces as well as in the Cascades, temperatures and adaptive seasonality are negatively correlated. In the higher elevation Rocky Mountains, the correlation is positive. Correlations are significant (p < 0.05) when r = 0.17. literature on a number of insect species and forest types [e.g., Bale et al., 2002; Carroll et al., 2004; Fastie et al., 1995; Fleming, 2000]. Specifically, Williams and Leibhold [2002] developed a statistical model that related climate variables to mountain pine beetle outbreak in the Pacific Northwest to explore changes in MPB range in response to prescribed step changes in temperature. These authors found a positive correlation between temperature and MPB distribution, and that as temperatures increased by 2°, 4°, and 8°C, the MPB range decreased in area and moved upward in elevation. Our results using a process-based model with transient climate scenarios over the entire western United States confirmed these findings. 4.2. Modeling Considerations [36] Different populations of mountain pine beetle have different developmental rates [Bentz et al., 2001]. More southern populations are adapted to higher temperatures through reduced developmental rates, thus requiring more thermal input to complete the life cycle. Our study utilized rates from Idaho populations, and this assumption may have led to the unsuccessful prediction of adaptive seasonality in areas of known outbreak in some southern areas. Related to this variability, we assumed that mountain pine beetles do not evolve in response to warmer conditions over the time period of this study. [37] Two sources of temperature variability were not represented here. Differences between air and phloem temperatures will influence beetle populations; our results here used air temperatures. A comparison of Figure 1 with a model run using a conversion to phloem temperatures resulted in little change in the overall regional patterns. In addition, spatial variability in microclimate within a 0.5° grid cell was not captured by the VEMAP temperatures, and therefore some areas actually suitable for outbreak within a grid cell could not be accurately modeled. [38] As with any model, the predictions of the adaptive seasonality model do not match expectations over the full range of possibilities (e.g., lack of adaptive seasonality in areas of known outbreaks). In addition to the variability in insect populations and temperatures discussed above, the model framework may be too constraining. The adaptive seasonality model predicts that annual temperatures are either suitable or not for outbreak for the median individual in a MPB population. This binomial modeling construct ignores variability in developmental rates and emergence (and thus timing of attack) across the population that occurs in reality. Further research will apply a process model that 8 of 12 G02019 HICKE ET AL.: CLIMATE CHANGE AND INSECT OUTBREAKS G02019 Figure 7. Mean annual temperature (black) and area of adaptive seasonality (gray) from 1895– 2100 (bottom) for all areas and (top four plots) four elevation bands in the western United States: 0 –1500 m, 1500 –2500 m, 2500– 3000 m, 3000 – 3500 m. Note changes in y axis ranges for area of adaptive seasonality. Temperatures were computed using only grid cells with at least 1 year of adaptive seasonality. The shift from historical to projected temperatures in 1993 is indicated by the vertical dotted line. Projected warming leads to reduced area of adaptive seasonality except at the highest elevations, which have increases. considers this population variability, and will investigate other sources of temperature to improve the predictions of temperature effects on population levels. 4.3. Additional Factors to Predicting Outbreaks [39] In this paper we discuss adaptive seasonality, or the suitability of temperatures for mountain pine beetle outbreaks. However, adaptive seasonality is only one of several conditions that need to be met for an outbreak to actually occur. Other contributors include stand characteristics and other weather conditions. Presence of host species is an obvious requirement. Favored hosts of mountain pine beetle include lodgepole and ponderosa pine, and to a lesser extent other pine species. Stand conditions that influence tree vigor and therefore resistance to attack also contribute to susceptibility [Amman and McGregor, 1985]. Shore and Safranyik [1992] identified three stand characteristics that increase susceptibility of a stand to outbreak: sufficiently large basal area of host species, stand age >60 –80 years, and interme- 9 of 12 G02019 HICKE ET AL.: CLIMATE CHANGE AND INSECT OUTBREAKS diate stem density. The current structure of western conifer forests, with a higher proportion of older stands [Ferry et al., 1995; Taylor and Carroll, 2004], may play a major role in driving recent infestations. Other weather variables used to assess outbreak potential include minimum winter temperatures that may cause beetle mortality, August temperatures associated with beetle flight, and drought indices that may reflect lowered tree resistance through reduced soil moisture [e.g., Carroll et al., 2004]. [40] Several other bark beetles are present at the southern edge of the mountain pine beetle distribution, such as the roundheaded pine beetle (Dendroctonus adjunctus Blandford), the larger Mexican pine beetle (Dendroctonus mexicanus Dietz), and the southern pine beetle (Dendroctonus frontalis Zimmermann). In addition, southern populations of MPB have lower developmental rates and thus are better adapted to warmer conditions. These MPB populations and different Dendroctonus species may expand their range northward to fill locations that we predict will become unsuitable for mountain pine beetle. 4.4. Implications for Forest Ecosystems [41] At lower elevations, the future reduction in adaptive seasonality area suggested by this study will have important implications for forest ecosystems. Removal of a significant disturbance may shift more stands from intermediate successional stages to climax stages. For example, in the Middle Rockies, the reliance of lodgepole pine on disturbance for regeneration implies that decreased outbreak frequency resulting from climate change (in the absence of changes in fire regime) may result in shifts to subalpine fir [Despain, 1990]. Changing disturbance regimes and increases in stand ages will impact biogeochemical cycling, including net primary production [e.g., Hicke et al., 2003] and net carbon fluxes [e.g., Kurz and Apps, 1999]. Responses of fire and outbreak disturbance to climate change and their interaction may substantially alter forest ecosystems. For instance, warming may lead to increased fire frequency, which in turn may lead to reduced stand ages across the landscape. Since older stands are more susceptible to mountain pine beetle outbreak [Shore and Safranyik, 1992], this reduced stand age may result in decreases in outbreak. [42] Our predictions of increased area of adaptive seasonality at higher elevations in coming decades will increase stresses to high-elevation pine ecosystems. In particular, whitebark pine (Pinus albicaulis Engelm.) grows at high elevations in the western United States, and has a life history that makes its populations particularly susceptible to elimination by bark beetles. Although mountain pine beetle outbreaks have occurred in these forests in the past, these were episodic events, affecting whitebark pine stands for limited time periods (e.g., the warm periods in 1930 – 1940 seen in Figure 5 [Logan and Powell, 2001]). This contrasts with lodgepole pine, which lives in many locations in thermal conditions suitable for outbreak (Figure 2). Whitebark pine appears to be particularly vulnerable to mountain pine beetle depredation for a variety of reasons. The relatively thick phloem provides an improved food resource as compared to lodgepole pine [Amman, 1982]. Additionally, whitebark pine may be less well protected by chemical defenses than lodgepole pine, perhaps in response G02019 to a lack of significant adaptive pressure from bark beetles (J. A. Logan and J. A. Powell, unpublished data, 2005). Our model estimates of increased adaptive seasonality at high elevations are particularly alarming for this sensitive ecosystem [Logan and Powell, 2001]. In recent years, widespread outbreaks of mountain pine beetles have occurred across the northern U.S. Rocky Mountains concurrent with an observed unprecedented warming [Powell and Logan, 2005]. If this trend continues, these communities, already at risk from widespread mortality from an introduced pathogen (white pine blister rust), will face an increasingly uncertain future [Tomback, 2002]. In particular, the substantial effort that has gone into establishing blister rust resistant strains of whitebark pine could be sidetracked by mountain pine beetle outbreaks that are not affected by disease resistance. [43] Our results forecast reduced area of adaptive seasonality in the western United States as a result of higher projected temperatures. However, the distribution of lodgepole pine extends farther north into British Columbia. The northern range limit of mountain pine beetle currently is limited by climate, not host availability (i.e., lodgepole pine exists well north of the beetle range limit) [Carroll et al., 2004]. Recent warming has been implicated as responsible for the dramatic expansion in outbreak area in British Columbia, with continued northward migration forecast with increasing temperature [Carroll et al., 2004]. Logan and Powell [2001] hypothesized that with sufficient warming, range expansion of mountain pine beetle in the Canadian Rockies may lead to invasion into stands of a possible host, jack pine (Pinus banksiana Lamb.), with widespread distribution across Canada. 5. Summary and Conclusions [44] A process-based model that predicts whether input temperatures are suitable for outbreaks of mountain pine beetle was run with gridded temperatures across the western United States. Temperatures and adaptive seasonality were analyzed for historical (1895 – 1993) and projected (1994– 2100) time periods as defined in the VEMAP data set. Temporally, adaptive seasonality occurred less frequently rather than more frequently across the region, with 80% of the grid cells with any adaptive seasonality having fewer than 55 years of climate suitability (out of 99). We found that adaptive seasonality occurred across a range of mean annual temperatures and mean annual cycles of temperature. This range resulted from variability in winter temperatures, when developmental rates are low and therefore life stage development is less sensitive to temperature variability. [45] Model estimates of adaptive seasonality resulted in areas that matched the distribution of a favored host, lodgepole pine, as well as agreeing with a potential range map of mountain pine beetle. This finding was obtained with the parameterization of the model using data from central Idaho, and is an indication that developmental rate estimates for this population are probably characteristic for most mountain pine beetle populations occurring in the U.S. distribution of lodgepole pine. [46] From the mid-1970s to the early 1990s, historical patterns in the Northern and Middle Rockies ecoprovinces 10 of 12 G02019 HICKE ET AL.: CLIMATE CHANGE AND INSECT OUTBREAKS revealed an upward shift of adaptive seasonality to higher elevations in periods of higher temperatures with a concurrent decrease in adaptive seasonality at lower elevations. These shifts are associated with an increase in outbreak reported in novel environments (i.e., high-elevation whitebark pine). In contrast, in the warmer Cascades, increases in temperature resulted in decreases in adaptive seasonality at all elevations. [47] Our modeled results suggest that projected warming across the western United States will lead to a decrease in overall modeled area of adaptive seasonality. At higher elevations, we estimate that increases in the area of adaptive seasonality will occur early in the twenty-first century, but as temperatures will continue to rise, conditions will become too warm to support mountain pine beetle outbreaks and area will decline. [48] Adaptive seasonality modeling is a first step toward process-based predictions of mountain pine beetle outbreak. Additional processes needed to predict outbreaks include mapping host species stand characteristics, including distribution, basal area, age, and density, as well as including other effects of climate on insect populations. Migration of other populations of MPB with different developmental rates [Bentz et al., 2001] or other bark beetle species in response to warming may fill in where we predict reductions in outbreaks will occur. Future research to incorporate these processes as well as to develop process-based modeling of additional bark beetle species will allow us to better understand the effects of climate change on forests in western North America. [49] Consequences of changing outbreak regimes range from effects at local scales (such as bark beetles as invasive species in high elevation pine ecosystems [Logan and Powell, 2001]) to effects at regional scales (such as the northward migration of beetles into jack pine ecosystems [Carroll et al., 2004]) to effects at global scales (such as biogeochemical cycling [Kurz and Apps, 1999]). Improved understanding of the role of climate in driving insect outbreaks will enhance our ability to forecast future forest responses to projected climate change. [50] Acknowledgments. Thanks are owed to Linda Joyce for helpful discussions and to Lee Casuto for providing programming support. We appreciate the thoughtful and constructive suggestions of two anonymous reviewers, which improved the manuscript. This work was funded by the USDA Forest Service Cooperative Agreement 03-CS-11222033-315, the USGS Western Mountain Initiative (USGS Cooperative Agreement 04CRAG0004/4004CS0001), NASA grant NNG04GH63G, and NSF grant DMS-0077663. References Amman, G. D. (1973), Population changes of the mountain pine beetle in relation to elevation, Environ. Entomol., 2, 541 – 547. Amman, G. D. (1982), Characteristics of mountain pine beetles reared in four pine hosts, Environ. Entomol., 11, 590 – 593. Amman, G. D. (1985), The beetle, in Integrating Management Strategies for the Mountain Pine Beetle with Multiple-Resource Management of Lodgepole Pine Forests, edited by M. D. McGregor, and D. M. Cole, pp. 2 – 7, Intermountain For. and Range Exp. Stn., USDA For. Serv., Ogden, Utah. Amman, G. D., and W. E. Cole (1983), Mountain pine beetle dynamics in lodgepole pine forests: Part II. Population dynamics, report, USDA For. Serv., Washington, D. C. Amman, G. D., and M. D. McGregor (1985), Hazard rating and predicting tree loss in unmanaged stands, in Integrating Management Strategies for the Mountain Pine Beetle with Multiple-Resource Management of Lodgepole Pine Forests, edited by M. D. McGregor and D. M. Cole, pp. 29 – G02019 30, Intermountain For. and Range Exp. Stn., USDA For. Serv., Ogden, Utah. Amman, G. D., M. D. McGregor, and R. E. Dolph (1990), Mountain pine beetle, For. Insect and Disease Leaflet 2, USDA For. Serv., Washington, D. C. Bailey, R. G., and H. C. Hogg (1986), A world ecoregions map for resource reporting, Environ. Conserv., 13(3), 195 – 202. Bale, J. S., et al. (2002), Herbivory in global climate change research: Direct effects of rising temperature on insect herbivores, Global Change Biol., 8(1), 1 – 16. Bentz, B. J., J. A. Logan, and G. D. Amman (1991), Temperature dependent development of mountain pine beetle and simulation of its phenology, Can. Entomol., 123, 1083 – 1094. Bentz, B. J., J. A. Logan, and J. C. Vandygriff (2001), Latitudinal variation in Dendroctonus ponderosae (Coleoptera: Scolytidae) development time and adult size, Can. Entomol., 133, 375 – 387. Burns, R. M., and B. H. Honkala (1990), Silvics of North America, vol. 1, Conifers, 877 pp., USDA For. Serv., Washington, D. C. Carroll, A. L., S. W. Taylor, J. Regniere, and L. Safranyik (2004), Effects of climate change on range expansion by the mountain pine beetle in British Columbia, in Mountain Pine Beetle Symposium: Challenges and Solutions, edited by T. Shore, J. E. Brooks, and J. E. Stone, pp. 223 – 232, Pac. For. Cent., Can. For. Serv., Nat. Resour. Can., Kelowna, B. C., Canada. Clements, F. E. (1910), The life history of lodgepole burn forests, Bull. 79, USDA For. Serv., Washington, D. C. Dale, V. H., et al. (2001), Climate change and forest disturbances, Bioscience, 51(9), 723 – 734. Daly, C., G. H. Taylor, and W. P. Gibson (1997), The PRISM approach to mapping precipitation and temperature, paper presented at 10th Conference on Applied Climatology, Am. Meteorol. Soc., Reno, Nevada. Despain, D. G. (1990), Yellowstone Vegetation: Consequences of Environment and History in a Natural Setting, 239 pp., Roberts Rinehart, Boulder, Colo. Fastie, C. L., T. W. Swetnam, and E. E. Berg (1995), Annual report of the Kenai National Wildlife Spruce Bark Beetle History Project, report, U.S. Fish and Wildlife Serv., Soldotna, Alaska. Ferry, G. W., R. G. Clark, R. E. Montgomery, R. W. Mutch, W. P. Leenhouts, and G. T. Zimmerman (1995), Altered fire regimes within fire-adapted ecosystems, in Our Living Resources A Report to the Nation on the Distribution, Abundance, and Health of U.S. Plants, Animals, and Ecosystems, edited by E. T. LaRoe et al., pp. 222 – 224, Natl. Biol. Serv., U.S. Dep. of the Interior, Washington, D. C. Fleming, R. A. (2000), Climate change and insect disturbance regimes in Canada’s boreal forests, World Resour. Rev., 12(3), 520 – 555. Fleming, R. A., J. N. Candau, and R. S. McAlpine (2002), Landscape-scale analysis of interactions between insect defoliation and forest fire in Central Canada, Clim. Change, 55(1 – 2), 251 – 272. Furniss, M. M. (1997), American forest entomology comes on stage, Am. Entomol., 43, 40 – 47. Furniss, R. L., and V. M. Carolin (1977), Western forest insects, Misc. Publ. 1339, 346 pp., USDA For. Serv., Washington, D. C. Hall, J. P., and B. Moody (1994), Forest depletions caused by insects and diseases in Canada 1982 – 1987, report, 14 pp., For. Insect and Disease Surv., Can. For. Serv., Nat. Resour. Can., Ottawa. Hanks, E., J. A. Powell, J. A. Logan, and B. J. Bentz (2001), Estimating phloem temperature for studying mountain pine beetle phenology, report, 25 pp., Dep. of Math. and Stat., Utah State Univ., Logan. Hicke, J. A., G. P. Asner, E. S. Kasiske, N. H. F. French, J. T. Randerson, G. J. Collatz, B. J. Stocks, C. J. Tucker, S. O. Los, and C. B. Field (2003), Postfire response of North American boreal forest net primary productivity analyzed with satellite observations, Global Change Biol., 9(8), 1145 – 1157. Holsten, E. H., P. Hennon, L. Trummer, and M. Schultz (2001), Insects and diseases of Alaskan forests, report, 243 pp., Alaska Region, USDA For. Serv., Washington, D. C. Houghton, R. A., J. L. Hackler, and K. T. Lawrence (2000), Changes in terrestrial carbon storage in the United States: 2. The role of fire and fire management, Global Ecol. Biogeogr., 9(2), 145 – 170. Intergovernmental Panel on Climate Change (2001), Climate Change 2001:The Scientific Basis – Contribution of the Working Group I to the Third Assessment Report of the Intergovernmental Panel on Climate Change, 881 pp., Cambridge Univ. Press, New York. Kittel, T. G. F., et al. (2004), VEMAP Phase 2 bioclimatic database: I. Gridded historical (20th century) climate for modeling ecosystem dynamics across the conterminous USA, Clim. Res., 27, 151 – 170. Kurz, W. A., and M. J. Apps (1999), A 70-year retrospective analysis of carbon fluxes in the Canadian forest sector, Ecol. Appl., 9(2), 526 – 547. Leenhouts, B. (1998), Assessment of biomass burning in the conterminous United States, Conserv. Eco., 2(1), 1. (Available at www.consecol.org/ vol2/iss1/art1) 11 of 12 G02019 HICKE ET AL.: CLIMATE CHANGE AND INSECT OUTBREAKS Little, E. L. (1971), Atlas of United States Trees, vol. 1., Conifers and Important Hardwoods, 211 pp., USDA For. Serv., Washington, D. C. Logan, J. A., and G. D. Amman (1986), A distribution model for egg development in mountain pine beetle, Can. Entomol., 118, 361 – 372. Logan, J. A., and B. J. Bentz (1999), Model analysis of mountain pine beetle (Coleoptera: Scolytidae) seasonality, Environ. Entomol., 28, 924 – 934. Logan, J. A., and J. A. Powell (2001), Ghost forests, global warming and the mountain pine beetle (Coleoptera: Scolytidae), Am. Entomol., 47, 160 – 173. Logan, J. A., and J. A. Powell (2004), Modeling mountain pine beetle phenological response to temperature, in Mountain Pine Beetle Symposium: Challenges and Solutions, edited by T. Shore, pp. 210 – 222, Pac. For. Cent., Can. For. Serv., Nat. Resour. Can., Kelowna, B. C., Canada. Logan, J. A., P. V. Bolstad, B. J. Bentz, and D. L. Perkins (1995), Assessing the effects of changing climate on mountain pine beetle dynamics, in Workshop on Interior West Global Climate, edited by R. W. Tinus, pp. 92 – 105, USDA For. Serv., Fort Collins, Colo. Logan, J., J. Regniere, and J. A. Powell (2003), Assessing the impacts of global warming on forest pest dynamics, Frontiers Ecol. Environ., 1(3), 130 – 137. McCullough, D. G., R. A. Werner, and D. Neumann (1998), Fire and insects in northern and boreal forest ecosystems of North America, Annu. Rev. Entomol., 43, 107 – 127. Ministry of Forests (2005), 2004 aerial overview survey estimates (preliminary), report, Gov. of B. C., Vancouver, B. C., Canada. Parlange, M. B., and R. W. Katz (2000), An extended version of the Richardson model for simulating daily weather variables, J. Appl. Meteorol., 39(5), 610 – 622. Pfister, R. D., and D. M. Cole (1985), The host, in Integrating Management Strategies for the Mountain Pine Beetle with Multiple-Resource Management of Lodgepole Pine Forests, edited by M. D. McGregor and D. M. Cole, pp. 7 – 28, Intermountain For. and Range Exp. Stn., USDA For. Serv., Ogden, Utah. Powell, J. A., and J. A. Logan (2005), Insect seasonality: Circle map analysis of temperature-driven life cycles, Theor. Popul. Biol., 67, 161 – 179. Powell, J. A., J. L. Jenkins, J. A. Logan, and B. J. Bentz (2000), Seasonal temperatures alone can synchronize life cycles, Bull. Math. Biol., 62, 977 – 998. Raffa, K. F., and A. A. Berryman (1987), Interacting selective pressures in conifer-bark beetle systems: A basis for reciprocal adaptations?, Am. Nat., 129(2), 234 – 262. G02019 Shore, T. L., and L. Safranyik (1992), Susceptibility and risk rating systems for the mountain pine beetle in lodgepole pine stands, report, For. Can., Victoria, B. C., Canada. Stuart, J. D., J. K. Agee, and R. I. Gara (1989), Lodgepole pine regeneration in an old, self-perpetuating forest in south central Oregon, Can. J. For. Res., 19, 1096 – 1104. Taylor, S. W., and A. L. Carroll (2004), Disturbance, forest age dynamics and mountain pine beetle outbreaks in BC: A historical perspective, in Mountain Pine Beetle Symposium: Challenges and Solutions, edited by T. Shore, Pac. For. Cent., Can. For. Serv., Nat. Resour. Can., Kelowna, B. C., Canada. Tomback, D. F. (2002), The rapid decline of whitebark pine communities, in Ecology and Earth Sciences in Mountain Areas, edited by L. Taylor et al., pp. 31 – 42, Banff Cent., Banff, Alberta, Canada. Turner, M. G., W. H. Romme, and R. H. Gardner (1999), Prefire heterogeneity, fire severity, and early postfire plant reestablishment in subalpine forests of Yellowstone National Park, Wyoming, Int. J. Wildland Fire, 9(1), 21 – 36. U.S. Department of Agriculture Forest Service (2002), Western bark beetle report: A plan to protect and restore western forests, report, 7 pp., Washington, D. C. U.S. Department of Agriculture Forest Service (2003), Forest insect and disease conditions in the Rocky Mountain region, 2002, report, 28 pp., Washington, D. C. U.S. Department of Agriculture Forest Service (2004), Forest insect and disease conditions in the United States, 2003, report, 156 pp., Washington, D. C. VEMAP Members (1995), Vegetation/ecosystem modeling and analysis project: Comparing biogeography and biogeochemistry models in a continental-scale study of terrestrial ecosystem responses to climate change and CO2 doubling, Global Biogeochem. Cycles, 9, 407 – 438. Williams, D. W., and A. M. Liebhold (2002), Climate change and the outbreak ranges of two North American bark beetles, Agric. For. Entomol., 4, 87 – 99. J. A. Hicke and D. S. Ojima, Natural Resource Ecology Laboratory, Colorado State University, Fort Collins, CO 80523, USA. (jhicke@nrel. colostate.edu) J. A. Logan, U.S. Department of Agriculture Forest Service, Logan, UT 84321, USA. J. Powell, Department of Mathematics and Statistics, Utah State University, Logan, UT 84322, USA. 12 of 12