10 pericyclic
advertisement

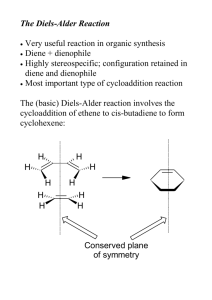
Pericyclic reactions Pericyclic reaction • Concerted reactions with cyclic transition states (breaking and making of bonds occur simultaneously ) • No intermediates Pericyclic reaction • • • • Cycloaddition Reactions Electrocyclic Reactions Sigmatropic Rearrangements Ene Reactions Cycloaddition Reactions • Using electrons in π bonds → form two σ bonds and become one ring • Diels-Alder reaction C (diene + dienophile) • HOMO / LUMO Selectivity in Diels-alder • Exo < Endo • Secondary orbital interactions exo endo If substituent is quite large… Asymmetric Diels-Alder reaction • Chiral catalyst BINOL,VANOL,VAPOL or Ti-BINOL… • Recent Study In this case they are using Boron Compounds Chiral ligand VANOL or VAPOL BH3 THF AcOH Good way to synthesis some natural products Xia Li, Jianguang Han, Alexander X. Jones, and Xiaoguang Lei J. Org. Chem., 2016, 81 (2), pp 458–468 Up to 97% ee Asymmetric Diels-Alder reaction • Chiral catalyst MacMillan’s Catalyst CHO…useful acrolein R Me Pr i-Pr Ph Furyl yield 75 92 81 99 89 5mol% cat. MeOH H2O 23℃ exo:endo 1:1 1:1 1:1 1.3:1 1:1 exo ee (%) 86 (2S) 86 (2S) 84 (2S) 93 (2S) 91 (2S) Kateri A. Ahrendt , Christopher J. Borths , and David W. C. MacMillan J. Am. Chem. Soc., 2000, 122 (17), pp 4243–4244 endo ee (%) 90 (2S) 90 (2S) 93 (2S) 93 (2S) 93 (2S) Asymmetric Diels-Alder reaction • Chiral catalyst chiral oxazaborolidine (R)-2-Methyl-CBS -oxazaborolidine Only 4mol% catalyst For high yield and ee 4mol% 99% yield 94% ee Duan Liu , Eda Canales , and E. J. Corey J. Am. Chem. Soc., 2007, 129 (6), pp 1498–1499 Intramolecular Diels-alder Intramolecular Diels-alder • MacMillan’s Catalyst (S) Exo selective Up to 20:1 selectivity (R) Endo selective Joel F. Hooper, Natalie C. James, Esra Bozkurt, Viktorya Aviyente, Jonathan M. White, Mareike C. Holland, Ryan Gilmour, Andrew B. Holmes, and K. N. Houk J. Org. Chem., 2015, 80 (24), pp 12058–12075 Electrocyclic reactions • Use one π bond to form one σ bond (and become ring) • Thermal and photochemical ways Electrocyclic reactions conrotatory Δ hv disrotatory Hv → electron be excited Number of π Thermal reaction bonds Photochemical reaction even • A conrotatory disrotatory odd disrotatory conrotatory Can be explained by Woodward-Hoffman rule example Up to 15.7 : 1 Ashay Patel, Gregg A. Barcan, Ohyun Kwon, and K. N. Houk J. Am. Chem. Soc., 2013, 135 (12), pp 4878–4883 Sigmatropic rearrangements • one σ-bond is changed to another σ-bond • Claisen rearrangement keto-enol tautomerism Δ Good way to get alcohol • Cope rearrangement Δ examples • Not real pericyclic… 10 mol% cat AcOEt 0℃ 2h 94% yield 92% ee 10 mol% cat CH2Cl2 -50℃ 2h 68% yield 94% ee Duffey, T. A.; Shaw, S. A.; Vedejs, E. J. Am. Chem. Soc. 2009, 131, 14-14. Joannesse, C.; Johnston, C. P.; Concellon, C.; Simal, C.; Philp, D.; Smith, A. D. Angew. Chem. Int. Ed. 2009, 48, 8914-8918. Fisher indole synthesis • phenylhydrazine + aldehyde or ketone (acid condition) [H+] [H+] rearrangement -NH3 indole Nowadays, many other synthesis are reported example Not good example… But these sigmatropic rearrangements are good for asymmetric synthesis (well controlled, doesn’t affect) 7steps 3steps Asymmetric hydrovinylation 99% yield 96% ee 4-Et-7-vinylindole 22% yield Wang Liu, Hwan Jung Lim, and T. V. RajanBabu J. Am. Chem. Soc., 2012, 134 (12), pp 5496–5499 Indole derivatives • Many significant derivatives are reported Biologically active Ene reaction • Using C-H σ bond to form new bond (so not real pericyclic reaction?) • Need high temp • Lewis acid / metal catalyst → lower temp At the end… Pericyclic • Medicine • Natural products Many have rings