Using a Note to File (NTF)
advertisement

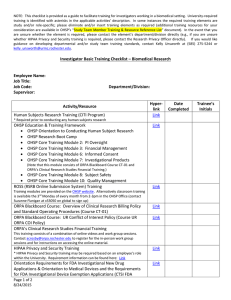
Office for Human Subject Protection 4th Quarter 2013 Contact us: Director OHSP: Kelley O’Donoghue 273-4631 Executive Director RSRB: Tiffany Gommel 273-4574 Director Research Education: Bill Kelvie 275-5244 RSRB Specialists: Emily Flagg, Sr. Specialist 276-5537 Kathleen Buckwell, Sr. Specialist 275-7446 Linda Palm-Montalbano Vol. 2, No. 4 CITI Information: The transition to CITI for initial certification for human subject protection was initiated on September 13, 2013. If you previously recertified with CITI, ensure you use your existing username and password to be directed to the appropriate program. If you have forgotten your username or password, please follow the instructions on the CITI login page. If you have any questions about which human subject protection program to complete, please contact OHSP. 273-4578 Igor Milosevic 273-2117 Michelle Giglio 273-4576 Kelly Unsworth 276-3856 Assistant Specialists: Jim Filingeri 273-4577 Nicole Mason 276-5544 Achieving High Quality Clinical Research Seminar Series November 19, 2013 “Review of the Investigational Drug and Device Regulations and Changes to the RSRB Online Submission System (ROSS) Application for Drugs and Devices” Director Quality Assurance: Kelley O'Donoghue and Kathleen Wessman 273-2118 Tiffany Gommel Quality Assurance Associate For questions regarding ROSS (RSRB Online Submission System) , please contact: Sue Flanigan 275-3050 Upcoming Educational Research Education News Time: 12:15 p.m.-1:15p.m. Place: Helen Wood Hall Auditorium Workshops December 4-6, 2013 “Conducting Human Subject Research” 3-day Workshop on GCP Techniques CLICK HERE: For more information and to register. OHSP OFFICE HOLIDAY CLOSURE: November 27th at 2pm November 28th and 29th December 24th at Noon December 25th December 31st at 2pm January 1st OHSP Website: www.rochester.edu/ohsp Office for Human Subject Protection Quality Improvement News Using a Note to File (NTF): Notes to file need to be well written, relevant, timely, and address the root causes of the problem. It is important the NTF identify how the root cause of the problem will be corrected, not just document what occurred. Notes to file can help a study team to recognize a bad pattern or trend, and lead to a change in policy and procedure, if necessary. Best practice is to include the NTF with the document to which it refers (subject or regulatory file). Ensure a consistent and logical system for filing all notes to file. Components of a NTF: NTF should be printed on department letterhead. Date: <When the NTF was written> To: <Protocol number and documented related file ‘Regulatory Binder or Subject File #’> From: <Name and title of person writing the NTF along with their signature> Issue: <Describe the problem> Root Cause: <Explain why the problem occurred> Corrective/Preventative Action: <Describe how the problem was corrected, by whom, when it was corrected, and how the problem will be prevented in the future. If a problem cannot be corrected, explain why it cannot be corrected, e.g. missing data. > Additional Information: <comments or other relevant information> NOW AVAILABLE!!!! Study Documentation Tool Box: OHSP-QI is pleased to announce a new resource for PIs and Study Coordinators! Research study forms and templates can be accessed here: http://www.rochester.edu/ohsp/qualitystudyDocumentationToolBox.html Research QI ‘Gold Star’ Award!!!!! The QI division would like to recognize Dr. Jill Halterman and the Preventive Care Program for Urban Children with Asthma Program members for their quality work on the ‘School-Based Telemedicine Enhanced Asthma Management: A Randomized Control Trial Using Novel Technology to Improve Preventive Asthma Care’ trial. This dynamic study team also includes Maria Fagnano and Reynaldo Tajon, and is located in the Saunders Research Building. They received the ‘Gold Star’ award for demonstrating attention to regulatory compliance, exceptional subject recruitment/retention practices, teamwork amongst site staff, and high quality, organized study-related documentation. CONGRATULATIONS!!!! Office for Human Subject Protection Research Subjects Review Board News HAPPY HOLIDAYS from the RSRB!! RSRB Submission During the Holidays The holidays are fast approaching and we would like to remind you of the importance of submitting continuing reviews a little earlier than usual to ensure uninterrupted RSRB approval. For those studies that require full board review, a list of upcoming meeting dates is noted below. Keep in mind that agendas fill up quickly especially at this time of the year. Please contact your RSRB Specialist with any questions about submission requirements or timelines. November Board 01: Board 02: Board 03: Board 04: Board 05: November 12th & 26th Cancelled November 19th November 14th & 21st November 13th December Board 01: Board 02: Board 03: Board 04: Board 05: 18th) Investigators leaving the University of Rochester Principal Investigators (PI) leaving the institution are responsible for notifying the RSRB well in advance of their departure so arrangements can be made to either close the study or name another appropriately qualified individual at the institution to serve as PI. Keeping Your Email Address Up to Date It is critical that your email address and other contact information is UP-TO-DATE date in the RSRB Online Submission System (ROSS). If your email address is not accurate or you do not frequently check your email, you may miss important email notifications, reminders, change requests, etc. Please contact the OHSP office to ensure that your contact information is accurate. Study Personnel Amendments in ROSS th December 10 December 5th December 3rd & 17th December 5th, 12th, & 19th December 11th (possibly move to The name of the person who will be added, that individuals certification and role on the study MUST BE included in the amendment request form when adding study personnel to your study. In addition you MUST go into the “Amended Application” and add the person to the appropriate section. This is routinely section 1.6 or 85.1. If this is not done, the amendment request will be returned to make these changes.