EVIDENCE OF CHARGE A MICROSCOPIC VIEW OF CHARGE A
advertisement

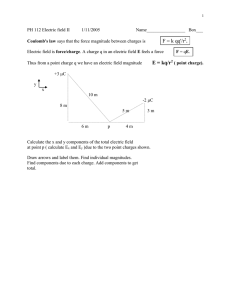
4/8/2016 CHAPTER 20, SECTION 1 – ELECTRIC CHARGE Main Idea Like charges repel, and unlike charges attract EVIDENCE OF CHARGE Electrostatics is the study of electric charges that can be collected and held in one place. Scientist have determined that there are two types of electric charge Benjamin Franklin named them positive and negative charges Two objects with like charges always repel each other. Two objects with unlike charges always attract each other. If you rub your hair with a balloon, it will stand on end because of the electrostatic force between your hair and the oppositely charged balloon. A MICROSCOPIC VIEW OF CHARGE A MICROSCOPIC VIEW OF CHARGE All materials contain light, negatively charged particles called electrons. In addition, each atom has a massive, positively charged nucleus, containing protons. For a neutral object, the amount of positive charge exactly balances the amount of negative charge. With the addition of energy, the outer electrons can be removed from atoms. An atom missing electrons has an overall positive charge, and consequently, any matter made of these electron-deficient atoms is positively charged. The freed electrons can remain unattached or become attached to other atoms, resulting in negatively charged particles. A MICROSCOPIC VIEW OF CHARGE Electric charge carriers are electrons rather than protons because the electrons: Have a lighter mass Are located far from the nucleus Are loosely bound to the nucleus A MICROSCOPIC VIEW OF CHARGE From a microscopic viewpoint, acquiring charge is a process of transferring electrons. If two neutral objects are rubbed together, each can become charged. 1 4/8/2016 A MICROSCOPIC VIEW OF CHARGE For instance, when rubber shoes are rubbed on a wool rug, electrons are removed from the atoms in the wool, and transferred to the shoes. The extra electrons on the rubber result in a net negative charge. The electrons missing from the wool result in a new positive charge. Charge is conserved. This is, individual charges never are created or destroyed, they just move around. HAVE YOU EVER EXPERIENCED AN ELECTRIC CHARGE? Ever been shocked by a door knob or car door handle? Had the saran wrap stick to itself? Had clothes stick to each other when getting them out of the dryer (static cling)? Gone down a slide and had your hair stick up? These are all examples of electric charge. CONDUCTORS AND INSULATORS A material through which a charge will not move easily is called an electric insulator. A material that allows charges to move about easily is called an electric conductor. Metals are good conductors because at least one electron on each atom can be removed easily. These electrons move freely throughout the piece of metal. CHAPTER 20, SECTION 2 – ELECTROSTATIC FORCE Main Idea Forces between charged particles are mathematically related to charge and distance. CONDUCTORS AND INSULATORS Under certain conditions, charges move through air as if it were a conductor The spark that jumps between your finger and a doorknob after you have rubbed your feet on a carpet discharges you. In other words, you have become neutral because the excess charges have left you. Similarly, lightning discharges a thundercloud. In both of these cases, air became a conductor for a brief moment. Excess charges in the cloud and on the ground are great enough to remove electrons from the molecules in the air. FORCES ON CHARGED OBJECTS The electrostatic force can be demonstrated by suspending a charged rod so that it turns easily. The results of these experiments and the actions of the charged rods can be summarized in the following way: There are two kinds of electric charge: positive and negative Charges exert forces on other charges at a distance. The force is stronger when the charges are closer together. Like charges repel; opposite charges attract. 2 4/8/2016 ELECTRIC FORCE When two charged objects are brought close together, they may experience motion either toward or away from each other. The closer the two objects are to each other, the stronger the force between them. The greater the magnitude of the charges the stronger the force between them COULOMB’S LAW qa qb r2 The electric force, like all other forces, is a vector quantity. COULOMB’S LAW FK qa qb r2 Used to collect soot in smokestacks, there by reducing air pollution Tiny paint droplets, charged by induction, can be used to paint automobiles and other objects uniformly Photocopy machines use static electricity to place black toner on a page to make a copy FK qa qb r2 The standard unit for Charge is called the Coulomb (C). The symbol for charge is q. One coulomb is the charge of 6.24 x 1018 electrons or protons. The charge on a single electron is -1.60 x 10-19 C. The magnitude of the charge of an electron is called the elementary charge. The charge on a single proton is +1.60 x 10-19 C. EXAMPLE – COULOMB’S LAW •Coulomb’s Law is used to calculate the force between 2 charged objects Electric force Coulomb constant There are many applications of electric forces on particles COULOMB’S LAW According to Coulomb’s Law, the magnitude of the force between two point charges (qa and qb) a distance r apart can be written as follows. FK APPLICATIONS OF ELECTROSTATIC FORCES (charge 1)(charge 2) (distance) 2 •Coulomb’s Constant, Kc = 8.99 X 109 N.m2/C2 •qa and qb = Charge on object 1 and 2 •r = distance between the objects. •F = Electric Force What is the magnitude of the electric force between a proton and an electron in a hydrogen atom? The two particles are separated by a distance of 5.3 x 10-11 m on average. Given: qa = -1.60 x 10-19 C. qb = 1.60 x 10-19 C. r = 5.3 x 10-11 m Unknown: F=? The equation: F k Ans. 8.2 x qa qb r2 10-8 N 3