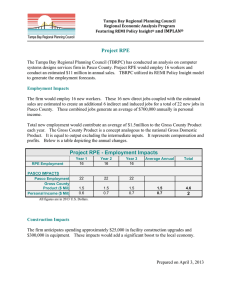
Session 111 RPE
advertisement

ARVO 2016 Annual Meeting Abstracts 111 RPE Sunday, May 01, 2016 8:30 AM–10:15 AM Exhibit/Poster Hall Poster Session Program #/Board # Range: 232–268/B0238–B0274 Organizing Section: Retinal Cell Biology Program Number: 232 Poster Board Number: B0238 Presentation Time: 8:30 AM–10:15 AM Comparative proteomic profiling of cultured RPE cells derived from human embryonic stem cells vs. rhesus monkey Bruce A. Pfeffer1, 2, Chengjian Tu3, Jun Li3, Jun Qu3, 4, Steven J. Fliesler1, 2. 1Research Service, VAWNYHS, Buffalo, NY; 2 Ophthalmology, Biochemistry, and SUNY Eye Institute, University at Buffalo, Buffalo, NY; 3Pharmaceutical Sciences, University at Buffalo, Buffalo, NY; 4SUNY Eye Institute, University at Buffalo, Buffalo, NY. Purpose: Cultured retinal pigment epithelial (RPE) cells from nonhuman primate, and those derived from human embryonic stem cells (hesc), each with their own strategic and translational merits and deficiencies, are potential substitutes for RPE cell lines and cultures established from human donor eyes. A fair consensus with regard to human RPE signature gene and protein expression has emerged from analysis of native and cultured RPE in published reports. We generated proteomic profiles to compare cultured hesc-derived and rhesus macaque RPE cells in terms of correlation with human RPE signature genes and proteins. Methods: Human (hesc-derived) and rhesus RPE cells (4th and 3rd passage, respectively) were maintained as stable, confluent monolayers for 8 weeks. Both strains displayed a polarized, pigmented epithelial phenotype in vitro. Triplicate sets of cell lysates were subjected to on-pellet proteolytic digestion and separations by nano-reverse-phase LC/ion-current-based tandem mass spectroscopy [Tu et al., Mol Cell Proteomics 2013]. Identifications from the UniProt human protein database were made on the basis of at least two unique peptides for each protein. Proteins detected were quantified by AUC using SIEVE, and ranked by relative abundance. Results: Approximately 1100 – 1300 proteins per sample were identified. Intensity vs. retention time plots showed consistent alignment for replicates within each species (87.6% and 95.1% for hesc- and rhesus RPE, respectively), but only a mean of 41.3% between species. For the 110 most abundant proteins detected in either hesc- or rhesus RPE, 70.0% vs. 77.3% of these overlapped with the other subset, respectively. Of the 50 most abundant proteins detected, 35 from either hesc-derived or rhesus cultured RPE corresponded to an entry from at least one comprehensive, recently published list of human RPE signature genes or proteins [Strunnikova et al., Hum Mol Genet 2010; West et al., Mol Cell Proteomics 2003]. Cytoskeletal, retinoid-processing, ion and small molecule transport, and antioxidant/pro-survival functional categories were highly represented for both species. RPE65, an exemplary signature protein for native RPE, was detected in both hesc- and rhesus RPE, with comparative normalized abundance values of 0.041 and 0.350, respectively. Conclusions: Proteomic profiling may be used to authenticate cultured hesc-RPE and non-human primate RPE. Commercial Relationships: Bruce A. Pfeffer, None; Chengjian Tu, None; Jun Li, None; Jun Qu, None; Steven J. Fliesler, None Support: NIH EY007361 (SJF); U54HD071594 and HL103411 (JQ); Center of Protein Therapeutics Industrial Award (JQ); RPB Unrestricted Grant (SJF); and VAWNYHS facilities and resources (SJF, BAP) Program Number: 233 Poster Board Number: B0239 Presentation Time: 8:30 AM–10:15 AM In vivo quantification of macular RPE cell density in infant monkeys using fundus photography Lauren M. Renner1, Emily Johnson1, Kasie W. Paul1, Travis B. Smith2, Trevor J. McGill2, 1, Martha Neuringer1, 2. 1Neuroscience, Oregon Health & Science University, Beaverton, OR; 2Casey Eye Institute, Oregon Health & Science University, Portland, OR. Purpose: The density of retinal pigment epithelial (RPE) cells in adult human and monkey eyes peaks in the fovea and decreases with eccentricity, as demonstrated by histology and by AOSLO autofluorescence, but has not been examined during development. Surprisingly, we have been able to visualize macular RPE cells in infant monkeys with standard fundus photography techniques, possibly as a result of the lack of RPE and choroidal pigmentation, and/or increased magnification due to small eye size. This study utilized this phenomenon to quantify the development of the RPE cell density profile longitudinally in infant monkeys in vivo. Methods: RPE cells were visualized longitudinally in infant rhesus macaques (n=11) at 2, 4, 8, 16 and 24 weeks of age by color and redfree fundus photography and fluorescein angiography using a Zeiss FF450 fundus camera. For each animal and time point, the clearest central fundus image of any imaging modality showing visible RPE cells was selected. Images were analyzed with Mosaic Analytics Software (courtesy of Joseph Carroll) with manual correction. A custom MATLAB program was used to calculate RPE density in the 0.5 mm diameter foveal center and in three concentric annuli at 0.25-0.5, 0.5-0.75, and 0.75-1.00 mm eccentricities, with additional measurements at greater eccentricities when possible. Results: At 2 weeks of age, RPE cells were visible in the macula of all 11 infant monkeys. Density was highest in the central fovea (2,400 cells/mm2) and decreased progressively with eccentricity. By 8 weeks of age, RPE density increased in all regions; the increase was greatest in the central 0.5 mm, with density reaching 3,000 cells/mm2. The area in which RPE cells were visible decreased as pigmentation increased, and in some infants RPE cells were visible only in the central 0.5 mm of the fovea. By 16 weeks, RPE cells were no longer visible in most infants. Conclusions: RPE cell density can be visualized and quantified in the infant primate macula using standard ophthalmic imaging techniques. Quantification of RPE cell density revealed the presence of a foveal peak as early as 2 weeks and a progressive density increase similar to the developmental pattern of foveal cone photoreceptor density. Commercial Relationships: Lauren M. Renner; Emily Johnson, Abbott Nutrition (F); Kasie W. Paul, Abbott Nutrition (F); Travis B. Smith, None; Trevor J. McGill, Abbott Nutrition (F); Martha Neuringer, Abbott Nutrition (F) Support: Abbott Nutrition through the Center for Nutrition, Learning, and Memory at the University of Illinois and NIH grant P51OD011092 Program Number: 234 Poster Board Number: B0240 Presentation Time: 8:30 AM–10:15 AM Impact of platelet-derived growth factor-C on junctional adhesion molecule-C expression in human blood-retinal barrier cells Xu Hou. ophthalmology, Eye institute of Chinese PLA, Xi'an, China. Purpose: To investgate the expression change of junctional adhesion molecule-C (JAM-C) in human retinal pigment epithelial cells (RPEs) and retinal capillary endothelial cells (RCECs) induced by platelet-derived growth factor-C (PDGF-C). Methods: Human RPE cells and RCECs were treated by VEGF-A or PDGF-C, respectively. The expression quantity of JAM-C These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record. ARVO 2016 Annual Meeting Abstracts was analyzed by real-time PCR, flow cytometry and western blot, and the expression distribution of JAM-C was observed by immunofluorescence staining. Results: After the treatment with VEGF-A, the total JAM-C expression in the two kinds of cells was not significantly changed. Meanwhile, the expression level in cytoplasm was decreased, and the expression level in membrane was increased. After the treatment with PDGF-C, the total JAM-C expression in the two kinds of cells was increased. The expression level in both cytoplasm and membrane were increased. Conclusions: VEGF-A may promote the transfer of JAM-C expression from cytoplasm to cell membrane. PDGF-C may not only promote the transfer of JAM-C expression, but also promote the total JAM-C expression. Thus, PDGF-C should play a bigger role in the regulation of JAM-C in the two kinds of blood-retinal barrier cells. Commercial Relationships: xu hou, None Program Number: 235 Poster Board Number: B0241 Presentation Time: 8:30 AM–10:15 AM Assessment of Phagocytic Function of Human Retinal Pigment Epithelium in Vitro George Inana1, Chris Murat1, Weijun An1, Ian Harris2, Jing Cao2. 1 Bascom Palmer Eye Institute, Miami, FL; 2Janssen Research and Development, Spring House, PA. Purpose: The diurnally regulated phagocytosis of the outer tips of photoreceptors that occur every morning is a key function of the retinal pigment epithelium (RPE) in sustaining the health of the light-sensing photoreceptors. Its importance is illustrated in the well-known animal model of retinal degeneration, the Royal College of Surgeons rat, in which a defect in the RPE phagocytic function leads to retinal degeneration. In fact, the genetic defect (mutation in the Mertk gene) causing this defect has also been found in some humans with retinitis pigmentosa. This brings up the possibility that a defect in the RPE phagocytic function may play a role in other retinal degenerative conditions, including age-related macular degeneration (AMD), especially since AMD is characterized by the accumulation of abnormal intra- and extracellular debris (lipofuscin, drusen) which have been shown to be composed of material from the photoreceptors which ended up in the RPE via phagocytosis. With the goal of studying the status of phagocytic function in normal and AMD human RPE, we first set up an in vitro phagocytosis assay system for the normal human RPE and carried out an initial assessment of the phagocytic function. These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record. ARVO 2016 Annual Meeting Abstracts Methods: Harvesting of human RPE, culture, isolation of rod outer segments (ROS) and labeling with fluorescein, and phagocytosis assay were mostly based on the methods used for rat RPE phagocytosis assay that we have published (McLaren, Inana, Li IOVS 1993). RPE was obtained from 8 post-mortem human eyes. ROS was prepared from human, rat, and pig retinas. Results: Feeding of fluorescently labeled ROS to human RPE cultured for at least 1 week demonstrated evidence of phagocytosis of the ROS, clearly distinguishable microscopically as ingested phagosomes, after ~8 hours of incubation as seen in our rat RPE phagocytosis assays. The kinetics of phagocytosis by the human RPE was also similar to that of the rat, with binding of the ROS first, then ingestion after 7-8 hours. The human RPE preferred the human ROS over the rat and pig for phagocytosis. A baseline of phagocytic level was established for the normal human RPE, and a moderate decrease in the level was observed with age (28 to 79yo). Conclusions: Characteristics of ROS phagocytosis by the human RPE were similar to that for the rat, and the initial findings will be useful for the analysis of AMD eyes in the near future. Commercial Relationships: George Inana, Janssen R&D (R), Janssen R&D (F); Chris Murat, None; Weijun An, None; Ian Harris, Janssen R&D; Jing Cao, Janssen R&D Support: NIH Center Core Grant P30EY014801, Research to Prevent Blindness Unrestricted Grant to the University of Miami Program Number: 236 Poster Board Number: B0242 Presentation Time: 8:30 AM–10:15 AM Semaphorin 4D inhibits photoreceptor outer segment phagocytosis by retinal pigment epithelial cells Ayelen Bulloj, Silvia C. Finnemann. Department of Biological Sciences, Center for Cancer, Genetic Diseases, and Gene Regulation, Fordham University, Bronx, NY. Purpose: Daily clearance phagocytosis by the retinal pigment epithelium (RPE) of photoreceptor outer segment fragments (POS) is critical for photoreceptor function and longevity. In this process, POS bind to ανβ5 integrin to stimulate signaling via Mertk and AKT kinases, which ultimately target Rac1-dependent F-actin reorganization that is required for POS engulfment. Semaphorins belong to a diverse family of secreted or membrane associated glycoproteins that are ligands for the plexin receptor family. The receptor for semaphorin 4D (Sema4D) is plexinB1 (PlB1). PlB1 can reorganize the cytoskeleton by interacting with active Rac1 in a ligand dependent manner and/or by promoting integrin-dependent activation of AKT through RhoA/Rho kinase (ROCK). Here, we set out to study if Sema4D and its receptor PlB1 contribute to the molecular mechanism of RPE phagocytosis. Methods: Bound and internalized POS were quantified by feeding primary fetal human RPE cells with POS with or without recombinant Sema4D followed by confocal microscopy (CM) analysis. RhoA and Rac1 activity were measured by ELISA. Retinal Sema4D protein levels before and after light onset were studied by immunoblotting (IB) and by live tissue staining followed by CM in wild-type (wt) and Mertk null (RCS) rat eyes. Phosphorylation of PlB1 in rat eyes were analyzed by anti-phosphotyrosine immunoprecipitation followed by IB. Phagosome quantification and retina structure analysis were performed on sections of wt, Sema4D and PlB1 null mouse retina. Results: Added Sema4D abolished POS internalization by RPE cells in culture without affecting binding. ROCK inhibition or Rac1 activation restored POS internalization blocked by Sema4D. PlB1 phosphorylation and Sema4D levels were reduced during the peak of phagocytosis in wt rat eyes. This pattern was altered in RCS rat eyes, in which POS internalization does not occur. In mice with Sema4D or PlB1 gene deletion the RPE showed increased POS phagosome content, but otherwise their retina lacked obvious structural defects. Conclusions: Our findings identify a new function for the ligandreceptor pair Sema4D/PlB1 in the retina and add new players to the photoreceptor outer segment renewal mechanism that is essential to maintain health and functionality of the retina. Commercial Relationships: Ayelen Bulloj, None; Silvia C. Finnemann Support: NIH grant R01-EY026215 Program Number: 237 Poster Board Number: B0243 Presentation Time: 8:30 AM–10:15 AM Contribution of annexin A5 to diurnal phagocytosis by the retinal pigment epithelium CHEN YU1, Luis E. Munoz2, Sebastian Boeltz2, Silvia C. Finnemann1. 1 Department of Biological Sciences, Center for Cancer, Genetic Diseases and Gene Regulation, Fordham University, Bronx, NY; 2 Department of Internal Medicine 3, Rheumatology and Immunology, Friedrich-Alexander-Universität Erlangen-Nürnberg, Erlangen, Germany. Purpose: Diurnal phagocytosis of spent photoreceptor outer segment fragments (POS) by the retinal pigment epithelium (RPE) is critical for vision. POS recognition/binding by apical αvβ5 integrin receptors of the RPE triggers signaling that synchronizes POS engulfment in the eye. Annexin A5 is a Ca2+ dependent phospholipid binding protein with a binding domain for αvβ5 integrin. However, the physiological function of such interaction remains unknown. Here, we examined if and how annexin A5 contributes to RPE phagocytosis. Methods: Annexin A5 expression in rodent retina/RPE was examined using Western blotting and immunofluorescence microscopy. Retinal cell marker labeling was used to compare retinal morphology of annexin A5 knock-out (KO) and wild-type mice. POS phagosomes in the RPE at different times after light onset were quantified to assess the phagocytic activity of annexin A5 KO RPE in situ. POS phagocytosis assays of unpassaged primary RPE or of mouse embryonic fibroblasts (MEFs) were performed to assess the effect of silencing or overexpressing annexin A5 on binding and internalization of isolated POS particles. Results: We found that annexin A5 is expressed in the neural retina, RPE and choroid in mouse and rat eyes. 9-month old Annexin A5 KO mice showed normal retinal morphology. However, in situ phagosome quantification revealed that annexin A5 KO RPE lacks the peak of phagosome load at light onset that is characteristic for wild-type RPE. Silencing annexin A5 decreased and overexpressing annexin A5 increased POS binding by wild-type primary RPE cells in culture. Silencing annexin A5 did not directly affect internalization or F-actin phagocytic cup formation by primary RPE. Re-expressing annexin A5 was sufficient to rescue the impaired binding capacity of annexin A5 KO MEFs and to increase POS binding by wild-type MEFs above normal. In contrast, overexpressing annexin A5 in integrin β5 KO MEFs had no effect on POS binding. Conclusions: Our results demonstrate that annexin A5 plays an important role in diurnal outer segment renewal. Moreover, our cell culture assays indicate that annexin A5 specifically of the RPE contributes to this process. Like αvβ5 integrin receptors, manipulating annexin A5 alters POS binding but not POS internalization. Our results further suggest that the function in phagocytosis of annexin A5 is dependent on αvβ5 integrin. Commercial Relationships: CHEN YU, None; Luis E. Munoz, None; Sebastian Boeltz, None; Silvia C. Finnemann, None Support: NIH R01-EY026215 These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record. ARVO 2016 Annual Meeting Abstracts Program Number: 238 Poster Board Number: B0244 Presentation Time: 8:30 AM–10:15 AM Discovering Novel Genes and Pathways Involved in Maintenance of Epithelial Phenotype in Retinal Pigment Epithelium Cells Justin R. Chang1, Balendu Shekhar Jha1, Madhu Lal2, Ruchi Sharma1, Marc Ferrer2, Kapil Bharti1. 1National Eye Institute, National Institutes of Health, Bethesda, MD; 2National Center for Advancing Translational Sciences, National Institutes of Health, Rockville, MD. Purpose: Proliferative vitreoretinopathy (PVR) causes retinal pigment epithelium (RPE) dedifferentiation where RPE loses its epithelial characteristics. The goal of this study is to perform a series of high throughput screening assays and bioinformatics analyses to identify novel genes and pathways that are involved in maintenance of RPE epithelial phenotype to facilitate vision recovery after PVR surgery. Methods: RPE cells derived from a reporter line of human induced pluripotent stem cells (iPSCs) expressing RPE-specific GFP and constitutive housekeeping RFP were optimized to be cultured in 384-well plates. A library of 875 miRNA mimics was tested, and top 5% GFP up- and down-regulators (GFP, RFP signals measured by MetaXpress) were identified. Multiple miRNA databases were used to analyze these selected mimics for their target genes. Targets that had >90% scores and common occurrences between multiple databases per miRNA were chosen for subsequent siRNA screening. siRNA screening results and Ingenuity Pathway Analysis (IPA) software were used to compile a list of genes for further assays. Results: The miRNA assay had a high reproducibility with correlation coefficients of 0.87, 0.94, 0.91 in GFP, RFP, Hoechst signals, respectively, between two identical screenings. The top 5% of miRNA mimics (21 mimics) that decreased GFP by 50% compared to control and the top 5% miRNA mimics (10 mimics) that increased GFP by 250% compared to control were selected for target analysis. miRNA databases and IPA predicted novel genes and pathways that are involved in regulating the RPE epithelial phenotype. The results were confirmed by a subsequent siRNA screening comprising of 122 siRNAs. Small molecule inhibitors and activators of these pathways are currently being tested to modulate in vitro RPE phenotype. Conclusions: HTS on iPSC-RPE identified novel genes and pathways involved in RPE epithelial phenotype maintenance, and potential therapeutic targets for RPE-associated conditions like PVR. Additionally, these pathways can provide potential targets to enhance the protocol of RPE differentiation from pluripotent stem cells. Commercial Relationships: Justin R. Chang, None; Balendu Shekhar Jha, None; Madhu Lal, None; Ruchi Sharma, None; Marc Ferrer, None; Kapil Bharti, None Program Number: 239 Poster Board Number: B0245 Presentation Time: 8:30 AM–10:15 AM F-actin organization as potency indicator for high-quality, functional human RPE cell culture Claudia Müller, Silvia C. Finnemann. Department of Biological Sciences, Center for Cancer, Genetic Diseases and Gene Regulation, Fordham University, Bronx, New York, NY. Purpose: Retinal pigment epithelial (RPE) cell replacement therapies for treatment of age-related macular degeneration are under development. Cells used for transplantation must establish critical functional interactions with photoreceptors in the diseased retina. Phagocytosis of shed photoreceptor outer segment fragments (POS) is one important property of differentiated RPE. We investigated cellular markers that may predict robust phagocytic capacity. Methods: RPE cell lines derived from adult human RPE stem cells (RPESC-RPE) passage 2 were used for experiments. Western blot analysis, immunostaining and confocal microscopy were used to study cell morphology and marker protein expression. Protein secretion was assessed by ELISA. Binding and internalization of purified POS were quantified to assess phagocytic capacity of RPE cells in culture. Results: Phagocytosis assays showed that RPE cell lines could be categorized as either highly phagocytic (with 55-80% of cells engulfing POS in our assay) or poorly phagocytic (with 5-40% of cells engulfing POS). Regardless of phagocytic capacity, all RPE lines tested expressed known proteins of the phagocytic pathway and secreted phagocytic ligands (MFG-E8 and ProteinS). However, in poorly phagocytic cells, POS surface binding and apical ezrin localization were dramatically reduced indicating abnormal microvilli. In highly phagocytic cells, the majority of cells showed cobblestone epithelial morphology with an F-actin-rich apical brush border, a circumferential apical adhesion belt and basal F-actin at substrate attachment sites. In poorly phagocytic cells, less than 10% of cells showed a circumferential F-actin belt, and most of these cells contained less apical F-actin and abundant central and basal F-actin stress fibers. Fewer than 1% of cells expressed the mesenchymal marker smooth muscle actin (SMA). Inhibition of Rho-associated, coiled-coil protein kinase (ROCK) was sufficient to improve epithelial F-actin characteristics and to increase the fraction of cells engulfing POS. Conclusions: Our results suggest that F-actin cytoskeleton analysis is sufficient to assess epithelial morphology of RPE cells in culture. Moreover, alterations in F-actin directly correlate with and may contribute to diminished phagocytic function of RPE in culture. Commercial Relationships: Claudia Müller; Silvia C. Finnemann, None Support: Empire State Stem Cell Fund through New York State Department of Health Contract #C028505 Program Number: 240 Poster Board Number: B0246 Presentation Time: 8:30 AM–10:15 AM Molecular Mechanisms of Oxytocinergic Signaling and its Inhibition of Kir7.1 in the RPE Nathaniel York1, 2, Patrick Halbach1, Michelle Chiu1, Ian Bird1, De-Ann M. Pillers1, 2, Bikash R. Pattnaik1, 2. 1Pediatrics, University of Wisconsin - Madison, Madison, WI; 2McPherson Eye Research Institute, Madison, WI. Purpose: Oxytocin (OXT) is a neuropeptide that activates the oxytocin receptor (OXTR), a rhodopsin family G-protein coupled receptor. We have localized OXTR to the retinal pigment epithelium (RPE) and OXT has been found in the adjacent cone photoreceptors. We hypothesize that there is OXTR signaling in the retina and sought to characterize this signaling in the RPE and explore the downstream effects of OXT on cellular signaling, focusing on the regulation of inwardly rectifying K+ channel Kir7.1. Methods: Ca2+ response to OXT was measured in cultured human fetal RPE cells (hfRPE) using Fura-2AM in the presence of 2-APB and nifedipine, pharmacological inhibitors of Ca2+ signaling pathways. HEK-293 cells where used to establish stable expression of human OXTR and signaling was visualized using live cell imaging following transient expression of PH-GFP and PKC-GFP, monitors of GPCR metabolites PIP2 and DAG. Whole cell patch clamp electrophysiology was performed on HEK-OXTR cells transfected with GFP-fused Kir7.1 as well as freshly isolated mouse RPE cells to monitor Kir7.1 current. Results: OXT treatment of RPE cells in culture resulted in a transient increase in cytoplasmic Ca2+ that was reduced by 95% in the presence of the IP3R antagonist, 2-APB (P<0.001). Upon bathing the cells in Ca2+ free extracellular solution or nifedipine, the Ca2+ response to OXT was not altered. While the amplitude of responses was not These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record. ARVO 2016 Annual Meeting Abstracts altered, time to recover from the rise in [Ca2+]i peak was faster, with time constants (τ) of 0.53 (r2 = 0.983), 0.93 (r2 = 0.975), and 1.7 (r2 = 0.987) min for Ca2+-free, nifedipine, and Ringer’s solution, respectively. We also demonstrate that OXTR activation blunted Kir7.1 channel current, which has a physiologic role in RPE function. In isolated mouse RPE, we observed an average 61.81 ± 4.77 % decrease in K+ inward current amplitude and an average 11.4 ± 3.2 mV depolarization in resting membrane potential. Conclusions: We propose that OXTR utilizes multiple capacitative Ca2+ entry (CCE) mechanisms to sustain an increase in Ca2+ driven by intracellular signaling molecules coupled to OXTR/G-protein in the RPE. This OXT-OXTR signaling in the RPE cell also integrated mobilization of intracellular Ca2+ and the parallel modulation of the Kir7.1 channel. We suggest that novel OXT-OXTR signaling pathways in the outer retina will be of fundamental importance for eye development, health and visual function. Commercial Relationships: Nathaniel York; Patrick Halbach, None; Michelle Chiu, None; Ian Bird, None; De-Ann M. Pillers, None; Bikash R. Pattnaik, None Support: Meriter Grant MSN180934; UW Foundation Program Number: 241 Poster Board Number: B0247 Presentation Time: 8:30 AM–10:15 AM Choroid endothelial cells regulate RPE tight junctions through modulation of Bruch′s membrane assembly Ignacio Benedicto1, Guillermo Lehmann-Mantaras1, Michael Ginsberg2, Daniel J. Nolan2, Olivier Elemento4, Nazia M. Alam3, 4, Glen T. Prusky3, 4, Arvydas Maminishkis6, Sheldon S. Miller6, Shahin Rafii2, 5, Enrique J. Rodriguez-Boulan1. 1 Ophthalmology, Weill Cornell Medical College, New York, NY; 2 Angiocrine Bioscience, New York, NY; 3Burke Medical Research Institute, White Plains, NY; 4Physiology and Biophysics, Weill Cornell Medical College, New York, NY; 5Genetic Medicine, Weill Cornell Medical College, New York, NY; 6NEI, NIH, Bethesda, MD. Purpose: A hallmark of terminal retinal differentiation is the coordinated maturation of RPE, Bruch′s membrane (BM) and choroid vasculature. Recent studies demonstrate that endothelial cells (ECs) play key instructive roles in the differentiation and maintenance of parenchymal cells. We tested the hypothesis that choroid ECs regulate terminal differentiation of RPE and the outer bloodretina barrier by modulating BM assembly, which in turn triggers remodeling of RPE tight junctions (TJs). Methods: We co-cultured in Transwell chambers human fetal RPE (hfRPE) and either human umbilical vein ECs or mouse choroid ECs isolated by in vivo labeling of VE-cadherin and flow cytometry cell sorting. We assessed the effect of ECs on hfRPE TJs by measuring transepithelial electrical resistance (TER) and occludin subcellular localization by cell surface biotinylation and quantitative microscopy. We studied extracellular matrix (ECM) deposition by hfRPE by scanning electron microscopy and immunofluorescence assays. Lysyl oxidase activity, which catalyzes collagen and elastin crosslinking, was inhibited in EC-conditioned media by β-aminopropionitrile (BAPN) treatment. We carried out RNAseq analyses of choroid ECs from developing (P5) and adult (P30) mouse retinas. We evaluated newborn mice treated for 30 days with BAPN for visual function by optomotor response and for BM maturation by transmission electron microscopy. Results: EC-conditioned media induced a specific increase in hfRPE TER and the accumulation of occludin along hfRPE TJs. These effects were significantly impaired by BAPN treatment and EC lysyl oxidase-like 2 (LOXL2) knockdown. ECs enhanced the appearance of bundled collagen I-positive fibers in the ECM deposited by hfRPE, and increased the levels of active β1 integrin at the hfRPE basal plasma membrane. RNAseq analyses showed that several ECMrelated genes were differentially expressed in developing versus adult choroid ECs. In particular, LOXL2 was significantly more expressed in P5 than in P30 choroid ECs. In vivo, BAPN treatment during mouse terminal retinal differentiation induced defects in BM structure and visual function. Conclusions: Our results suggest that the regulation of BM composition and structure by choroid ECs is key for the maturation of RPE TJs and the establishment of the outer blood-retina barrier, essential for proper vision. Commercial Relationships: Ignacio Benedicto, None; Guillermo Lehmann-Mantaras, None; Michael Ginsberg, Angiocrine Bioscience, Angiocrine Bioscience (I); Daniel J. Nolan, Angiocrine Bioscience (I), Angiocrine Bioscience; Olivier Elemento, None; Nazia M. Alam, None; Glen T. Prusky, CerebralMechanics (I); Arvydas Maminishkis, None; Sheldon S. Miller; Shahin Rafii, Angiocrine Bioscience (I); Enrique J. Rodriguez-Boulan, None Support: NIH/NEI R01 EY08538-24-29 Program Number: 242 Poster Board Number: B0248 Presentation Time: 8:30 AM–10:15 AM Helping retina to breathe by increasing fetal hemoglobin production in RPE: relevance to outer retinal preservation in hypoxic conditions characteristic of aging and diabetes Pamela M. Martin1, 2, Wanwisa Promsote1, Folami L. Powell1, Alan Saul2, Vadivel Ganapathy3, Pamela M. Martin1, 2. 1Biochemistry & Molecular Biology, Georgia Regents University, Augusta, GA; 2 Ophthalmology and Culver Vision Discovery Institute, Georgia Regents University, Augusta, GA; 3Cell Biology & Biochemistry, Texas Tech University Health Science Center, Lubbock, TX. Purpose: In normal adults, 97% of total red cell hemoglobin (Hb) is adult Hb (HbA). Elevated fetal Hb (HbF) levels post-infancy generally indicate underlying metabolic stress and tissue hypoxia. HbF binds oxygen with higher affinity than HbA; increased HbF production is regarded as a “rescue” attempt by the body to increase oxygen extraction and subsequent delivery to tissues under low oxygen conditions. Hb production by RPE brings new perspectives regarding the understanding of outer retinal oxygenation under normal conditions. In light of this discovery, the known high metabolic activity and consequent demand of photoreceptors for oxygen, here we evaluated whether HbF modulation confers benefit in retina under hypoxic conditions characteristic of aging and diabetes. Additionally, we investigated the underlying molecular mechanisms responsible. Methods: Mice engineered to produce human Hb were used in this study: abnormal Hb (HbSS)-producing mice to study the effects of HbF augmentation on chronic, low-grade hypoxia that occurs in aging and, normal Hb (HbAA)-producing mice ± STZ, to examine its effects on diabetes-induced hypoxia. Animals were treated ± the HbFinducer monomethylfumarate (MMF; 15 mg/ml in drinking water). Retinal health/function was monitored by ERG, OCT, and routine histologic, RNA and protein analyses. MMF-induced alterations in gene/protein expression were evaluated also in ARPE-19 cells cultured ± MMF (100 μM) under normal, hyperglycemic (25 mM glucose) or hypoxic (100 μM CoCl2 or 1% oxygen) conditions. Results: Oral MMF-therapy increased γ-globin mRNA/HbF production systemically and in retina, reduced oxidative stress/ inflammation, and preserved outer retinal morphology/function positively in chronically hypoxic retinas (HbSS and HbAA+STZ). MMF induced Nrf2 and downregulated Bcl11A, a repressor of γ-globin transcription and regulator of cell metabolism and senescence. siRNA studies demonstrated the effect on Bcl11A to be Nrf2-dependent. These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record. ARVO 2016 Annual Meeting Abstracts Conclusions: This study exposes novel potential links between factor that govern globin gene switching, redox status, metabolism and senescence and suggests that HbF-inducing therapies might afford multiple levels of benefit in hypoxic retina. Commercial Relationships: Pamela M. Martin; Wanwisa Promsote, None; Folami L. Powell, None; Alan Saul; Vadivel Ganapathy, Georgia Regents University (P); Pamela M. Martin, Georgia Regents University (P) Support: NIH Grant EY022704 Program Number: 243 Poster Board Number: B0249 Presentation Time: 8:30 AM–10:15 AM Functional studies of sodium/proton exchanger 8 in adult mouse retina Chun-hong Xia, Mei Li, Audrey Kim, Ian Ferguson, Xiaohua Gong. School of Optometry and Vision Science Program, University of California, Berkeley, Berkeley, CA. Purpose: To determine the essential role of sodium/proton exchanger 8 (NHE8) in the retina of adult mice and to investigate the mechanism of how NHE8 regulates pH homeostasis in the retinal pigment epithelium (RPE). Methods: Using the novel clustered regularly interspaced short palindromic repeats (CRISPR)-associated endonuclease (Cas)9 from Neisseria meningitidis (NmCas9), we studied the role of NHE8 in the retina of adult mice. Recombinant adeno-associated viruses (AAV) carrying NmCas9 and guide RNAs were used to knockdown NHE8 in the RPE of 4-week-old adult wild-type mice by subretinal injection; eye samples were collected 4 weeks post injection; immunostaining was performed to determine the distribution of NHE8 and other proteins in mouse retina; and NHE8 expression was examined by real-time PCR. Cultured RPE cells infected with AAV-NHE8-pHluorin-mcherry were used for pH measurements in intracellular compartments by monitoring the fluorescent intensity ratios between pH-sensitive ecliptic pHluorin and pH-insensitive mcherry. Results: Our previous studies revealed that NHE8 knockout and NHE8-M120K point mutant mice developed retinal degeneration associated with aberrant RPE with disrupted cell polarity; NHE8 proteins were predominantly colocalized with the Golgi complex and intracellular vesicles in the RPE as well as in the inner segments of rod photoreceptors. Current study showed a loss of photoreceptor cells in eyes injected with AAV-Cas9 and AAV-sgRNA target NHE8, while normal retinal morphology was observed in mice injected with AAV-Cas9 and AAV-control sgRNA. Real-time PCR analysis showed about 32% reduction of NHE8 transcripts in NHE8-sgRNA injected retinas compared to control-sgRNA injected retinas. Fluorescent intensity ratios revealed different pH values in some of intracellular compartments including protein trafficking pathways between RPE cells expressing wild-type NHE8 and mutant NHE8-M120K, infected with AAV-NHE8-pHluorin-mcherry and AAV-NHE8-M120KpHluorin-mcherry. Conclusions: Sustained function and regulation of NHE8 are essential for maintaining the polarity and function of RPE cells in the retina of adult mice. NHE8 regulates pH homeostasis in some intracellular compartments of RPE. Impaired NHE8 such as mutant NHE8-M120K would disrupt the protein trafficking or recycling to affect the polarity, phagocytosis and other functions of RPE cells, leading to photoreceptor cell death. Commercial Relationships: Chun-hong Xia, None; Mei Li; Audrey Kim, None; Ian Ferguson, None; Xiaohua Gong, None Support: A grant from The East Bay Community Foundation. Program Number: 244 Poster Board Number: B0250 Presentation Time: 8:30 AM–10:15 AM Anticancer Drug Cisplatin Differentially Affects Retina Pigment Epithelial Cells Depending on Mitochondrial Backgrounds Tej Patel, Cristina M. Kenney, Catherine Y. Liu, Marilyn Chwa, Shari R. Atilano, Jon L. Norman. Gavin Herbert Eye Institute, Irvine, CA. Purpose: Mitochondrial (mt) DNA can be classified into haplogroups based upon single nucleotide polymorphism patterns that define racial populations. The anticancer drug, cisplatin, is known to cause toxicity to the retina, including retinal pigmentary changes. Our hypothesis is that retinal pigment epithelial (RPE) cells with the same nuclear DNA but containing different mtDNA backgrounds are differentially affected by cisplatin. Methods: Cytoplasmic hybrids (cybrids), cell lines with identical nuclear genomes but different mitochondrial genomes, are created by fusion of platelets (with mtDNA but no nuclear DNA) collected from various individuals to RPE cells devoid of mtDNA (H haplotype, 3 cell lines; J haplotype, 3 cell lines). H and J cybrids were treated for 48 hours with 25 or 50 µM cisplatin and cell viabilities were measured using trypan blue exclusion. H and J cybrids were also treated with 40µM Cisplatin for 48 hours and the mitochondrial membrane potential (ΔΨm) was measured to test for early apoptotic changes. QPCR was used to analyze cellular gene expression changes. Untreated cultures served as the controls. Statistical analysis was done using GraphPad Prism 5. Results: With 25 uM and 50 uM cisplatin treatment, J cybrids show decreased viability to 65% (p=0.043) and 42% (p=0.002) of untreated, respectively. H cybrids had cell viability that was less effected by cisplatin: 87% (p=0.58) and 62% (p=0.051) of untreated H-cybrids, respectively. Cisplatin-treated and untreated H-cybrids had similar ΔΨm: 5.3% ± 7.8% (p=0.5). In contrast, treated J-cybrids s showed a 24.0% ± 4.1% (p<0.0001) decrease in ΔΨm compared to untreated. Comparison of gene expression in J-treated versus H-treated cybrids showed increase in apoptotic gene BAX (p=0.03, fold 1.4), but a decrease in genes such as SFRP1 (p=0.0001, fold 0.21), DHRS2 (p=0.041, fold 0.58) and EFEMP1 (p=0.009, fold 0.45). Conclusions: Retinal Pigment Epithelial cells with identical nuclear DNA but H versus J mtDNA haplogroups show significant differences in cell viability, mitochondrial membrane potential and cellular gene expression levels when treated with the cancer drug cisplatin. We show that mitochondrial DNA changes alone can affect cellular response. These findings may help identify individuals who may be more susceptible to cisplatin retinal toxicity depending on their mitochondrial background. Commercial Relationships: Tej Patel, None; Cristina M. Kenney, None; Catherine Y. Liu, None; Marilyn Chwa, None; Shari R. Atilano, None; Jon L. Norman, None Support: This work was supported by Discovery Eye Foundation, Guenther Foundation, Beckman Initiative for Macular Research, Polly and Michael Smith Foundation, Max Factor Family Foundation, Iris and B. Gerald Cantor Foundation. Program Number: 245 Poster Board Number: B0251 Presentation Time: 8:30 AM–10:15 AM Dissecting Exosome Biogenesis in Adult Human Retinal Pigment Epithelial Cells (ARPE19): Specific Interaction of αB-crystallin with Rab GTPase27b Rajendra K. Gangalum1, Ankur M. Bhat1, Sirus A. Kohan3, Suraj P. Bhat1, 2. 1Jules Stein Eye Institute, UCLA, Los Angeles, CA; 2 Molecular Biology Institute and Brain Research Insitute, UCLA, Los Angeles, CA; 3Brain Research Insitute, UCLA, Los Angeles, CA. These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record. ARVO 2016 Annual Meeting Abstracts Purpose: We have previously demonstrated that the small heat shock protein, αB-crystallin (αB) is secreted from ARPE19 cells in culture via exosomes (ARVO 2009; J Biol Chem. 286:3261-9, 2011). Exosomes are 50 -200 nm proteolipid vesicles that carry molecular information (proteins and RNA) from one cell to the other and therefore represent potential vectors of intercellular communication between cells in the RPE; they may also have a role in the maintenance of this epithelial layer as a physiological and physical barrier at the blood/retina interface. As currently understood exosomes are produced from the endocytic pathway that maintains membrane protein (receptor) homeostasis. Here we examine the status of this pathway in ARPE19 cells that express αB-crystallin and those that do not. Methods: We have employed Flow cytometry, Immunofluorescence and Confocal Microscopy, Western blotting, Co-immunoprecipitation and Double Immunogold labeling and Electron Microscopy to elucidate the status of the endocytic pathway in αB expressing and non-expressing stable ARPE19 αBshRNA transfected) cell lines generated in our laboratory. Results: In cells not expressing αB, the distribution of CD63 (LAMP3), an exosome marker, is markedly altered from the normal dispersed pattern to a stacked perinuclear presence. However sucrose density gradient analyses of cellular extracts indicate continued exosome synthesis under conditions of impaired exosome secretion. Immunoconfocal microscopy shows increased presence of CD63(LAMP3) and LAMP1 indicating enhancement of the endolysosomal compartment. This is further corroborated by increased labelling of this compartment by Rab7, a Rab GTPase known to be associated with the late endosome maturation. No change is however, seen with Rab5. Interestingly, we find that αB interacts specifically with Rab GTPase27b and not with Rab GTPase 27a in these cells suggesting that this small heat shock protein may have a role in collaboration with other small GTPases in regulating the secretion of exosomes from these cells. Conclusions: These data point to a regulatory role for αB in exosome biogenesis possibly via its involvement at a branch-point in the endocytic pathway populated by Rab GTPases known to be involved in multiple phases of secretion from eukaryotic cells. Commercial Relationships: Rajendra K. Gangalum, None; Ankur M. Bhat, None; Sirus A. Kohan, None; Suraj P. Bhat, None Support: NIH Grant to SPB 1R01EY024929 Program Number: 246 Poster Board Number: B0252 Presentation Time: 8:30 AM–10:15 AM PYK2 plays a role in matrix contraction by RPE-derived cells Shigeo Tamiya, Kevin McDonald, Henry J. Kaplan. Ophthalmology and Visual Sciences, University of Louisville, Louisville, KY. Purpose: Contractile fibroblastic/myofibroblastic cells derived from retinal pigment epithelial (RPE) cells have been implicated in the fibrotic complications of proliferative vitreoretinopathy. We have previously demonstrated that matrix contraction by RPE-derived cells can be inhibited by dasatinib, an FDA-approved cancer medication that inhibits multiple tyrosine kinases. The purpose of this project was to identify molecular targets affected by dasatinib that contribute to matrix contraction. Methods: Primary cultured porcine RPE cells were used between passages 3 to 5. Cells were cultured for 3 days in 25% vitreous fluid supplemented DMEM in the presence or absence of tyrosine kinase inhibitors for the final 24 hours. Western blot analyses were used to determine the phosphorylation status of phospho-tyrosine proteins. A type I collagen contraction assay was utilized to examine matrix contraction by cells. Results: Dasatinib reduced tyrosine phosphorylation of multiple proteins. Interestingly, while FAK auto-phosphorylation on Tyr397 was unaffected, auto-phosphorylation on Tyr402 of PYK2, also known as FAK2, was significantly reduced. In agreement with the Western blot data, PF431396, a dual inhibitor of PYK2 and FAK, significantly reduced matrix contraction while PF573228, an inhibitor of FAK but not PYK2, was without effect. Conclusions: The inhibitory effect of dasatinib on matrix contraction by RPE-derived cells is, at least in part, due to the prevention of PYK2 activation, which plays a role in the contractile process. Commercial Relationships: Shigeo Tamiya, None; Kevin McDonald, None; Henry J. Kaplan, None Support: DoD/USAMRAA BAA W81XWH-15-1-0298; Research to Prevent Blindness, New York, NY Program Number: 247 Poster Board Number: B0253 Presentation Time: 8:30 AM–10:15 AM MDM2 inhibition suppresses NF-κB-mediated inflammation in retinal pigment epithelium cells Biraj Mahato1, 2. 1Cell Biology and Immunology, University of North Texas Health science Center, Fort Worth, TX; 2Laboratory of Ratinal Rehabilitation, North Texas Eye Research Institute, Fort Worth, TX. Purpose: Our group has shown that Nutlin-3, a MDM2 inhibitor inhibits pathologic retinal endothelial cell proliferation through a p53-dependent mechanism, but its p53-independent functions and effect on retinal pigment epithelial (RPE) cell viability have not been adequately elucidated. TNF-α induced RPE inflammation through NF-κB is a known underlying mechanism of uveitis. Here, we sought to investigate the non-canonical, p53-indendepent function of Nutlin-3, and hypothesized that MDM2 inhibitors possess an anti-inflammatory function benefiting patients suffering from retinal inflammatory diseases. Methods: For all experiments, ARPE-19 cells were cultured to >95% confluence using standard methodology. In vitro cell viability was assessed using a Hemacytometer and the MTT assay. To assess retinal toxicity in vivo, 200 mM of Nultin-3 or vehicle (DMSO) (n=12 rat eyes per condition) was injected once weekly for four weeks. Retinal toxicity was assessed using histopathology, ERG, SD-OCT, and fundus photography. To assess the anti-inflammatory effect of Nutlin-3, ARPE-19 cells were infected with lenti-virus carrying an NF-κB luciferase reporter. Results: Nutiln-3 did not demonstrate a statistical difference in cell viability at doses lower than 45 mM (N=3, p>0.5). Rats did not show evidence of retinal toxicity, but some rats did develop cataracts, which may be related to the vehicle or injection technique. ARPE-19 cells were pretreated with Nutlin-3 10 μM (a dose that does not alter RPE cell viability) for 30 minutes to 7 hours, and then stimulated with TNF-α (50 ng/ml) for 24 hours. TNF-α alone induced NFκB luciferase reporter gene activity, and Nutlin-3 pre-treatment significantly attenuated this TNF-α response (p<0.05). Conclusions: Based on these experiments, these data suggest that Nutlin-3 does not cause retinal toxicity at doses less than 45 mM in vitro and 200 mM in vivo. Interestingly, Nutlin-3 appears to have a novel p53-indepenent function in RPE cells by inhibiting TNF-α mediated NF-κB RPE inflammation. Commercial Relationships: Biraj Mahato, None Support: R01 EY026201-01 These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record. ARVO 2016 Annual Meeting Abstracts Program Number: 248 Poster Board Number: B0254 Presentation Time: 8:30 AM–10:15 AM Glutaredoxin 2 (Grx2) Protects Retinal Pigment Epithelial Cells from Oxidative Damage by Regulating Autophagy Xiaobin Liu1, 2, Christy Xavier1, 2, Hongli Wu1, 2. 1North Texas Eye Research Institute, Fort Worth, TX; 2Pharmaceutical Sciences, University of North Texas Health Science Center, Fort Worth, TX. Purpose: Glutaredoxin 2 (Grx2) is an oxidoreductase present in the mitochondria where it protects the organelle from oxidative damage and maintains its redox homeostasis. The purpose of this study is to evaluate the cytoprotective effects of Grx2 in human retinal pigment epithelial (RPE) cells and characterize its potential function in regulating autophagy. Methods: Primary RPE cells were isolated from Grx2 knockout (KO) mice and treated with or without 400 µM H2O2 for 4 h. Human retinal pigment epithelial (ARPE-19) cells were transfected with either a human Grx2 cDNA-containing plasmid (pCR3.1-hGrx2) or an empty vector pCR3.1. Cells were treated with or without 200 µM H2O2 for 16 h. Grx2 protein expression was detected by western blot analysis. Cell viability was measured by a colorimetric assay with WST8. The morphology of nuclear chromatin was assessed by staining with Hoechst 33342. Apoptosis was quantitatively analyzed by flow cytometry. The level of protein glutathionylation (PSSG) and autophagy pathway proteins were measured by immunoblotting. Results: Primary RPE cells laking Grx2 were more sensitive to oxidative damage. On the other hand, Grx2 overexpression protected RPE cells from H2O2-induced cell viability loss. Assessment of apoptosis indicated that cells transfected with Grx2 were more resistant to H2O2 with fewer cells undergoing apoptosis as compared to vector control cells. PSSG accumulation was also attenuated by Grx2 overexpression with acute H2O2 challenge. Furthermore, the protein level of Beclin-1, a key molecule to initiate autophagy, was inhibited in Grx2 overexpressed cells with H2O2 treatment. LC3II, a protein that finalizes autophagosome maturation, was also increased. Conversely, primary Grx2 KO RPE cells showed higher levels of Beclin-1 and LC3II under oxidative stress. Conclusions: Grx2 rescues RPE cells from lethal oxidative damage, possibly through alleviation of ROS-triggered autophagy and prevention of PSSG accumulation. Commercial Relationships: Xiaobin Liu; Christy Xavier, None; Hongli Wu, Reata Pharmaceuticals (F) Support: BrightFocus Foundation for Macular Degeneration (Grant No. M2015180 to Hongli Wu) Program Number: 249 Poster Board Number: B0255 Presentation Time: 8:30 AM–10:15 AM Optimization of Culture Conditions for Human Retinal Progenitor Cells Danhong Zhu2, Christine Spee1, David R. Hinton3, 1. 1Ophthalmology, University of Southern California, Los Angeles, CA; 2Pathology, University of Southern California, Los Angeles, CA; 3Pathology, University of Southern California, Los Angeles, CA. Purpose: The implantation of retinal progenitor cells (RPCs) is under investigation for the therapy of retinal degeneration diseases. However, methods to increase cell growth, promote survival, and inhibit differentiation during in vitro culture remain a major challenge in the production of quality RPCs for implantation. This study aimed at optimizing the culture conditions to obtain high yield of quality RPCs. Methods: 1). Human retinas were isolated from fetal eyes at 18-20 gestational weeks, sheared into small cell aggregates by forcing them through a 25-gauge needle several times, and cultured in suspension in RPC medium for 24 hours. 2). RPC were then cultured in different conditions: co-culture with polarized human embryonic stem cellderived (HES)-RPE cells, surface-attached culture, and suspension culture. 3). RPCs cultured in different conditions and at certain time points were collected and analyzed for gene profiles by qPCR. Results: Using newly isolated retinas as a standard, RPCs in suspension culture expressed the highest level of photoreceptor marker genes, such as arrestin and rhodopsin, while RPC co-cultured with polarized HES-RPE cells expressed the highest level of Ki67 (proliferation marker gene) among the three groups after 1 month of culture. RPC in attached culture only expressed higher levels of neuronal and photoreceptor marker genes at 15 days of culture, but decreased the expressions of those genes at 1 month of culture. Conclusions: The co-culture of RPCs with polarized RPE promotes RPC growth and inhibits RPC differentiation; while suspension culture increases RPC photoreceptor differentiation. Future studies will focus on how culture conditions (co-culture and suspension culture) may be used sequentially, to achieve the most efficacious RPG for successful retinal integration, differentiation and function. Commercial Relationships: Danhong Zhu; Christine Spee, None; David R. Hinton, None Support: DR3-07438, California Institute for Regenerative Medicine (CIRM) Program Number: 250 Poster Board Number: B0256 Presentation Time: 8:30 AM–10:15 AM Computational Analysis of iPSC-Derived RPE Shape and Primary Cilium Location to Create Predictive Models of Cell Function Nathan A. Hotaling1, 2, Nicholas Schaub2, Jorge Ferrari1, Balendu Jha1, Carl Simon Jr.2, Kapil Bharti1. 1Unit on Ocular and Stem Cell Translational Research, National Eye Institute, Washington, DC; 2Biosystems & Biomaterials Division, National Institute of Standards and Technology, Gaithersburg, MD. Purpose: The emergence of primary cilium (PC) on human retinal pigment epithelium (RPE) cells regulates maturation of RPE as demonstrated by polarized expression of surface markers, tight junction formation, enhanced phagocytosis of photoreceptor outer segments (POS), and improved transepithelial potential/resistance (TEP/TER). We hypothesized that by analyzing the location of the PC as well as morphometric data from induced pluripotent stem cell (iPSC) derived RPE we could predict RPE maturation as defined by phagocytosis of POS, TER and TEP. Methods: Primary human iPSC derived RPE were cultured in porous transwell membranes over 6 weeks in the presence of PC inducers/suppressors (Aphidicolin, PGE2, and HPI-4). At the end of 6 weeks cells were stained for PC markers ARL13B, and GT335, tight junction marker ZO-1 and actin cytoskeleton stain phalloidin. Phagocytosis of labeled POS was quantified by flow cytometry. TER and TEP was recorded using a modified Ussing chamber. Cell morphometric quantification and location of PC, cellular center of mass (COM) and cell centroid were assessed via a novel ImageJ plugin. Linear and logistic regressions correlating POS level, TER and TEP with mean distance of cellular PC from cell centroid and COM as well as various morphological features were performed. Results: Enhanced induction of PC in RPE cells improved cell compactness and the number of neighbors per cell to a level similar to that of native human RPE. The level of cellular phagocytosis of POS, TER, and TEP were significantly correlated to PC distance from both RPE centroid and COM. Additionally, models including cell area, number of neighbors and aspect ratio of cell major and minor axis were found to be significantly predictive of RPE functionality. Conclusions: Using linear and logistic regression we show that iPS cell-derived RPE maturation, as assessed by phagocytosis of These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record. ARVO 2016 Annual Meeting Abstracts POS, monolayer TER and TEP can be predicted with high statistical certainty using four metrics: either PC distance to RPE centroid or COM, and cell area, number of neighbors, and the ratio of the cell major and minor axis. Due to the significance of the predictive ability of the model we conclude that analysis of fluorescent imaging of cells can adequately predict functional cell phenotype in lieu of, or in conjunction with, physiological tests. Commercial Relationships: Nathan A. Hotaling; Nicholas Schaub, None; Jorge Ferrari, None; Balendu Jha, None; Carl Simon Jr., None; Kapil Bharti, None Support: NIH Grant EY000542-02 Program Number: 251 Poster Board Number: B0257 Presentation Time: 8:30 AM–10:15 AM MicroRNA-34a inhibits proliferation, migration and adhesion of RPEs through direct targeting LGR4 Qiang Hou, Linglin Zhou, Jiajia Tang, Nan Ma, Ancong Xu, Lili Tu. Ophthalmology, Wenzhou Medical University, Wenzhou, China. Purpose: We previously showed that microRNA-34a was significantly downregulted in subconfluent in vitro cultured RPE cells. Introduction of microRNA-34a or siRNA for its known targets c-Met inhibited proliferation and migration of RPE cells. As microRNAs can target multiple genes at the same time. We tried to sought new target genes of microRNA-34a and investigate its role in the regulation of RPE cells. Methods: TargetScan Human 5.0 software was used to predict potential targets of microRNA-34a. This was further validated by double luciferase reporter assay and Western blot analysis. The expression of target gene was knockdowned with siRNA interference in RPE cells. Subsequently, the proliferative potential of RPE cells was evaluated with MTS and Ki67 staining after transfection of siRNA or microRNA-34a. The migrative ability was investigated with transwell and in vitro scratch assays. The adhesion ability was analyzed with cell attachment assay. The expression of downstream molecules was detected by Western blot. Results: TargetScan results suggested LGR4 is a putative target of microRNA-34a. LGR4, also known as GPR48, is a member of G-protein coupled receptor. Its role in development and caner metastasis have been well defined. However, there is no report about its role in RPE cells. We validated the direct regulation of LGR4 by microRNA-34a by double luciferase and Western blot assays. Introduction of siRAN sequence or microRNA-34a inhibited the proliferation, migration and adhesion of RPE cells. Furthermore, LGR4 can regulate similar downstream molecules as microRNA-34a, such as E2F1, p-CDC2, CDK2, CDK4 and CDK6. Conclusions: Our results indicate that LGR4 is direct target of microRNA-34a. MicroRNA-34a can inhibit the proliferation, migration and adhesion of RPE cells through regulation of LGR4. As microRNAs have promising therapeutical roles in many diseases, and G-protein coupled receptors are ideal targets of drug development, our results provide new insights in the prevention of PVR and other related retinal diseases. Commercial Relationships: Qiang Hou, None; Linglin Zhou, None; Jiajia Tang, None; Nan Ma, None; Ancong Xu, None; Lili Tu, None Support: National Natural Science Foundation of China Grant 81100671, Zhejiang Provincial Natural Science Foundation of China Grant LY16H120009 and Y2110609. Program Number: 252 Poster Board Number: B0258 Presentation Time: 8:30 AM–10:15 AM Primary Cilia Regulates Human iPSC-RPE Maturation via Regulation of WNT Signaling Qin Wan1, Balendu Jha2, Helen May-Simera3, Ruchi Sharma2, Juliet Hartford2, Vladimir Khristov1, Kiyoharu Miyagishima1, Mostafa R. Lotfi1, Sheldon S. Miller1, Kapil Bharti2. 1SERPD, NEI/ NIH, Bethesda, MD; 2OSCTR, NEI/NIH, Bethesda, MD; 3Institute of Zoology, Johannes-Gutenberg University, Mainz, Germany. Purpose: Induced pluripotent stem cell (iPSC) derivatives often do not fully-mature in vitro, limiting their use in cell therapy and disease modeling. Retinal pigment epithelium (RPE), a ciliated monolayer critical for maintaining the health and integrity of adjacent photoreceptors, is an attractive candidate for stem cell therapies to treat blinding eye diseases including Age-related macular degeneration. The primary cilium serves as a signaling and sensory hub and controls developmental cellular processes in many cell types, but the role of primary cilium in human RPE development remains largely unknown. The goal of this study is to explore the function and underlying mechanism of primary cilium in RPE development and maturation. Methods: Human iPSC derived RPE (iPSC-RPE) were grown on semi-permeable transwells to generate a confluent monolayer. Primary cilia in iPSC-RPE were manipulated using cilium inducers or blockers, or canonical WNT agonist or antagonist. Electrophysiology, immunocytochemistry, electron microscopy, gene expression, and phagocytosis assays were used to determine the polarization and maturity of iPSC-RPE monolayer. Results: The progressive development of primary cilia in human iPSC-RPE coincides with the formation of RPE tight junctions and epithelial cell morphology. Experimentally enhanced activation of primary cilia led to extensive apical processes, improved pigmentation, increased expression of adult-specific RPE genes, increased phagocytic capability, and significantly enhanced electrical responses that mimic native human RPE, demonstrating improved RPE maturation and functionality. Electrophysiological recordings in combination with immunostaining and phagocytosis using canonical WNT activator or inhibitors revealed that primary cilia-induced iPSC-RPE maturation was regulated through the suppression of canonical WNT signaling. Conclusions: Our results demonstrate a developmental role for primary cilia in human iPSC-RPE maturation, which is mediated through the suppression of canonical WNT pathway. This study provides a mechanistic tool to mature RPE or other epithelial cell types derived from human iPSCs, and also provides insight into retinal degeneration caused by ciliopathies. Commercial Relationships: Qin Wan, None; Balendu Jha, None; Helen May-Simera; Ruchi Sharma, None; Juliet Hartford, None; Vladimir Khristov, None; Kiyoharu Miyagishima, None; Mostafa R. Lotfi, None; Sheldon S. Miller, None; Kapil Bharti, None Support: NEI Intramural Funds, NIH CRM, and NIH Common Fund Program Number: 253 Poster Board Number: B0259 Presentation Time: 8:30 AM–10:15 AM Ex-vivo assessment of metabolic alterations of retinal pigment epithelial cells using Fluorescence Lifetime Imaging Ophthalmoscopy (FLIO) Natalie Blimke1, Joachim Pruessner1, Lars Alt1, Gereon Huttmann2, Ralf Brinkmann1, 2, Yoko Miura2, 1. 1Medical Lasercenter Luebeck, Luebeck, Germany; 2Institut for Biomedical Optics, Luebeck, Germany. These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record. ARVO 2016 Annual Meeting Abstracts Purpose: Fluorescence lifetime (FLT) decays of endogenous fluorophores in the fundus is becoming of more interest as a noninvasive and non-labeling indicator of retinal metabolic status. The aim of this study is to investigate changes of FLT of retinal pigment epithelial (RPE) cells in static organ culture over time and its relevance to cell metabolic status. Methods: Porcine RPE-choroid-sclera explants were cultivated in static culture medium. At 2, 24 and 48h after the initiation of cultivation the FLT of the RPE was measured using a fluorescence lifetime imaging ophthalmoscope (FLIO: Heidelberg Engineering) with 80 MHz pulsed excitation at 473 nm and two emission spectral channels (channel 1: 498-560 nm, channel 2: 560-700nm) coupled with a time-correlated single photon counting system. A custommade artificial eye model mimicking optical characteristics of the human eye served as a measuring chamber for the explants. RPE cell viability was examined using calcein-AM staining, and cellular glucose uptake was estimated by measuring glucose concentration of the conditioned medium using GlucCell® glucose meter. Results: Calcein-positive living RPE cells in the culture was found in 99.4% ± 0.27 at 48h, which was not significantly different from at 2h. There was an increase in cellular glucose uptake over time, from 48.0 ± 14.2 mg/mm2/h at 2h to 56.2 ± 17.2 mg/mm2/h at 24h, and 56.3 ± 17.3 mg/mm2/h at 48h (p=0.035). In FLIO, the FLT decay curve of the RPE was well fitted to a biexponential curve. The t2 lifetime (longer component) in channel 2 was significantly longer at 24h and 48h than at 2h (2308 ± 76 ps at 2h, 2473 ± 90 ps at 24h (p=0.006), and 2430 ± 103 ps at 48h (P = 0.017)). The ratio of the amplitude of t1 and t2 (a1/a2) in channel 2 also showed a significant decrease over time, 12.4 ± 3.2 at 2h, 11.0 ± 1.7 at 24h, and 9.8 ± 1.4 at 48h (p=0.04 vs 2h). Conclusions: Results suggest that FLIO might be useful to investigate metabolic alterations of the ex-vivo RPE. The change of cell energy metabolisms which increases glucose uptake might result in the increase of the length of long lifetime component and its amplitude. Further investigations will provide insight into its clinical use, e.g. in evaluating cell health and metabolic activity of the in-vivo RPE, or of the RPE sheet for RPE cell transplantation therapy. Commercial Relationships: Natalie Blimke; Joachim Pruessner, None; Lars Alt, None; Gereon Huttmann, None; Ralf Brinkmann, None; Yoko Miura, None Support: Heidelberg Engineering GmbH Program Number: 254 Poster Board Number: B0260 Presentation Time: 8:30 AM–10:15 AM Feedback Regulation of IRE1 During Unfolded Protein Response in RPE Sarah Melissa P. Jacobo, Maximilian J. Gerhardt, Arogya Khadka, Daniel Diaz-Aguilar, Magali Saint-Geniez. Department of Ophthalmology, Harvard Medical School / SERI, Boston, MA. Purpose: Inositol-requiring enzyme 1 (IRE1) regulates the most ancient branch of the unfolded protein response (UPR), and is present in the endoplasmic reticulum (ER) of all eukaryotes. IRE1 signaling is mediated by its splicing of X-box binding protein 1 (sXBP1), a transcription factor that is essential for expression of UPR genes that are required for ER stress relief. We previously reported that ER stress induced HtrA1 expression in retinal pigment epithelial (RPE) cells. This up-regulation was protective and essential for surviving proteotoxic cell death. We tested the hypothesis that IRE1 influences HtrA1 expression during ER stress in the RPE. Methods: Chronic protein misfolding was stimulated in cultured ARPE-19 cells using tunicamycin. HtrA1 expression was assayed by immunoblot and real time qRT-PCR. Parallel experiments were performed in the presence of 4μ8C (100 nM), an active site inhibitor of IRE1α’s endoribonuclease activity. To test the effect of HtrA1 suppression on IRE1α signaling, we stably expressed HtrA1-specific shRNA or control GFP shRNA. The total RNA pool was used in a UPR array (Qiagen) and gene expression was compared for IRE1α -related targets in vehicle- or tunicamycin-treated cells. For randomly selected hits, qRT-PCR results were validated by immunoblot. Data are representative of n>3 experiments, and *p<0.05 was considered statistically significant. Results: We mimicked conditions of proteotoxicity in RPE by preventing the glycosylation and ER-to-Golgi export of nascent peptides and found that intracellular HtrA1 protein accumulated in a tunicamycin dose (0.01–11 μM) and time (0-24h) – dependent manner. HtrA1 protein levels correlated with enhanced mRNA transcription. The IRE1α ribonuclease blocker 4μ8C potently inhibited the accumulation of sXBP1 and HtrA1 mRNA during chronic protein misfolding. We examined the effect of HtrA1 on IRE1α signaling in the face of ER stress. We found that expression of IRE1α protein, and consequently that of sXBP1, was suppressed after HtrA1 knockdown. Reduction in IRE1α and sXBP1 altered the mRNA and protein expression of members of the IRE1 signaling cascade. These include proteins which are critical to folding, glycosylation, and export of nascent peptides. Conclusions: Our results unravel a positive feedback loop that integrates HtrA1 into the IRE1α pathway, and which has direct consequences on RPE’s toolbox for combating proteotoxic stress. Commercial Relationships: Sarah Melissa P. Jacobo, None; Maximilian J. Gerhardt, None; arogya Khadka, None; Daniel Diaz-Aguilar, None; Magali Saint-Geniez, None Support: BrightFocus Foundation Macular Degeneration Award M2014025 Program Number: 255 Poster Board Number: B0261 Presentation Time: 8:30 AM–10:15 AM Usher proteins are engaged in vesicular trafficking in human retinal pigment epithelial and fibroblasts cells Bhagwat V. Alapure, Marianne Hathaway, Jennifer J. Lentz. Neuroscience Center of Excellence, Louisiana State University Health Sciences Centre, New Orleans, LA. Purpose: Usher syndrome (Usher) is the most common genetic condition that affects both hearing and vision. Sixteen genes are associated with 3 clinical subtypes. Among the Usher genes, thirteen are known and studies on their encoded proteins suggest that they interact together and function in multiprotein complexes. Individual Usher proteins, including myosin VIIa (USH1B), harmonin (USH1C), cadherin 23 (USH1D), protocadherin 15 (USH1F), SANS (USH1G), CIB2 (USH1J), usherin, (USH2A), VLGR1/GPR98 (USH2C), whirlin (USH2D), clarin-1 (USH3A), HARS (USH3B), PZDZ7 (USH modifier) and CEP250 (atypical USH), are expressed in hair cells and photoreceptors and are predicted to perform a wide range of functions including intracellular trafficking, scaffolding, cell adhesion and signaling. The exact function of these proteins and mechanism by which they interact is unclear. The purpose of this study is to determine the sub-cellular localization of Usher proteins and the effect of blocking trafficking on their expression in human retinal pigment epithelial (ARPE-19) and human dermal fibroblast (hFB) cultured cells. Methods: ARPE-19 and hFB cells were cultured with and without trafficking inhibitors Brefeldin A (BFA) and Membrane Traffic Inhibitor A5. The expression and sub-cellular localization of Usher proteins were investigated using immunohistochemistry techniques. Immunofluorescence intensity was quantified and analyzed using ImageJ Software. These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record. ARVO 2016 Annual Meeting Abstracts Results: All Usher proteins were found expressed in both ARPE-19 and hFB cells. All were found in the cytoplasm associated with the endoplasmic reticulum. Myosin VIIa, harmonin and cadherin 23 were also observed co-localized with the Golgi apparatus. Additionally, myosin VIIa, harmonin, cadherin 23, protocadherin 15, SANS, usherin and VLGR1 were found associated with F-actin showing a punctate pattern; whereas SANS appeared to co-localize along the long F-actin fibers. Lysosomal co-localization was not observed with any of the Usher proteins. Reduced expression of all Usher proteins was observed in the presence of BFA and A5 inhibitors. Conclusions: These data suggest Usher proteins are involved in intracellular trafficking between the ER and Golgi in ARPE-19 and hFB cells. Commercial Relationships: Bhagwat V. Alapure; Marianne Hathaway, None; Jennifer J. Lentz Support: The National Institutes of Health, Foundation Fighting Blindness, Eye on Jacob Foundation, Usher in Sight and Sound, and Usher 2020 Foundation. Program Number: 256 Poster Board Number: B0262 Presentation Time: 8:30 AM–10:15 AM Treatment of Haplogroup H and K ARPE-19 Cybrids with βAmyloid Kunal Thaker, Marilyn Chwa, Shari R. Atilano, Tej Patel, Cristina M. Kenney. Ophthalmology, Gavin Herbet Eye Institute UCI, Irvine, CA. Purpose: Ashkenazi Jewish populations, mainly of the mitochondrial (mt) haplogroup K, have been observed to have increased rates of hypercholesterolemia, a risk factor for age-related macular degeneration (AMD), compared to other Caucasian populations. Apolipoprotein E (apoE) is a protein involved in lipid transport, and processing the pathogenic peptide amyloid-β (am-β), known to have a role in Alzheimer’s and AMD. Haplogroup H and K adult retinal pigmental epithelial (ARPE) cytoplasmic hybrids (cybrids) were treated with amyloid-β1-42 (am-β1-42) to determine if the differences in APOE expression affected cellular response. Methods: H (n=6) and K (n=8) haplogroup cybrids were created using platelets from individuals. RNA and DNA were isolated from cells cultured to the fifth passage. GeneChip Array Analysis was performed to find the highest differential gene expression between the two haplogroups and qPCR was performed on genes of interest. Cells were plated on 96-well plates (5x104 cells per well) and treated with 20µM am-β1-42 after 24 hours. An MTT assay was done to observe cell viability 24 hours after treating. Each sample’s data was normalized to its untreated average. Statistical significance was determined through a student’s t-test. Results: GeneChip Array Analysis found that the K cybrids had increased gene expression in the atherosclerosis pathways compared to H cybrids. QPCR showed that the K cybrids had a 9.3-fold increase in the expression of APOE compared to the haplogroup H cybrids (p<0.0001). APOC1, COL1A1, and MMP1 were also measured but no significant difference was found between the H and K cybrids. After treatment with am-β1-42 the cell viability in H cybrids dropped 30% ± 2.3% (p<0.001), while the K cybrids decreased 16% ± 2.0% (p<0.0001) relative to their respective untreated controls. The difference in viability between the H and K cybrids post treatment was 14.7% (p<0.0001). Conclusions: These findings demonstrate that cells with haplogroup K mtDNA transcribe higher levels of APOE and are more resistant to am-β1-42 toxicity compared to the cells with haplogroup H mtDNA. In other systems, apoE enhances the elimination of am-β and provides cyto-protection. Our results suggest that in human RPE cells, higher levels of APOE expression may have protective functions against the pathogenic effects of am-β. These findings indicate that mtDNA may mediate levels of APOE expression and play a role in apoE-related diseases. Commercial Relationships: Kunal Thaker, None; Marilyn Chwa, None; Shari R. Atilano, None; Tej Patel; Cristina M. Kenney, None Support: Discovery Eye Foundation, Guenther Foundation, Beckman Initiative for Macular Research, Polly and Michael Smith Foundation, Max Factor Family Foundation, Iris and B. Gerald Cantor Foundation. Program Number: 257 Poster Board Number: B0263 Presentation Time: 8:30 AM–10:15 AM Validation of pluripotent stem cell derived RPE (scRPE) as a pure and renewable source for disease modeling and assay development Ulrich F. Luhmann2, Sonja Schlicht1, Faye M. Drawnel2, Silke Zimmermann1, Carolin Willburger2, Claire Hippert1, Verena Küppers2, Jean-Baptiste Vallier1, Jean-Philippe Carralot1, Marco Prunotto1, Mark Burcin1. 1Roche Pharma Research and Early Development, Therapeutic Modalities, Roche Innovation Center Basel, F. Hoffmann - La Roche Ltd., Basel, Switzerland; 2Pharma Research and Early Development, Ophthalmology Discovery and Biomarkers, Roche Innovation Center Basel, F. Hoffmann - La Roche Ltd., Basel, Switzerland. Purpose: Impaired function and loss of the retinal pigment epithelium (RPE) impact on photoreceptor health and contribute to vision loss in degenerative retinal diseases such as age-related macular degeneration, one of the most common causes for blindness in the developed world. To identify a reliable and renewable source of human RPE cells for disease modelling and assay development we implemented a differentiation protocol for pluripotent stem cell derived RPE cells (scRPE) and characterized their biological activity and function relative to ARPE19 cells and commercially available primary human RPE under different culture and assay conditions. Methods: To obtain RPE from pluripotent stem cells we developed a stepwise differentiation protocol based on small molecules inhibiting the Wnt and Nodal pathways during early cell development. To be assay compatible, we developed cultivation conditions to routinely expand, bank and mature scRPE. To validate their functional properties and compare scRPE to primary human RPE and ARPE-19 cells, we utilized gene expression profiling by PCR and immunocytochemistry to identify gene products that represent important functional properties of RPE. Further, we established assay conditions to test scRPE for their oxygen consumption capacity, ROS and cytokine production, cell polarization and barrier function. Results: Under identical cultivation conditions scRPE show a similar morphology to primary RPE. PCR analysis and immunostaining show that scRPE and primary RPE express RPE specific marker genes such as ZO-1 (tight junction), RPE65 (visual cycle), CRALBP (visual cycle), Tyrosinase (melanin biosynthesis), CD68 (phagocytosis) and VEGFA (vascular endothelial cell proliferation and survival). In comparison, ARPE-19 cells clearly show differences in morphology and gene expression. By measuring oxygen consumption, cell polarization, barrier function, and stressor-induced ROS and cytokine production scRPE cells produce comparable results to primary human RPE. Conclusions: ScRPE obtained by our differentiation protocol provide a reliable and renewable source of RPE cells that are functionally comparable to primary human RPE in several disease-relevant assay formats. This uniform source of scRPE cells will be useful for disease modelling in vitro and the development of novel assays for drug development. These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record. ARVO 2016 Annual Meeting Abstracts Commercial Relationships: Ulrich F. Luhmann, F. HoffmannLa Roche Ltd; Sonja Schlicht, F. Hoffmann-La Roche Ltd; Faye M. Drawnel, F. Hoffmann-La Roche Ltd; Silke Zimmermann, F. Hoffmann-La Roche Ltd; Carolin Willburger, F. HoffmannLa Roche Ltd; Claire Hippert, F. Hoffmann-La Roche Ltd; Verena Küppers, F. Hoffmann-La Roche Ltd; Jean-Baptiste Vallier, F. Hoffmann-La Roche Ltd; Jean-Philippe Carralot, F. HoffmannLa Roche Ltd; Marco Prunotto, F. Hoffmann-La Roche Ltd; Mark Burcin, F. Hoffmann-La Roche Ltd Program Number: 258 Poster Board Number: B0264 Presentation Time: 8:30 AM–10:15 AM A Porcine Model of Retinal Pigment Epithelium (RPE) Injury to Test the Efficacy of Human Induced Pluripotent Stem Cell– derived RPE (hiPSC-RPE) Transplants Juan Amaral1, 2, Maria M. Campos1, 3, Arvydas Maminishkis1, 4, Vladimir Khristov1, 4, Raymond Zhou1, 4, Steve T. Charles5, 1, Boris Stanzel6, 1, Balendu Jha1, 2, Irina Bunea1, 2, Sheldon S. Miller1, 4, Kapil Bharti1, 2. 1National Eye Institute, Bethesda, MD; 2Ocular and Stem Cell Translational Research, Bethesda, MD; 3NEI Histology Core, Bethesda, MD; 4Section on Epithelial and Retinal Phisiology and Disease, Bethesda, MD; 5Charles retina Institute, Memphis, TN; 6 Ophthalmology, University of Bonn, Bonn, Germany. Purpose: RPE replacement therapy has potential for the treatment of retinal degenerative diseases. Autologous hiPSC-RPE transplantation avoids immune rejection and the complications of long-term immunosuppression. We have developed a porcine model of localized retinal degeneration and a surgical procedure to test the ability of hiPSC-RPE cell transplants to protect RPE injury induced retinal degeneration. Methods: A micropulse laser with a 3% duty cycle and power between 600-750 mW was used to create a 5.4 mm2confluent grid lesion in 3 months old pigs. Histologic evaluation (n=15) was done on days 0, 2, and weeks 1, 2, 3 and 6 after laser. In selected cases, (n=14) 2 days after laser a standard 25G 4 port pars plana vitrectomy was done and a localized retinal detachment (RD) was created with a 38G cannula. Using a custom made injection tool, hiPSC-RPE cells on a PLGA biodegradable scaffold were introduced into the subretinal space. The retina was attached using fluid air exchange (FAX) without retinopexy. Results: Laser treatment leads to an immediate RPE detachment with edema. Bruch’s membrane and choriocapillaries are not damaged. By day 2 there is extensive RPE and photoreceptor damage but the photoreceptor nuclear layer (ONL) is still present. Over the next 3 weeks the ONL shrinks in the areas not transplanted by the hiPSCRPE scaffold with slow recovery after 5 - 6 weeks. During surgery a localized RD is induced in the laser treated area. The injector is introduced into the subretinal space through a retinotomy and the RPE scaffold is released. FAX is used to attach the RD and to close the enlarged sclerotomy via surface tension. Major complications include hemorrhage, proliferative vitreous retinopathy, and immune reaction against human cells. The overall success rate of the procedure is more than 50%. Conclusions: We have developed a porcine laser model for RPE ablation with selective outer retina damage. Our model and surgical techniques are suitable for in- vivo functional evaluation of hiPSCRPE transplants and their ability to rescue laser-damaged outer retina. Commercial Relationships: Juan Amaral, None; Maria M. Campos, None; Arvydas Maminishkis, None; Vladimir Khristov, None; Raymond Zhou, None; Steve T. Charles, None; Boris Stanzel, None; Balendu Jha, None; Irina Bunea, None; Sheldon S. Miller, None; Kapil Bharti, None Program Number: 259 Poster Board Number: B0265 Presentation Time: 8:30 AM–10:15 AM Development of a high content imaging photoreceptor outer segment phagocytosis assay in inducible pluripotent stem cell (iPSc)-derived RPE cells Megan Jabour, Heather MacLeod, Frada Berenshteyn, Arnaud Lacoste, Kathryn McAllister. Novartis Institutes for BioMedical Research, Cambridge, MA. Purpose: Daily phagocytosis of shed photoreceptor outer segments (OS) by the retinal pigmented epithelium (RPE) is essential to maintaining the health of the retina, and dysfunction of this process has been implicated in retinal degeneration. Prior studies have focused on OS phagocytosis by transformed RPE cell lines or freshly isolated RPE cells. The aim of these studies is to use high content imaging to develop an OS phagocytosis assay with iPSc derived RPE cells. Methods: iPSc derived RPE cells and the transformed RPE cell line, ARPE-19, were cultured in 384-well plates for 3-5 weeks prior to phagocytic challenge. Isolated bovine OS were labeled with FITC isomer or Qtracker 800 nanocrystals. OS were incubated with RPE cells for 2-24 hours at 37°C, washed, and fixed with 4% paraformaldehyde. Cells were then permeabilized with ice cold methanol and stained for rhodopsin or ZO-1, to evaluate tight junction formation. Images were acquired on the Molecular Devices ImageXpress MicroXL, and OS counts quantified using MetaXpress Transfluor HT analysis module. Results: Both iPSc derived RPE and ARPE-19 cells form tight junctions when cultured in 384 well format, but only iPSc derived RPE develop the cobblestone morphology typical of RPE cells. Visualization of RPE phagocytosis was achieved through direct labeling of OS with either Qtracker nanocrystals or FITC isomer. Complete colocalization with rhodopsin staining was observed with FITC labeled OS. Further evaluation of serial diluted OS demonstrate that ARPE-19 and iPSc derived RPE cells phagocytose OS in a concentration-dependent manner with maximal phagocytosis occurring at approximately 4 hours. Conclusions: We have developed a 384-well high content imaging assay allowing for visualization and quantification of OS phagocytosis by iPSc derived RPE cells. This in turn may enable high throughput evaluation and deconvolution of pathways involved in the phagocytic process. Commercial Relationships: Megan Jabour, Novartis; Heather MacLeod, Novartis; Frada Berenshteyn, Novartis; Arnaud Lacoste, Novartis; Kathryn McAllister, Novartis Program Number: 260 Poster Board Number: B0266 Presentation Time: 8:30 AM–10:15 AM Autophagy machinery is functional in human pluripotent stem cell-derived retinal pigment epithelial cells Kati M. Juuti-Uusitalo1, Niko Kivinen2, Johanna Viiri2, Juha Hyttinen2, 7, Arto Koistinen3, Hannu M. Uusitalo4, 5, Debasish Sinha6, Heli Skottman1, Kai Kaarniranta2. 1University of Tampere, BioMediTech, Tampere, Finland; 2Department of Ophthalmology, Institute of Clinical Medicine, University of Eastern Finland, Kuopio, Finland; 3SIB Labs, University of Eastern Finland, Kuopio, Finland; 4Department of Ophthalmology, University of Tampere, Tampere, Finland; 5SILK,TAUH Eye Center, Tampere University Hospital, Tampere, Finland; 6The Wilmer Eye Institute, The Johns Hopkins University School of Medicine, Baltimore, MD; 7 Department of Ophthalmology, Kuopio University Hospital, Kuopio, Finland. Purpose: The impairment in autophagic and proteasomal cleansing has been documented in retinal pigment epithelial (RPE) cells and These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record. ARVO 2016 Annual Meeting Abstracts age-related macular degeneration (AMD) pathology, but not studied with human embryonic stem cell (hESC)-RPE cells. Methods: Here the role of autophagy and proteolytic machinery was assessed in the regulation of melanocytic pigmentation with a tandem fluorescently tagged LC3 (GFP-mCherry-LC3) transfected or nontransfected mature hESC-RPE cells which were treated either with MG-132, AICA ribonucleotide, 5-aminoimidazole-4-carboxamide-1β-D-ribofuranoside (AICAR), bafilomycin A1 or their combinations. The effects were analysed by western blotting (WB), confocal and electron microscopy (EM) and calcein fluorescence. Results: Inhibition of proteasomal function with MG-132, or autophagy with bafilomycin A1 increased the accumulation of premelanosomes seen in EM. The upregulation of autophagy markers SQSTM/p62 and MAP1LC3A/LC3-II, and the perinuclear localization of them was observed in WB and confocal microscopy, respectively. The AMP-dependent protein kinase activator AICAR treatment decreased the proteasome inhibitor -induced accumulation of melanosomes observed in EM, and also eradicated the SQSTM/ p62 and LC3-II detected in WB. The autophagy flux was shown to be induced during the MG-132 proteasome inhibition, and was further enhanced by simultaneous AICAR treatment, observed in confocal microscopy with the GFP-mCherry-LC3. Bafilomycin A1 increased amount of autophagosomes, premelanosomes and LC3-II observed in EM, calcein fluorescence or WB. Conclusions: Our result suggests that proteasomal inhibition increases the expression of the autophagy flux markers SQSTM1/p62 and LC3-II and lead to elevated accumulation of autophagosomes and melanosomal granules in the hESC-RPE. Furthermore, the AMPK activator AICAR promoted cleansing of up-regulated melanin granules, SQSTM/p62 and LC3-II, indicating autophagic cleansing. These results reveal that autophagy machinery is functional in hESCRPE cells and regulates cellular pigmentation with proteasomes. Commercial Relationships: Kati M. Juuti-Uusitalo, None; Niko Kivinen, None; Johanna Viiri, None; Juha Hyttinen, None; Arto Koistinen, None; Hannu M. Uusitalo, None; Debasish Sinha, None; Heli Skottman, None; Kai Kaarniranta, None Program Number: 261 Poster Board Number: B0267 Presentation Time: 8:30 AM–10:15 AM Ano2 contributes to the outwardly rectifying Ca2+-dependent Clconductance in the retinal pigment epithelium Olaf Strauss, Nadine Reichhart, Susanne Keckeis. Experimental Ophthalmology, Charite University Medicine Berlin, Berlin, Germany. Purpose: The retinal pigment epithelium (RPE) expresses a variety of Cl channels which contribute to transepithelial transport of Cland water. The Ca2+-dependent Cl channel in the RPE can adopt the transepithelial transport to the functional needs by rises of intracellular free Ca2+ as second messenger. Patch-clamp data from different groups have described the Ca2+-dependent Cl channel in the RPE as an outwardly rectifying channel which shows properties of anoctamin-2 (Ano2), a member of a Ca2+-dependent ion channel family. The purpose of the study is to characterize the molecular identity of the Ca2+-dependent Cl channel in the RPE. Methods: Analysis of the expression pattern of Ano2 in the mouse retina and human ARPE-19 cell line; patch-clamp analysis of ATPinduced Cl- conductance in ARPE-19 cells; siRNA approach against Ano2. Results: In sagittal sections of the mouse retina, we found Ano2 in the basolateral membrane of the RPE. ARPE-19 cells showed Ano2 expression at the mRNA and the protein level. Stimulation of ARPE-19 cells with 500µM ATP, which leads to a transient increase in intracellular free Ca2+, transiently stimulated an outwardly rectifying Cl- conductance which showed reversal potentials close the Cl- equilibrium potential and was blocked by DIDS and niflumic acid. siRNA knock-down of Ano2 in ARPE-19 cells reduced the Ano2 protein and Ca2+-dependent Cl- conductance. Conclusions: Ano2 contributes to the basolateral Ca2+-dependent Cl – conductance in the RPE. Its biophysical properties correspond with that of previous publications. Thus Ano2 is a major Ca2+dependent Cl channel in the RPE. Commercial Relationships: Olaf Strauss, None; Nadine Reichhart, None; Susanne Keckeis, None Support: DFG grant STR480/14-1 Program Number: 262 Poster Board Number: B0268 Presentation Time: 8:30 AM–10:15 AM Evaluation of the proliferative capacity of canine retinal pigment epithelial cells harvested from different regions of the fundus Freya M. Mowat1, Jonathan Hash1, Philip Mzyk2, Jill Harned2, Steven Nagar2, Mary C. McGahan2. 1Clinical Sciences, North Carolina State University, Raleigh, NC; 2Molecular Biomedical Sciences, North Carolina State University, Raleigh, NC. Purpose: Regional susceptibility to disease of the foveomacular region is challenging to model in rodents due to the absence of a region of enhanced cone photoreceptor density in these species. The canine retina has a well-defined cone photoreceptor enriched region known as the area centralis. We tested the hypothesis that the canine central retinal pigment epithelium (RPE) is less capable of proliferation in response to stress in culture using an established tissue culture model. Methods: We harvested RPE cells from young adult healthy mixedbreed dogs (n = 6 eyes) following humane euthanasia. Cells were harvested and separate low-density cultures were made from the RPE of the area centralis, nasal visual streak, superior peripheral retina and inferior peripheral retina. As cultures approached confluency, cells were fixed and immunocytochemistry for RPE65 and the cell proliferation marker Ki67 was performed. We quantified the number of Ki67 positive nuclei compared with all nuclei (DAPI nuclear counterstain). A repeated measures ANOVA with Bonferroni post-test was used to compare values from the area centralis with other regions of the eye. Results: As expected, the RPE cells of the area centralis contained no observable melanosomes. Cells from all regions expressed RPE65, supporting an RPE cell-type. There was no significant difference in the density of the cells, at the time of initial culture (p = 0.27), or at the time of assay (p = 0.2). The area centralis region (6.1 ± 5.3% SEM) contained a significantly smaller proportion of proliferating cells than the superior peripheral retina (38.5 ± 9.1%, p <0.05). Conclusions: Mature retinal pigment epithelium cells of the canine area centralis region have reduced capacity to proliferate. Our results are consistent with the hypothesis that the retinal pigment epithelium of the cone-rich canine area centralis remains in a more quiescent state. Regional cultures of dog retinal pigment epithelium could be used to further study the underlying causes of regional susceptibility to foveomacular disease involving the RPE. Commercial Relationships: Freya M. Mowat, None; Jonathan Hash; Philip Mzyk, None; Jill Harned, None; Steven Nagar, None; Mary C. McGahan, None These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record. ARVO 2016 Annual Meeting Abstracts Program Number: 263 Poster Board Number: B0269 Presentation Time: 8:30 AM–10:15 AM Increased expression and apical distribution of betaA3/A1Crystallin in polarized RPE cells Hiroto Terasaki1, 2, Parameswaran G. Sreekumar1, Shozo Sonoda2, Christine Spee3, Taiji Sakamoto2, David R. Hinton3, 4, Ram Kannan1. 1 Arnold and Mabel Beckman Macular Research Center, Doheny Eye Institute, Los Angeles, CA; 2Ophthalmology, Kagoshima University Graduate School of Medical and Dental Sciences, Kagoshima, Japan; 3 Ophthalmology, Keck School of Medicine of the University of Southern California, Los Angeles, CA; 4Pathology, Keck School of Medicine of the University of Southern California, Los Angeles, CA. Purpose: While recent studies have shown that alpha-crystallins play an important role in retinal function, not much is known about expression and function of beta-crystallins in the retinal pigment epithelium (RPE). The purpose of this study was to investigate the expression of betaA3/A1-crystallin in human RPE cells and the effect of polarity and oxidative stress on its expression. Methods: Polarized human fetal RPE cells with a mean TER of 385 ± 13.1 Ω·cm2 were cultured following an established protocol in our laboratory (Sonoda et al. Nature protocols 2009). Initial microarray analysis of polarized and non-polarized H-RPE cells (Lonza, Walkersville, MD) was carried out using a commercial array service (Agilent Expression Array; Takara Bio, Yokkaichi, Japan). Expression of betaA3/A1-crystallin in polarized and non-polarized human RPE cells was assessed by western blot and real-time PCR. Apical/ basolateral localization of beta A3/A1 crystallin was determined by immunostaining. To study the effect of oxidative stress on expression of betaA3/A1 crystallin, polarized RPE cell were exposed to 500uM of H2O2 for 24 h and expression was determined by western blot and real-time PCR analysis. Results: Microarray analysis revealed that, among crystallins, betaA3/A1 crystallin was highly expressed in polarized RPE cells (about 6.5 times higher compared to non-polarized RPE cells). This finding was confirmed by western blot, confocal microscopy and realtime PCR which showed higher expression of betaA3/A1 crystallin in polarized RPE cells while non-polarized RPE cells had negligible expression. Oxidative stress caused a decrease in gene and protein expression of betaA3/A1 crystallin in polarized RPE cells. Conclusions: BetaA3/A1 crystallin was highly expressed in polarized RPE cells and its expression decreased with oxidative stress. The increased expression in the apical domain suggests a possible role for betaA3/A1 crystallin to protect the neural retina. Commercial Relationships: Hiroto Terasaki, None; Parameswaran G. Sreekumar, None; Shozo Sonoda, None; Christine Spee, None; Taiji Sakamoto, None; David R. Hinton, None; Ram Kannan, None Support: EY01545 and a grant from the Arnold and Mabel Beckman Foundation Program Number: 264 Poster Board Number: B0270 Presentation Time: 8:30 AM–10:15 AM Modulation of Epithelial Mesenchymal Transition (EMT) Through the Notch Signaling Pathway in Human Retinal Pigment Epithelial Cells Jinggang Yin1, Junming Yue2, Gunnan Zhao2, Wenying Huo2, Weihong Huo1, Edward Chaum1. 1Ophthamology, University of Tennessee Health Science Center, Memphis, TN; 2Pathology, University of Tennessee Health Science Center, Memphis, TN. Purpose: Epithelial mesenchymal transition (EMT) of the RPE plays a central role in the development of proliferative vitreoretinopathy (PVR), a blinding clinical complication of retinal detachment and trauma. The Notch signaling pathway has been shown to regulate the expression of EMT proteins; however, its role in PVR is unknown. We examined the impact of Notch signaling in the RPE using a genome editing CRISPR/cas9 approach to knock out Notch 2/3 expression in ARPE19 cells and evaluated its impact on EMT protein expression and cell phenotype. Methods: Two gRNAs were cloned into lentiviral CRISPR/Cas9 vectors and used to target the Notch1-4 receptors. Lentiviruses were packaged in HEK293FT cells and ARPE19 cells were transduced with lentiviral Notch KO constructs. Transfected cells were isolated using puromycin and KO status confirmed by sequencing. EMT protein markers were evaluated in the Notch KO cell lines by Western blotting, and the cell phenotype was examined including proliferation, migration, and susceptibility to apoptosis. Results: Disruption of Notch2 in ARPE19 cells leads to the upregulation of N-cadherin, β-catenin, Snai1, α-SMA and downregulation of the E-cadherin and ZO-1 compared to the CRISPR/Cas9 empty vector infected controls. Knockout of Notch2 promotes ARPE19 cell migration. Conclusions: Disruption of the Notch2 gene indirectly promotes EMT-associated gene expression in ARPE19 cells. Notch pathway signaling may play an important role in contributing to the PVR phenotype expressed by transformed RPE cells following retinal detachment. Commercial Relationships: Jinggang Yin; Junming Yue, None; Gunnan zhao, None; Wenying Huo, None; Weihong Huo, None; Edward Chaum, None Support: Research to Prevent Blindness, Plough Foundation, Hamilton Eye Institute NEI Core Grant for Vision Research (P30EY013080), US Army Medical Research and Materiel Command (W81XWH-15-1-0023) Program Number: 265 Poster Board Number: B0271 Presentation Time: 8:30 AM–10:15 AM A cell-based model to study wound repair in retinal pigmented epithelial cells YingHsuan Shih1, 2, Monte J. Radeke1, Pete J. Coffey1. 1Neuroscience Research Institute, University of California-Santa Barbara, Santa Barbara, CA; 2Molecular, Cellular, and Developmental Biology, University of California-Santa Barbara, Santa Barbara, CA. Purpose: In rodents, RPE can repair laser-induced wounds by migration and proliferation. Whether human RPE employs a similar mechanism is unclear. To gain further understanding on wound healing of differentiated human RPE, we developed a cell-based system to study RPE behavior, morphology, and gene expression in response to chronic wounds. Methods: Human fetal RPE were cultured on Electric Cell-substrate Impedance Sensing (ECIS) 96-well plates for 30-60 days to differentiate. Each well contains 2 electrodes, which can introduce elevated electrical pulses to kill overlying RPE and create 0.25mm2 wounds. To create chronic wounds, electrical pulses were applied to kill RPE once a day. TGFβ1 was applied to study the roles of TGFβ signaling pathway on RPE repair. Wound healing was monitored by real-time impedance recording. Cell proliferation was detected using EdU labeling and cell size was measured based on ZO-1 staining. Gene expression was analyzed by RNA-Seq. Results: Immediately after wounding, cell death was detected as an abrupt drop in impedance. After 3 hours, bystander cells began to repopulate the electrodes. After 15 hours, the wounds were enclosed by cells that originated from the peripheral. Both EdU+ and EdUcells were observed in/outside of the enclosed wounds indicating that monolayer repair resulted from cell proliferation and migration. Cell size and density remained stable following a single wound. Conversely, after 20 treatments, RPE became larger and less dense. These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record. ARVO 2016 Annual Meeting Abstracts After repeated treatments, gene expression of the whole population remained constant despite the substantial changes in cell behavior, morphology and cell numbers. These results suggest that monolayer repair resulted from localized signaling. Moreover, activation of the TGFβ signaling pathway repressed cell proliferation, prolonged repair, and led to larger and less dense RPE. Conclusions: Here we used a cell-based system to study the repair mechanism of differentiated human RPE in response to chronic wounding. We showed that human RPE employs both cell proliferation and migration to repair disruption of the monolayer. However, the capability to repair is reduced by chronic cell loss and activation of the TGFβ signaling pathway. This cell-based system not only allows us to dissect the detailed mechanisms that regulate RPE repair under different conditions, but may also provide a platform to understand the lack of repair in diseased RPE. Commercial Relationships: YingHsuan Shih, None; Monte J. Radeke, None; Pete J. Coffey Support: LA1-02086; Garland Initiative for Vision Research Grant Program Number: 266 Poster Board Number: B0272 Presentation Time: 8:30 AM–10:15 AM Fluorescence Lifetime Imaging Ophthalmoscopy of in-vitro retinal pigment epithelial cells under different glucose conditions Joachim Pruessner1, Natalie Blimke1, Lars Alt1, Gereon Huttmann2, Ralf Brinkmann1, 2, Yoko Miura2, 1. 1Medical Laser Center Lübeck, Lübeck, Germany; 2Institute of Biomedical Optics, University of Lübeck, Lübeck, Germany. Purpose: Fluorescence lifetime imaging ophthalmoscopy (FLIO) is a new method for measuring fluorescence lifetimes (FLT) of endogenous fundus autofluorescence (AF). The metabolically active retinal pigment epithelium (RPE) may largely contribute to the fundus AF signal, and thus it is of great importance to elucidate its FLT characteristics under different pathological conditions. The aim of this study is to utilize the FLIO system to investigate in-vitro RPE cells cultivated under different glucose conditions. Methods: Second passage confluent porcine RPE cells cultured on 12-mm Transwell® membrane inserts were used for the study. The cells were cultured under glucose concentrations of 1000 (low glucose: LG), 4500 (normal glucose: NG) or 9000 (high glucose: HG) mg/L for 6 days. FLT was measured on day 3 and day 6, using a prototype system of FLIO (Heidelberg Engineering), which is based on a laser scanning ophthalmoscope with excitation at 473 nm and two emission spectral channels (ch. 1: 498-560 nm, ch. 2: 560-700 nm). Measurements were performed by placing the membrane insert into a custom-made artificial eye model. RPE cell viability was evaluated using calcein-AM cell viability indicator. Results: There was no significant change in RPE cell viability among different glucose conditions. A biexponential fit was performed for FLT decay analysis, thus each FLT consists of the short (t1) and long (t2) components. Under LG and HG conditions, elongation of t1 and t2 was typically seen (e.g. in ch. 1 on day 3, t1 was 355 ps ± 6.6, 366 ps ± 12, and 368 ps ± 16, and t2 was 2101 ps ± 64, 2123 ps ± 129 and 2179 ps ± 100, under NG, LG and HG conditions, respectively). The apparent difference between LG and HG conditions was shown in tm (mean FLT) of both channels on both days, where tm under LG is significantly longer than under other conditions. Regarding component contribution ratio (a1/a2), LG and HG conditions led to the significantly higher a1/a2 of ch. 2 on day 6 (2.43±0.03 and 2.57±0.22, respectively), compared to the NG condition (2.15±0.18). Conclusions: Our original setup is useful for measuring FLT of cultivated RPE cells while maintaining cell viability. The results suggest that glucose concentration-induced alterations of RPE metabolic status can result in change of the length and contribution of autofluorescence lifetime components, which can be detected by using FLIO. Commercial Relationships: Joachim Pruessner; Natalie Blimke, None; Lars Alt, None; Gereon Huttmann, None; Ralf Brinkmann, None; Yoko Miura, None Support: Financial support by Heidelberg Engineering, Germany Program Number: 267 Poster Board Number: B0273 Presentation Time: 8:30 AM–10:15 AM Phagocytosis of photoreceptor outer segments regulates mTORC1 activity in the retinal pigment epithelium Bo Yu, Pei Xu, Zhen-Yang Zhao, Yan Chen. Ophthalmology and Visual Science, University of Texas Medical Branch, Galveston, TX. Purpose: Mechanistic target of rapamycin complex 1 (mTORC1) is a central regulator of metabolism and integrates environmental cues to adjust cellular metabolic activities in a timely manner. Retinal pigment epithelial (RPE) cells remove daily shed photoreceptor outer segments (POS) by phagocytosis. Degradation of ingested POS and recycling its components generate demanding metabolic loads to the RPE. In this study, we investigated whether POS can stimulate mTORC1 activity in the RPE and explored the underlying molecular mechanisms. Methods: RPE mTORC1 activity was monitored during morning burst of OS disc shedding, the time when synchronized phagocytosis occurs. RPE cells were harvested from C57BL6/J mice started 30 minutes before light on to 3 hours afterwards. mTORC1 activity was analyzed by monitoring the phosphorylation status of downstream substrate proteins S6 and 4EBP-1. Subcellular localization of mTORC1 components was measured by immunostaining of RPE flat mounts. For mechanistic studies, cultured human RPE cells were treated with purified porcine POS. To investigate the roles of lysosomes in POS-induced mTORC1 activation, cells were either treated with lysosome inhibitor chloroquine, or transfected with small interference RNA targeting p18, V-ATPase6V0C, V-ATPase6V1A, or SLC38A9. The activity and distribution of mTORC1 were then measured by western blot or immunostaining. Results: RPE mTORC1 was transiently activated during morning burst of phagocytosis. Treating RPE cells with purified POS also led to activation of mTORC1 in culture. Co-localization of rhodopsin and LAMP1 with mTOR, p18, or RagA was detected by immunostaining, indicating their distribution on phagolysosomes. In contrast to the response to nutrient signals, disrupting proton gradients of the lysosome or downregulating the V-ATPase transporter protein did not affect POS-induced mTORC1 activation. Conclusions: POS are physiological stimuli of mTORC1 in the RPE. The daily fluctuation in mTORC1 activity responding to POS can be critical in maintaining normal functions of the RPE and retina. Commercial Relationships: Bo Yu; Pei Xu, None; ZhenYang Zhao, None; Yan Chen, None Support: NIH grant EY 019706 and the International Retinal Research Foundation Program Number: 268 Poster Board Number: B0274 Presentation Time: 8:30 AM–10:15 AM hiPSC-derived RPE cells: Characterization of blood-retinal barrier properties and drug permeability Jenni J. Hakkarainen1, Julien Maruotti2, Aila Seppänen1, Brigitte Onteniente2, Giedrius Kalesnykas1, Mika Reinisalo1. 1R&D department, Experimentica Ltd., Kuopio, Finland; 2Phenocell, Evry, France. Purpose: The retinal pigment epithelium (RPE) has a central role in maintenance of the outer blood-retinal barrier (oBRB). The oBRB efficiently limits drug access to the retina from systemic circulation These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record. ARVO 2016 Annual Meeting Abstracts (i.e. choroidal blood). The purpose of this study was to investigate the permeability, tight junction proteins and oBRB properties of a novel in vitro RPE cell model, derived from human induced pluripotent stem cells (hiPSC). Methods: hiPSC-derived RPE cells (PCi-RPE1426, Phenocell, France) were cultured on permeable Transwell® cell culture inserts and the apparent permeability coefficient (Papp) values for standard molecules were determined. Expression of the tight junction proteins, occludin and ZO-1, was characterized in the PCi-RPE1426 model by immunostaining. In addition, tert-butyl hydroperoxide (tBHP)induced oxidative stress in PCi-RPE1426 cells was assessed by using a resazurin cell viability assay. Results: The experimentally determined Papp value for the low permeability standard molecule 6-carboxyfluorescein was 1.0×10-6 cm/s, representative of a tight paracellular barrier between PCiRPE1426 cells. The dynamic range was >14 in PCi-RPE1426 indicating high discrimination between high and low Papp values. Furthermore, PCi-RPE1426 cells showed low susceptibility to tBHPinduced oxidative stress (IC50 = 2.2 mM). Conclusions: We provide evidence that PCi-RPE1426 cells form differentiated cell monolayers with intercellular tight junctions characteristic of the oBRB. This correlated with a decreased Papp for the low standard molecule, representing a 3-7 fold tighter barrier compared to commonly used ARPE-19 cells. In addition, PCiRPE1426 cells were able to tolerate high levels of tBHP-induced oxidative stress suggesting a potent endogenous antioxidant defense system. Commercial Relationships: Jenni J. Hakkarainen, Experimentica Ltd.; Julien Maruotti, Phenocell (I), Phenocell; Aila Seppänen, Experimentica Ltd.; Brigitte Onteniente, Phenocell, Phenocell (I); Giedrius Kalesnykas, Experimentica Ltd. (S), Experimentica Ltd. (I), Experimentica Ltd.; Mika Reinisalo, Experimentica Ltd. These abstracts are licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International License. Go to http://iovs.arvojournals.org/ to access the versions of record.