Get PDF - Wiley Online Library
advertisement

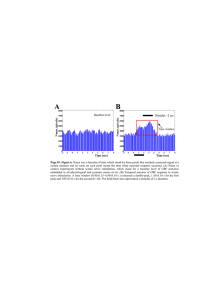
The Laryngoscope C 2014 The American Laryngological, V Rhinological and Otological Society, Inc. Stimulation Threshold Greatly Affects the Predictive Value of Intraoperative Nerve Monitoring Daniel L. Faden, MD; Lisa A. Orloff, MD; Tokunbo Ayeni, BS; Daniel S. Fink, MD; Katherine Yung, MD Objectives/Hypothesis: Using a standardized, graded, intraoperative stimulation protocol, we aimed to delineate the effects of various stimulation levels applied to the recurrent laryngeal nerve on the postoperative predictive value of intraoperative nerve monitoring. Study Design: A total of 917 nerves at risk were included for analysis. Intraoperatively, patients underwent stimulation of the recurrent laryngeal nerve at 0.3, 0.5, 0.8, and 1.0 mA followed by postoperative laryngoscopy for correlation with intraoperative findings. Methods: Sensitivity, specificity, positive predictive value, and negative predictive value were calculated at each stimulation level. Results: Sensitivity, specificity, positive predictive value, and negative predicative values ranged from 100% to 37%, 6% to 99%, 2% to 39%, and 100% to 99%, respectively at 0.3 to 1.0 mA. No demographic variables affected sensitivity or specificity. Receiver operating characteristic analysis identified 0.5 mA as the level of stimulation that optimizes sensitivity and specificity. Conclusions: The predictive value of intraoperative nerve monitoring varies greatly depending on the stimulation levels used. At low amplitudes of stimulation, nerve monitoring has high sensitivity and negative predictive value but low specificity and positive predictive value, related to the high rate of false positives. At high levels of stimulation, specificity and negative predictive value are high, sensitivity is low, and the positive predictive value rises as the rate of false negatives increase and the rate of false positives decrease. A stimulation level of 0.5 mA optimizes the predictive value of nerve monitoring; however, stimulation at multiple levels significantly improves the predictive value of intraoperative nerve monitoring. Key Words: Recurrent laryngeal nerve, intraoperative nerve monitoring, thyroid surgery. Level of Evidence: 2b. Laryngoscope, 125:1265–1270, 2015 INTRODUCTION Surgery that puts the recurrent laryngeal nerve (RLN) at risk, including thyroidectomy, parathyroidectomy and central neck dissection, is on the rise in the United States as the population ages and the incidence of thyroid cancer increases.1 Although occurring in only about 3% of patients,2,3 damage to one or both RLNs can result in significant morbidity including voice changes, dysphagia, dyspnea, or airway distress, and need for tracheostomy or other interventions. Previously published Additional Supporting Information may be found in the online version of this article. From the Department of Otolaryngology–Head and Neck Surgery (D.L.F., K.Y.) and School of Medicine (T.A.), University of California, San Francisco, San Francisco, California; Department of Otolaryngology– Head and Neck Surgery (L.A.O.), Stanford University, Stanford, California; and the Department of Otolaryngology–Head and Neck Surgery (D.S.F.), Louisiana State University Health Sciences Center, New Orleans, Louisiana, U.S.A. Editor’s Note: This Manuscript was accepted for publication September 15, 2014. Presented at the Triological Society 117th Annual Meeting at COSM, Las Vegas, Nevada, U.S.A., May 15–16, 2014. The authors have no funding, financial relationships, or conflicts of interest to disclose. * Send correspondence to Daniel Faden, MD, UCSF Department of Otolaryngology–Head and Neck Surgery, 2380 Sutter Street, First Floor, San Francisco, CA, 94115. E-mail: dfaden@ohns.ucsf.edu DOI: 10.1002/lary.24960 Laryngoscope 125: May 2015 studies have not shown that use of intraoperative nerve monitoring (IONM) decreases the rate of injury to the RLN2,3; however, use of IONM has become increasingly widespread. In recent surveys, approximately 80% of otolaryngologists and 48% of general surgeons reported using IONM, and 44% of otolaryngologists and 31% of general surgeons use IONM routinely.4,5 More recently, interest has focused on the predictive value of IONM for intraoperative decision making (i.e., decision to proceed with contralateral dissection after loss of IONM signal on the first side) and prognostication.3,6–9 These topics have garnered interest considering that <15% of injuries to the RLN are predicted intraoperatively.10–13 However, a lack of standardization of how IONM is used makes interpretation of the literature difficult. Although numerous studies have looked at the sensitivity and specificity of IONM, many of these studies have significant limitations, including small sample sizes, different IONM techniques, and lack of standardized and accurate assessment of RLN function postoperatively. To our knowledge no studies have examined the effects of various stimulation thresholds on the predictive value of IONM using an endotracheal tube surface electrode system. Thus, few practical guidelines exist as to what level of stimulation to apply to the RLN intraoperatively. In this study, using a standardized, graded, intraoperative stimulation protocol, we aimed to Faden et al: Predictive Value of Nerve Monitoring 1265 TABLE I. Definitions. Term Definition True positive Inability to stimulate RLN, abnormal VF function True negative False positive Intact stimulation of RLN, normal VF function Inability to stimulate RLN, normal VF function False negative Intact stimulation of RLN, abnormal VF function Sensitivity Specificity Ability to detect a damaged RLN Ability to detect a normally functioning RLN PPV Likelihood that when an RLN does not stimulate, VF function is abnormal NPV Likelihood that when an RLN stimulates, VF function is normal NPV 5 negative predictive value; PPV 5 positive predictive value; RLN 5 recurrent laryngeal nerve; VF 5 vocal fold. calculate predictive values of IONM at various stimulation levels to identify thresholds for maximizing postoperative predictive values. MATERIALS AND METHODS Patient Selection and Study Protocol Data were collected from the charts of 732 consecutive patients who underwent surgery that placed the right and/or left RLN at risk (thyroid surgery, parathyroid surgery, central neck dissection) at the University of California, San Francisco (UCSF) from 2005 to 2013. All patients underwent surgery by a single surgeon (L.A.O.). This retrospective cohort study was approved by the institutional review board at UCSF. IONM was performed using the Medtronic-Xomed (Minneapolis, MN) nerve integrity monitoring system (NIM version 2.0 or 3.0) in conjunction with the Medtronic-Xomed nerve integrity monitor electromyography endotracheal tube. All patients underwent preoperative laryngoscopy, and vocal fold (VF) mobility was documented as normal, paresis, or paralysis All patients were seen in follow-up 5 to 7 days after surgery and again underwent laryngoscopy and grading. If there was concern for abnormal VF mobility based on voice changes, abnormal indirect laryngoscopy, or difficult exam, the patient underwent laryngovideostroboscopy, and again the VF mobility was graded. In patients with an abnormal exam, laryngoscopy was repeated at 4- to 8-week intervals until recovery or until deemed stable. Patients who had an abnormal preoperative exam, had the RLN sacrificed or transected during surgery, did not have the RLN clearly identified during surgery, did not complete a follow-up appointment within 1 week of surgery, had incomplete documentation in their chart, or were <18 years old were excluded from analysis. Surgical Protocol Patients were orally intubated with a nerve integrity monitor endotracheal tube by an anesthesiologist. Confirmation of placement of the electrodes in contact with the VFs was done with direct visualization. The endotracheal tube was secured, with care taken not to rotate the tube. A shoulder roll was placed, and a "tap test" was used to ensure the circuit was intact. In accordance with the International Standards Guideline Statement,14 impedance values for each electrode were confirmed to be <5 kilohms, and the imbalance between electrodes<1 kiliohm. If these values were above the threshold, Laryngoscope 125: May 2015 1266 the tube was repositioned. The evoked potential threshold was set at 150 mV. This value was chosen based on prior experience of suboptimal signal-to-noise ratio, as well as occasional evoked events being triggered by low-level respiratory spontaneous waveforms, when a threshold of 100 mV was used. This value is within the range suggested by the International Standards Guideline Statement of 100 to 200 mV. After surgical dissection was complete, the most proximal portion of the nerve was identified and stimulated at 0.3 mA. If the nerve did not stimulate at 0.3 mA, the current was increased in a graded fashion to 0.5, 0.8, and 1.0 mA until stimulation was obtained. The electrodes were left in place at the end of the procedure until the patient was extubated so that those patients who exhibited spontaneous VF mobility could be identified. Statistical Analysis and Definitions Data analysis was performed in nine cohorts: normal VF motion versus abnormal (paralysis and paresis), normal VF motion versus paralysis, and normal VF motion versus paresis, for left-sided surgeries, right-sided surgeries, and again as a combined cohort, with the nerves treated as independent observations. Because patients who had bilateral surgery represented two data points that were treated as independent observations in the combined cohort, we calculated a kappa statistic. Kappa was 0.37, indicating fair agreement, and giving an actual percentage agreement of 97.5% compared to a chance level of agreement of 96.1%. This small difference was deemed acceptable for the purposes of this study. Calculations of sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and receiver operator characteristic (ROC) curves were calculated in the standard fashion using SAS statistical software (SAS 9.4; SAS Institute Inc., Cary, NC). If a nerve stimulated at a given level, all levels above that were assumed to be functional. Statistical significance was set at P <.05 using the Fisher exact test. Paralysis is defined as immobility of the VF due to nerve injury, paresis as decreased mobility of the VF due to nerve injury, and abnormal function as either paresis or paralysis. A positive result in our study was set as inability to stimulate the RLN intraoperatively. Table I lists the corresponding definitions used in this study. Because of the concern for bias in having the operating surgeon also grading postoperative VF motion, we utilized a reliability cohort to ensure accuracy. Fifty-two nerves (5% of the cohort) from patients who underwent laryngovideostroboscopy by the operating surgeon underwent blinded review and grading by an independent laryngologist (D.S.F.). These results were then compared with a kappa statistic to existing grading. The calculated kappa was 0.65. This is considered substantial interrater agreement and was deemed acceptable for this study. RESULTS A total of 614 patients met inclusion criteria. Fortynine percent of patients had bilateral surgery, resulting in a total of 917 nerves at risk. The mean patient age was 50.2 years (range, 18–96 years). Additional patient characteristics, and operative and postoperative statistics are described in Table II. Cases of paresis and paralysis were equally distributed between the left and right sides. In patients who suffered paresis or paralysis, spontaneous recovery occurred in 49%, on average, 38 days postoperatively. Of the patients with abnormal postoperative vocal fold motion, including those after RLN sacrifice, 55% had an intervention, most commonly Faden et al: Predictive Value of Nerve Monitoring TABLE II. Descriptive Statistics. Category No. % White Asian 442 89 73.9 14.7 Other 54 9.8 Black Gender 21 3.5 Race Female Male Previous surgery on the neck Yes No Previous radiation to head and neck No Yes Sternotomy 484 78.8 130 21.2 208 34.0 404 66.0 562 92.0 49 8.0 Yes 4 0.7 No RLN transected, total 610 6 99.4 0.7 RLN sacrificed, total 7 0.8 Principle diagnoses Papillary thyroid cancer 236 38.0 Parathyroid adenoma 97 16.0 Follicular adenoma Thyroiditis 75 42 12.0 7.0 Goiter 42 7.0 Other Neoplasm 122 20.0 338 55.1 276 4 44.9 0.9 No Yes Left VF paresis Left VF paralysis 7 1.5 Total abnormal function on left Right VF paresis 11 4 2.4 0.9 Right VF paralysis 9 2.0 Total abnormal function on right Total paresis 13 8 2.8 0.9 Total paralysis 16 1.7 Total abnormal function Bilateral paresis 24 1 2.6 0.1 Bilateral paralysis 3 0.3 RLN 5 recurrent laryngeal nerve; VF 5 vocal fold. injection laryngoplasty, with a median time to intervention of 51 days. Sensitivity, specificity, PPV, and NPV were calculated at 0.3-, 0.5-, 0.8-, and 1.0-mA stimulation levels for normal versus abnormal, normal versus paralysis, and normal versus paresis, for left, right, and combined cohorts, giving nine distinct cohorts at each stimulation level (see Supporting Table I in the online version of this article). No appreciable differences were observed between these cohorts; therefore, we focused our analyLaryngoscope 125: May 2015 sis on normal versus abnormal function in the left- and right-sided combined cohort. Sensitivity and specificity were inversely related. Sensitivity was 100% at 0.3 mA and dropped nearly linearly as the stimulation current increased to 37% at 1.0 mA (Fig. 1). Specificity was 6.0% at 0.3 mA but increased rapidly from 0.5 mA upward to 99% at 1.0 mA. PPV was poor at all stimulation levels but increased with increasing stimulation, whereas NPV was high at all levels (100%–98%). The ROC identified 0.5 mA as the optimal level of stimulation if sensitivity and specificity are weighted equally (nearest to 0.1 on Fig. 2). We examined the effects of selected variables (age, gender, tumor size, nodal status, previous neck surgery, previous radiation to the neck, malignant disease vs benign disease, need for sternotomy) on the sensitivity and specificity of IONM. There were no statistically significant differences in sensitivity or specificity for any of the variables. DISCUSSION Surgeries that place the RLN at risk are increasing in frequency in the United States as the average life expectancy increases and the incidence of thyroid cancer rises.1 IONM has gained popularity despite a lack of data showing that its use decreases RLN injury.2,3 More recently, studies have attempted to assess the value of IONM for intraoperative decision making, postsurgical monitoring, and long-term prognosis.3,6–9 However, there is considerable variability in methodology between these studies, making comparisons and thus the ability to draw meaningful, clinically relevant conclusions, challenging. For example, studies assessing the predictive value of IONM include various techniques for measurement of RLN function including laryngeal palpation,15 placement of intramuscular electrodes with stimulation of the RLN16 or vagus nerve,17 and more recently, endotracheal tube surface electrodes, as in our study. We identified five studies in the English literature that report predictive values for IONM using surface electrodes on the endotracheal tube, stimulation of the RLN, sample sizes greater than 100 and pre/postoperative laryngoscopy to confirm VF findings.9,18–21 Rates of sensitivity and specificity, according to our definitions, vary considerably in these studies (sensitivity 52%– 100%, mean 73%; and specificity 94%–99%, mean 96%). Four of these studies calculated PPV and NPV (PPV 29%–72%, mean 43%; and NPV 97%–100%, mean 99%).18–21 One, of many variables that differ between these studies is the level of stimulation applied to the RLN intraoperatively, ranging from 0.5 to 2 mA. Four of the five studies used multiple levels of stimulation, and one study used a single level.20 None of these studies examined the predictive value of IONM at various levels of stimulation within the same patient or between patients. In this study, we examined the predictive value of IONM at various levels of stimulation, within the same patient (internal control), in 917 nerves at risk. All nerves underwent stimulation at 0.3, 0.5, 0.8, and 1 mA, Faden et al: Predictive Value of Nerve Monitoring 1267 Fig. 1. Relationship of sensitivity, specificity, positive predicitive value (PPV), and negative predictive value (NPV) at 0.3, 0.5, 0.8, 1.0 mA stimulation levels for normal versus abnormal exam in a left- and rightsided combined cohort. [Color figure can be viewed in the online issue, which is available at www.laryngoscope.com.] or until stimulation was achieved. Sensitivity, specificity, NPV, and PPV were then calculated at each level. The starting value was chosen as 0.3 mA because it has been reported as the level at which the RLN first begins to stimulate if the nerve is dry and dissected free from fascia.14 0.3 mA is assumed to induce depolarization of only a subset of RLN fibers and is thus "subphysiologic." Furthermore, 0.8 mA is the level described in the literature as corresponding to complete depolarization of the RLN, and thus this was used as the initial level for confirmation of identification of the RLN. 1.0 mA was chosen as Fig. 2. Receiver operator characteristic curve for normal versus abnormal exam in a left- and right-sided combined cohort showing 0.5 mA as the optimal level of stimulation. [Color figure can be viewed in the online issue, which is available at www.laryngoscope.com.] Laryngoscope 125: May 2015 1268 the upper limit of stimulation as it has been considered in the literature as a safe "suprathreshold" level of stimulation.14 The total rate of VF motion impairment in our study was 2.6%, consistent with previously published series.2,3 Findings did not differ significantly between the left and right side or between paresis and paralysis. The relationships between sensitivity, specificity, PPV, and NPV are demonstrated graphically in Figure 1 (and numerically in Supporting Table IA in the online version of this article). In essence, at low amplitudes of stimulation (0.3 mA), nerve monitoring has high sensitivity and NPV but low specificity and PPV, meaning it is excellent at confirming that a nerve that stimulates intraoperatively will have normal function postoperatively, but is poor at predicting that a nerve that does not stimulate intraoperatively will not function postoperatively, due to the high rate of false positives. At high levels of stimulation (1.0 mA), the NPV and specificity are high, the sensitivity is low, and the PPV rises, meaning inability to stimulate the RLN at this level is a better predictor of loss of function than at lower levels as the rate of false positives decreases. Ability to stimulate the RLN remains a good predictor of normal postoperative function at this level; however, there is an increased rate of false negatives leading to a lower sensitivity. Predictive values from this study, despite stringent surgical protocols and internal control, show considerable variability between stimulation levels, suggesting the threshold of stimulation is paramount in the predictive value of IONM. The ranges in predictive values in this study parallel the ranges seen in our review of the literature above. When we combine values to estimate the best possible predictive values at all ranges of stimulation, our sensitivity, specificity, PPV, and NPV are 100%, 99%, 39%, and 100%, respectively. These values again closely Faden et al: Predictive Value of Nerve Monitoring approximate the mean values for similar studies, presented above. This broad data range highlights the importance of the level of stimulation when using IONM to guide intra- or postoperative decision making and is vital information for the operating surgeon. We used an ROC to identify the single threshold of stimulation to optimize sensitivity and specificity, which we found to be 0.5 mA (sensitivity 68% and specificity 84%). However, an ROC assumes that sensitivity and specificity are equally weighted in the eyes of the test user. In reality, the most important function of IONM is to capture all true positives, and thus sensitivity should be weighted more heavily than specificity. Although it is tempting to suggest that 0.3 mA would be an excellent level at which to simulate (100% sensitivity), the number of false positives (849) is unreasonably high. This is in agreement with the idea that 0.3 mA is subphysiologic. Sensitivity drops off significantly after 0.3 mA. Low sensitivity exists for numerous reasons in addition to this low level of stimulation, including malfunction of the IONM system due to tube malposition, transient nerve dysfunction due to traction injury, light soft tissue coverage (most common), or wetness of the nerve,14 among others. At 1.0 mA, stimulation of the RLN is a supraphysiologic event, implying simply that a portion of the RLNVF circuit is intact.14 As seen in our data, although the NPV remains high, stimulation at this level increases the number of false negatives, decreasing the sensitivity. Because of the dichotomous relationship of sensitivity and specificity at low- and high-stimulation levels, a graded stimulation protocol, as was done in this study, is advocated with use of the current IONM system. If a single value of stimulation is to be used, 0.5 mA optimizes predictive value. When we examined the effects of selected variables on sensitivity and specificity, no differences were seen. This implies the predictive value of IONM is not dependent on any intrinsic patient factors or on previous exposure of the RLN, despite revision surgery itself being riskier. This is in contrast to recent studies that report differences in predictive values based on pathology, among other factors.9,19 However, small samples sizes within these subgroups and lack of statistical analysis make interpretation of their data difficult. We acknowledge a number of limitations of this study. First, this is a retrospective cohort study, and thus analysis is confined to the information that exists in the medical chart. As is true for similar studies of IONM, low rates of nerve injury make the event being studied infrequent. Thus, our sample size may not have been adequate to analyze certain variables. The true rate of VF motion abnormality may have been underestimated in this study as laryngovideostroboscopy, the gold standard to identify abnormalities of VF motion, was not performed in all patients. Last, this study used a single evoked potential amplitude threshold of 150 mV. Several recent articles have focused on the potential importance of the evoked response in the predictive value of IONM.20,22 This variable was not examined in our study. The relationship between stimulation levels applied to Laryngoscope 125: May 2015 the RLN, the evoked response, and predictive measures of IONM remains a topic for further investigation. CONCLUSION Existing studies of IONM have not examined how stimulation levels applied to the RLN affect predictive values. Our data support the hypothesis that the predictive value of IONM varies greatly depending on the stimulation threshold. At low levels of stimulation, IONM has high sensitivity and NPV but low specificity and PPV. This means that IONM, at this level, is excellent at confirming that a nerve that stimulates intraoperatively will have normal function postoperatively, but is poor at predicting that a nerve that does not stimulate intraoperatively will not function postoperatively, related to the high rate of false positives. At high levels of stimulation, the NPV and specificity are high, the sensitivity is low, and the PPV rises, meaning the inability to stimulate the RLN at this level is a better predictor of loss of function than at lower levels, as the rate of false positive decreases. Ability to stimulate the RLN remains a good predictor of normal postoperative function at this level; however, the rate of false negatives increases leading to a lower sensitivity. A 0.5 mA stimulation level optimizes the sensitivity and specificity of nerve monitoring; however, stimulation at multiple levels increases the predictive value of IONM. BIBLIOGRAPHY 1. Howlader N, Noone AM, Krapcho M, et al. SEER cancer statistics review, 1975–2008. Available at: http://seer.cancer.gov/csr/1975_2008. Accessed March 1, 2014. 2. Higgins TS, Gupta R, Ketcham AS, Sataloff RT, Wadsworth JT, Sinacori JT. Recurrent laryngeal nerve monitoring versus identification alone on post-thyroidectomy true vocal fold palsy: a meta-analysis. Laryngoscope 2011;121:1009–1017. 3. Dralle H, Sekulla C, Lorenz K, Brauckhoff M, Machens A. Intraoperative monitoring of the recurrent laryngeal nerve in thyroid surgery. World J Surg 2008;32:1358–1366. 4. Sturgeon C, Sturgeon T, Angelos P. Neuromonitoring in thyroid surgery: attitudes, usage patterns, and predictors of use among endocrine surgeons. World J Surg 2009;33:417–425. 5. Ho Y, Carr MM, Goldenberg D. Trends in intraoperative neural monitoring for thyroid and parathyroid surgery amongst otolaryngologists and general surgeons. Eur Arch Otorhinolaryngol 2013;270:2525–2530. 6. Melin M, Schwarz K, Lammers BJ, Goretzki PE. IONM-guided goiter surgery leading to two-stage thyroidectomy—indication and results. Langenbecks Arch Surg 2013;398:411–418. 7. Perie S, Ait-Mansour A, Devos M, Sonji G, Baujat B, St Guily JL. Value of recurrent laryngeal nerve monitoring in the operative strategy during total thyroidectomy and parathyroidectomy. Eur Ann Otorhinolaryngol Head Neck Dis 2013;130:131–136. 8. Sadowski SM, Soardo P, Leuchter I, Robert JH, Triponez F. Systematic use of recurrent laryngeal nerve neuromonitoring changes the operative strategy in planned bilateral thyroidectomy. Thyroid 2013;23:329–333. 9. Eid I, Miller FR, Rowan S, Otto RA. The role of nerve monitoring to predict postoperative recurrent laryngeal nerve function in thyroid and parathyroid surgery. Laryngoscope 2013;123:2583–2586. 10. Poveda MD, G. Sitges-Serra A, Barczynski M, et al. Intraoperative monitoring of the recurrent laryngeal nerve during thyroidectomy: a standardized approach (part 1). World J Endocr Surg 2011;3:144–150. 11. Bergenfelz A, Jansson S, Kristoffersson A, et al. Complications to thyroid surgery: results as reported in a database from a multicenter audit comprising 3,660 patients. Langenbecks Arch Surg 2008;393:667–673. 12. Lo CY, Kwok KF, Yuen PW. A prospective evaluation of recurrent laryngeal nerve paralysis during thyroidectomy. Arch Surg 2000;135:204–207. 13. Patow CA, Norton JA, Brennan MF. Vocal cord paralysis and reoperative parathyroidectomy. A prospective study. Ann Surg 1986;203:282–285. 14. Otto RA, Cochran CS. Sensitivity and specificity of intraoperative recurrent laryngeal nerve stimulation in predicting postoperative nerve paralysis. Ann Otol Rhinol Laryngol 2002;111:1005–1007. 15. Hermann M, Hellebart C, Freissmuth M. Neuromonitoring in thyroid surgery: prospective evaluation of intraoperative electrophysiological Faden et al: Predictive Value of Nerve Monitoring 1269 16. 17. 18. 19. responses for the prediction of recurrent laryngeal nerve injury. Ann Surg 2004;240:9–17. Hamelmann WH, Meyer T, Timm S, Timmermann W. A critical estimation of intraoperative neuromonitoring (IONM) in thyroid surgery [in German]. Zentralbl Chir 2002;127:409–413. Chan WF, Lang BH, Lo CY. The role of intraoperative neuromonitoring of recurrent laryngeal nerve during thyroidectomy: a comparative study on 1000 nerves at risk. Surgery 2006;140:866–872; discussion 872–863. Chan WF, Lo CY. Pitfalls of intraoperative neuromonitoring for predicting postoperative recurrent laryngeal nerve function during thyroidectomy. World J Surg 2006;30:806–812. Genther DJ, Kandil EH, Noureldine SI, Tufano RP. Correlation of final evoked potential amplitudes on intraoperative electromyography of the recurrent laryngeal nerve with immediate postoperative vocal fold func- Laryngoscope 125: May 2015 1270 tion after thyroid and parathyroid surgery. JAMA Otolaryngol Head Neck Surg 2014;140:124–128. 20. Koulouris C, Papavramidis TS, Pliakos I, et al. Intraoperative stimulation neuromonitoring versus intraoperative continuous electromyographic neuromonitoring in total thyroidectomy: identifying laryngeal complications. Am J Surg 2012;204:49–53. 21. Randolph GW, Dralle H, Abdullah H, et al. Electrophysiologic recurrent laryngeal nerve monitoring during thyroid and parathyroid surgery: international standards guideline statement. Laryngoscope 2011; 121(suppl 1):S1–S16. 22. Caragacianu D, Kamani D, Randolph GW. Intraoperative monitoring: normative range associated with normal postoperative glottic function. Laryngoscope 2013;123:3026–3031. Faden et al: Predictive Value of Nerve Monitoring