BD CellFIX: Flow Cytometry Fixation Reagent
advertisement

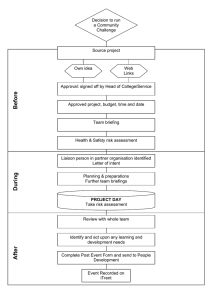
1. INTENDED USE BD CellFIX™ is intended for the fixation of human peripheral blood cell suspensions after immunofluorescence staining with monoclonal antibodies and prior to flow cytometric analysis. BD CellFIX™ 10X concentrate—Catalog No. 340181 2. SUMMARY AND EXPLANATION BD CellFIX is a premixed fixing concentrate, especially formulated for the treatment of samples prior to analysis with (automated) flow cytometers such as the BD FACS™ brand instruments. 3. PRINCIPLES OF THE PROCEDURE 03/2015 The interaction of BD CellFIX with human peripheral blood cells results in cross-linking of membrane-bound proteins. BD CellFIX effectively preserves stained human lymphocytes for at least 24 hours without significant alteration of lymphocyte subset enumeration values and fluorescence intensity results. The fixative may be used in conjunction with monoclonal antibodies conjugated to FITC, PE, PerCP, PerCP-Cy™5.5*, PECy™7, APC and APC-Cy7†. Prior to flow cytometric analysis, whole blood is stained with monoclonal antibodies, treated with BD FACS™ lysing solution to lyse erythrocytes, then washed and fixed with BD CellFIX. 23-12416-02 IVD BD, BD Logo and all other trademarks are property of Becton, Dickinson and Company. © 2015 BD Becton Dickinson Benelux N.V. Becton, Dickinson and Company Erembodegem-Dorp 86 B-9320 Erembodegem Belgium Tel 32.2.400.98.95 Fax 32.2.401.70.94 Benex Limited Pottery Road, Dun Laoghaire, Co. Dublin, Ireland Tel +353.1.202.5222 Fax +353.1.202.5388 BD Biosciences European Customer Support Tel +32.2.400.98.95 Fax +32.2.401.70.94 help.biosciences@europe.bd.com * Cy™ is a trademark of GE Healthcare. This product is subject to proprietary rights of GE Healthcare and Carnegie Mellon University, and is made and sold under license from GE Healthcare. This product is licensed for sale only for in vitro diagnostics. It is not licensed for any other use. If you require any additional license to use this product and do not have one, return this material, unopened, to BD Biosciences, 2350 Qume Drive, San Jose, CA 95131, and any money paid for the material will be refunded. † APC-Cy7: US patent 5,714,386 bdbiosciences.com ClinicalApplications@bd.com 1 capable of transmitting infection1,2 and dispose of with proper precautions in accordance with federal, state, and local regulations. Never pipette by mouth. Wear suitable protective clothing, eyewear, and gloves. 4. REAGENT Reagent Provided BD CellFIX 10X concentrate, 50 mL (sufficient for 1000 fixations) of a proprietary buffered solution. Precautions Dilution Instructions For use, dilute BD CellFIX 1:10 with reagent-grade (distilled and deionized) water. Store in a glass container at room temperature (20°C–25°C) for up to one month. Solution contains 10.0% formaldehyde, CAS number 50-00-0, 3.55% methanol, CAS number 67-56-1, and 0.93% sodium azide, CAS number 26628-22-8. Danger Storage and Handling When stored at 20°C–25°C, the reagent concentrate is stable until the expiration date shown on the label. Do not use after the expiration date. H311 Toxic in contact with skin. H331 Toxic if inhaled. H341 Suspected of causing genetic defects. H350 May cause cancer. Route of exposure: Inhalative. H371-H335 May cause damage to organs. May cause respiratory irritation. H373 May cause damage to the kidneys through prolonged or repeated exposure. Route of exposure: Oral. Alteration in the appearance of the reagent, such as precipitation or discoloration, indicates instability or deterioration. In such cases, the reagent should not be used. H318 Causes serious eye damage. 5. SPECIMEN COLLECTION AND PREPARATION Collect blood aseptically by venipuncture into a sterile BD Vacutainer® EDTA blood collection tube or equivalent. Store anticoagulated blood at room temperature (20°C–25°C) for up to 6 hours until ready for staining. H302 Harmful if swallowed. H315 Causes skin irritation. H317 May cause an allergic skin reaction. H413 May cause long lasting harmful effects to aquatic life. Wear protective gloves / eye protection. Wear protective clothing. Avoid breathing mist/vapours/spray. IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continue rinsing. IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing. IF SWALLOWED: Call a POISON CENTER/doctor if you feel unwell. CAUTION Use standard precautions when obtaining, handling, and disposing of all human blood samples and/or potentially carcinogenic reagents. 6. PROCEDURE Reagent provided BD CellFIX, 10X concentrate (Catalog No. 340181) WARNING All biological specimens and materials coming into contact with them are considered biohazards. Handle as if 2 use (IFU). Use care to protect tubes from direct light. Reagents and Materials Required but Not Provided • BD Vacutainer EDTA blood collection tubes or equivalent. • Falcon®‡ disposable 12 x 75-mm polystyrene test tubes or equivalent. • Micropipettor with tips. • 5- or 10-mL serological pipet. • BD monoclonal antibodies to human leucocyte antigens. • Vortex mixer. • Low-speed centrifuge (minimum speed 200g) with swinging bucket rotor with 12 x 75-mm tube carriers. • Vacuum aspirator with trap. • Reagent-grade (both distilled and deionized) water. • BD FACS lysing solution (10X) (Catalog No. 349202) • BD CellWASH™ (Catalog No. 349524) or a wash buffer of phosphate buffered saline (PBS) with 0.1% sodium azide. • 2. In the case of PBMCs, proceed to step 3. In the case of whole blood samples, lyse red blood cells following staining, using diluted BD FACS lysing solution (1X). Use care to protect tubes from direct light. Perform the procedure (incubation and centrifugation) as directed in the specific reagent data sheet or IFU or the BD FACS lysing solution IFU. Discard or aspirate the supernatant, leaving approximately 50 µL of residual fluid in the tube to avoid disturbing the pellet. 3. Vortex thoroughly at low speed to resuspend the cell pellet. Add 2 mL of BD CellWASH solution or 1X PBS with 0.1% sodium azide to the cell suspensions. Vortex thoroughly at low speed for 3 seconds. Centrifuge at 200g for 5 minutes at room temperature (20°C–25°C). 4. Discard or aspirate the supernatant, leaving approximately 50 µL of residual fluid in the tube to avoid disturbing the pellet. 1X PBS (Dulbecco’s modified PBS, pH 7.2 ±0.2, 0.01 mol/L PO4 and 0.15 mol/ L NaCl)3. This reagent does not contain calcium, magnesium, phenol red, or sodium azide. Filter PBS through a 0.2µm filter before use. Store at 2°C–8°C. 5. Vortex thoroughly at low speed to resuspend the cell pellet in the residual fluid. 6. Dilute room-temperature (20°C– 25°C) 10X BD CellFIX to 1X, following the instructions in Reagent, Section 4. Add 0.5 mL of 1X BD CellFIX solution to each tube except if the monoclonal antibody instructions for use specify other volumes. Vortex thoroughly at low speed for 3 seconds. Make sure that the cell suspension is well mixed with BD CellFIX. Preparing Samples 1. Stain whole blood or peripheral blood mononuclear cells (PBMCs) with monoclonal antibodies using the amounts, incubation times, and temperatures specified in the appropriate reagent instructions for ‡ Falcon is a registered trademark of Corning Incorporated. 3 7. Tubes are now ready to be analyzed on the flow cytometer. Cap or cover the prepared tubes and store at 2°C– 8°C in the dark until flow cytometric analysis. Analyze the fixed cells within 24 hours. Vortex the cells thoroughly at low speed to help reduce aggregation before running them on the flow cytometer. 8. PERFORMANCE CHARACTERISTICS Performance Results obtained for cell preparations fixed with BD CellFIX were compared to non-fixed cell preparations (PBS) and to cell preparations fixed with a freshly prepared fixative solution (1X PBS, 1% paraformaldehyde, 0.1% sodium azide). Blood samples of one donor were prepared for analysis with BD Multitest reagents CD3/CD8/CD45/CD4 and CD3/ CD16+CD56/CD45/CD19. Lymphocyte subsets (in %) were determined in nonfixed samples, in samples treated with the reference fixative solution, and in samples treated with 6 different BD CellFIX lots identified as a through f. Cells were analyzed immediately after preparation (T=0 h) and after 24 hours (T=24 h). CAUTION Some PE-Cy7 conjugates show changes in their emission spectra with prolonged exposure to paraformaldehyde. For overnight storage of stained cells, wash and resuspend in buffer without paraformaldehyde after 1 hour of fixation. 7. EXPECTED RESULTS Figure 1 % T lymphocytes in non-fixed blood cell preparations (--), in cell preparations fixed with 1% paraformaldehyde solution/0.1% sodium azide (reference fixative), and in blood cell preparations fixed with BD CellFIX lot a, b, c, d, e or f. % NK lymphocytes in non-fixed blood cell preparations (--),in cell preparations fixed with 1% paraformaldehyde solution/0.1% sodium azide (reference fixative), and in blood cell preparations fixed with BD CellFIX lot a, b, c, d, e, or f. % B lymphocytes in non-fixed blood cell preparations (--), in cell preparations fixed with 1% paraformaldehyde solution/0.1% sodium azide Aliquots of blood specimens from normal and abnormal donors were stained with BD Multitest™ reagents CD3/CD8/CD45/ CD4 and CD3/CD16+CD56/CD45/ CD19, then lysed with BD FACS lysing solution, washed, and fixed with BD CellFIX (see Section 6, Preparing Samples). Samples were analyzed on a BD FACSCalibur™ flow cytometer with BD Multiset™ software, and lymphocyte subset populations (in %) were determined. The reference ranges for the parameters studied are presented in the following table. Refer to Section 9 for limitations about reference ranges. Lymphocyte subset BD Multiset software (n=29) Reichert et al4 (n=271) Mean Reference range Mean Reference range %CD3+ 68 55–84 73 (6)a 61–85 %CD16+CD56+ 15 5–27 14 (6)a 6–29 %CD19+ 14 6–25 14 (4)a 7–3 a. Values given in parentheses as standard deviations (SD). 4 with BD CellFIX, and lymphocyte subsets were determined. Samples were run on the BD FACSCalibur instrument immediately after treatment (T = 0 h) and after 24 hours (T = 24 h). (reference fixative), and in blood cell preparations fixed with BD CellFIX lot a, b, c, d, e, or f. T=0h T=24h % T Lymphs, no fixation CellFIX lot f CellFIX lot e CellFIX lot d CellFIX lot c CellFIX lot b Mean SD %CVa 30 71 0.79 1.11 %CD16+CD56+ 30 12 0.63 5.30 %CD19+ 30 14 0.77 5.35 n Mean SD %CVa T=0h %CD3+ 30 72 0.81 1.12 T=24h %CD16+CD56+ 30 11 0.96 8.79 % NK Lymphs, no fixation %CD19+ 30 14 0.97 7.12 40 30 30 20 20 10 10 a. CV = coefficient of variation Accuracy To determine accuracy of BD CellFIX, flow analysis of BD CellFIX treated and non-BD CellFIX treated 4-color stained cell preparations was performed, and resulting lymphocyte subsets were compared. Aliquots of blood specimens from 29 normal or abnormal donors were stained, lysed, washed, and fixed with BD CellFIX and immediately run on a BD FACSCalibur flow cytometer. Aliquots of same blood specimens were prepared in a similar way for 4-color analysis but were not fixed with BD CellFIX. CellFIX lot f CellFIX lot e CellFIX lot d CellFIX lot c 0 CellFIX lot b 0 CellFIX lot a n %CD3+ a. CV = coefficient of variation T=0h T=24h % B Lymphs, no fixation 40 40 % B Lymphocytes Lymphocyte subset at T=0h Lymphocyte subset at T=24h 40 Reference Fixative % NK Lymphocytes CellFIX lot a 100 90 80 70 60 50 40 30 20 10 0 Reference Fixative % T Lymphocytes 100 90 80 70 60 50 40 30 20 10 0 30 30 20 20 10 10 0 Figure 2 % T lymphocytes in non-fixed blood cell preparations vs % T lymphocytes in cell preparations fixed with BD CellFIX; % NK lymphocytes in non-fixed blood cell preparations vs % NK lymphocytes in cell preparations fixed with BD CellFIX; % B lymphocytes in CellFIX lot f CellFIX lot e CellFIX lot d CellFIX lot c CellFIX lot b CellFIX lot a Reference Fixative 0 Precision Repeatability was assessed using the BD Multitest reagents CD3/CD8/CD45/ CD4 and CD3/CD16+CD56/CD45/ CD19. Thirty aliquots of the same blood sample were stained, lysed, and treated 5 non-fixed blood cell preparations vs % B lymphocytes in cell preparations fixed with BD CellFIX. % T Lymphocytes % T Lymphs in blood cell preparations fixed with CellFIX Expected values 100 90 80 70 60 50 40 30 20 10 0 EDTA is the anticoagulant of choice. BD Biosciences has limited information concerning use of other anticoagulants. • Samples should be processed within 6 hours of collection. Store blood samples in BD Vacutainer blood collection tubes or equivalent at room temperature prior to staining, lysing, and fixing. • Incubation times or temperatures other than those specified may be a source of error. • The volumes of reagent recommended are based on studies of normal human blood. y = 0,9774x + 1,1417 R 2 = 0,9894 0 10 20 30 40 50 60 70 80 90 100 % T Lymphs in non-fixed blood cell preparations % NK Lymphocytes WARRANTY Expected values Unless otherwise indicated in any applicable BD general conditions of sale for non-US customers, the following warranty applies to the purchase of these products. 40 % NK Lymphs in blood cell preparations fixed with CellFIX • y = 1,022x - 0,1176 30 R 2 = 0,9818 20 THE PRODUCTS SOLD HEREUNDER ARE WARRANTED ONLY TO CONFORM TO THE QUANTITY AND CONTENTS STATED ON THE LABEL 10 OR IN THE PRODUCT LABELING AT THE TIME OF DELIVERY TO THE CUSTOMER. BD DISCLAIMS HEREBY ALL OTHER WARRANTIES, 0 0 10 20 30 EXPRESSED OR IMPLIED, INCLUDING WARRANTIES OF 40 MERCHANTABILITY AND FITNESS FOR ANY PARTICULAR PURPOSE AND % NK Lymphs in non-fixed blood cell preparations NONINFRINGEMENT. BD’S SOLE LIABILITY IS LIMITED TO EITHER REPLACEMENT OF THE PRODUCTS OR REFUND OF THE PURCHASE PRICE. BD IS NOT LIABLE FOR PROPERTY DAMAGE OR ANY INCIDENTAL OR CONSEQUENTIAL DAMAGES, INCLUDING PERSONAL % B Lymphocytes INJURY, OR ECONOMIC LOSS, CAUSED BY THE PRODUCT. Expected values REFERENCES 50 40 % B Lymphs in blood cell preparations fixed with CellFIX 1. Protection of Laboratory Workers from Occupationally Acquired Infections; Approved Guideline—Third Edition. Wayne, PA: Clinical and Laboratory Standards Institute; 2005. M29-A3. 2. Centers for Disease Control. Perspectives in disease prevention and health promotion update: universal precautions for prevention of transmission of human immunodeficiency virus, hepatitis B virus, and other bloodborne pathogens in health-care settings. MMWR. 1988;37:377-388. 3. Jackson A, Warner N. Preparation, staining, and analysis by flow cytometry of peripheral blood leukocytes. In: Rose N, Friedman H, Fahey J, eds. Manual of Clinical Laboratory Immunology. 3rd ed. Washington, DC: American Society for Microbiology; 1986:226-235. y = 0,9774x + 0,3194 2 R = 0,9884 30 20 10 0 0 10 20 30 40 50 % B Lymphs in non-fixed blood cell preparations 9. LIMITATIONS • Each laboratory should establish normal ranges for leucocyte subsets using its own test conditions. 6 4. Reichert TA, DeBruyere M, Deneys V, et al. Lymphocyte subset reference ranges in adult Caucasians. Clin Immunol Immunopathol. 1991; 60:190-208. 7