Guidelines for Allergen Immunotherapy Extracts Label Documentation
advertisement

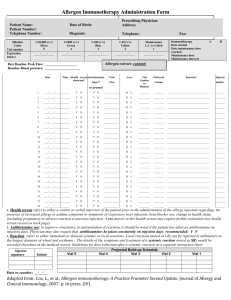
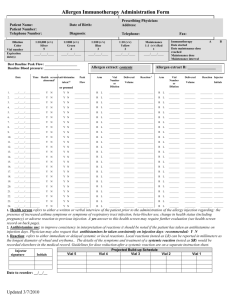
Guidelines for Allergen Immunotherapy Extract Label Documentation 1 Each vial of allergen immunotherapy extract should be labeled in a way that permits easy identification. Each label should include the following information • Appropriate patient identifiers may include the patient’s name, patient number, patient’s picture and birth date. • The contents of the allergen immunotherapy extract in a general way. The detail with which this can be identified depends on the size of the label and the number of allergens in the vial. Ideally, allergens should be identified as trees, grasses, weeds, mold, dust mite, cockroach, cat, and dog, etc. Due to space limitations, it may be necessary to abbreviate the antigens e.g., T, G, W, M, DM, Cr, C, and D respectively A full and detailed description of vial contents should be recorded on the prescription/content form • The dilution from the maintenance concentrate in volume per volume (v/v). If colors, numbers, or letters are used to identify the dilution, they also should be included • The expiration date should be included Examples of Possible Abbreviations for Allergen Immunotherapy Extract Components: Tree T Grass G Bermuda B WeedsVial Nomenclature: W Recommended a consistent and redundant labeling system for Ragweed R allergen extract dilutions is recommended Mold M Alternaria Alt During theCladosporium build-up phase a Cla number of dilutions of the maintenance vial are delivered. A redundantPenicillium labeling system may Pcn reduce administration errors. In addition to labeling dilutions from maintenance, Cat Ca color-coding, numerical or alphabetical system can be used. Dog D Cockroach Cr Dust Mite DM D. farinae Df D. pteronyssinus Dp Updated on 4/5/2011 Mixture Mx 2 Recommended Vial Nomenclature: A consistent and redundant labeling system for allergen extract dilutions is recommended During the build-up phase a number of dilutions of the maintenance vial are delivered. A redundant labeling system may reduce administration errors. In addition to labeling dilutions from maintenance, a color-coding, numerical or alphabetical system can be used. Dilution from Maintenance Concentrate Maintenance Concentrate 10-fold 100-fold 1000-fold 10,000-fold *V/V label 1:1 1:10 1:100 1:1000 1:10,000 Number 1 2 3 4 5 Color Red Yellow Blue Green Silver *Volume per volume • If a numbering system is used, the highest concentration should be numbered 1 and subsequent dilutions should be numbered from the maintenance concentration. This is will allow for consistency in labeling because if a greater number of dilutions are made for some patients, the number of the maintenance vial will remain the same. • If a color-coding system is used, the highest concentration should be red, the next highest yellow, followed by blue, green and silver in that order. • Specific contents each allergen immunotherapy extract vial should be listed separately, including final concentrations of each of its constituents. Consistency is essential as a basis for adoption of a universal standardized system. Updated on 4/5/2011 Example of color-coded allergen immunotherapy maintenance concentrate vial with dilutions. 3 All of the above information is derived from: Cox L, Nelson H, Lockey R, et al. Allergen immunotherapy: a practice parameter third update. J Allergy Clin Immunol 2011;127:S155 and accommpaning on-line supplementary material Updated on 4/5/2011