Small focal cortical dysplasia lesions are located at the bottom of a
advertisement

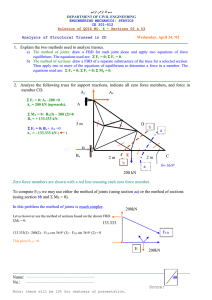
doi:10.1093/brain/awn224 Brain (2008), 131, 3246 ^3255 Small focal cortical dysplasia lesions are located at the bottom of a deep sulcus Pierre Besson, Frederick Andermann, Francois Dubeau and Andrea Bernasconi Department of Neurology and Neurosurgery and Brain Imaging Center, McGill University, Montreal Neurological Institute and Hospital, Montreal, Quebec, Canada Correspondence to: Andrea Bernasconi, MD, Montreal Neurological Hospital and Institute, 3801 University Street, Montreal, Quebec, Canada H3A 2B4 E-mail: andrea@bic.mni.mcgill.ca Keywords: epilepsy; focal cortical dysplasia; sulcal morphology; MRI; image analysis Received May 9, 2008. Revised August 11, 2008. Accepted August 18, 2008. Advance Access publication September 23, 2008 Introduction Taylor-type focal cortical dysplasia (FCD) (Taylor et al., 1971), a malformation due to abnormal neuroglial proliferation and organization (Barkovich et al., 2005), is an important cause of focal and often pharmacologically intractable epilepsy (Frater et al., 2000; Sisodiya, 2000, 2004). The term FCD designates a spectrum of histological abnormalities of the laminar structure of the cortex variably associated with cytopathological features, such as giant (or cytomegalic) neurons, dysmorphic neurons and balloon cells (Prayson et al., 2002; Palmini et al., 2004). Improvements of MRI techniques have allowed the in vivo recognition of FCD in an increasing number of patients (Barkovich and Raybaud, 2004a; Bernasconi, 2004). The most frequently described imaging features are cortical thickening, blurring of the grey–white matter (GM–WM) junction, and a hyperintense signal within the dysplastic lesion and its underlying white matter (Barkovich and Kuzniecky, 1996; Bronen et al., 1997; Colliot et al., 2006a). In patients with obvious malformations of cortical development gross sulco– gyral abnormalities may be noted and are characterized by a spectrum of changes ranging from clefts of various depth (Bronen et al., 2000) to broad gyri, shallow or deep sulci, or gyral simplification (Raymond et al., 1995; Yagishita et al., 1997). In many patients, however, FCD lesions go unrecognized by standard radiological analysis due to their subtlety. In their original observation, Taylor et al. (1971) noted that in all patients the cortical surface of the fixed tissue appeared normal to the naked eye and that histologically, in most cases, grotesque glial cells were present in the depths ß The Author (2008). Published by Oxford University Press on behalf of the Guarantors of Brain. All rights reserved. For Permissions, please email: journals.permissions@oxfordjournals.org Downloaded from http://brain.oxfordjournals.org/ by guest on September 30, 2016 Focal cortical dysplasia (FCD) is often characterized by minor structural changes that may go unrecognized by standard radiological analysis.Visual assessment of morphological characteristics of FCD and sulci harbouring them is difficult due to the complexity of brain convolutions. Our purpose was to elucidate and quantify the spatial relationship between FCD lesions and brain sulci using automated sulcal extraction and morphometry. We studied 43 consecutive FCD patients using high-resolution MRI. Lesions were classified into small and large using qualitative (detection on initial clinical assessment of conventional MRI) and quantitative (volume) criteria. Sulci were identified and labelled automatically using an algorithm based on a congregation of neural networks. Segmented FCD lesions and sulci were then simultaneously visualized in 3D. We measured mean and maximum depth of sulci related to each FCD and of the corresponding sulci in 21 healthy controls. In addition, we calculated sulcal depth within the FCD neighbourhood. Twenty-one (21/43 = 49%) patients had small FCD lesions (volume range: 128^3093 mm3). Among them, 17 (81%) had been overlooked during initial radiological evaluation and were subsequently identified using image processing. Eighteen (18/21 = 86%) small FCD lesions were located at the bottom of a sulcus. Two others were related to the walls of two sulci and one was located at the crown of a gyrus. Mean and maximum depth of sulci related to the FCD was higher than that of the corresponding sulci in controls (P _ 0.008). Sulcal depth within lesional neighbourhood had larger mean depth than that of the entire sulcus (P _ 0.0002). Evidence that small FCD lesions are preferentially located at the bottom of an abnormally deep sulcus may be used to direct the search for developmental abnormalities, particularly in patients in whom large-scale MRI features are only mildly abnormal or absent. Bottom of a sulcus cortical dysplasia Methods Subjects We studied 43 consecutive patients with FCD (21 males, mean age SD = 24 10 years) who had been investigated at the Montreal Neurological Institute and Hospital for refractory focal epilepsy. All patients had routine EEG recordings and prolonged video-EEG monitoring. The EEG background was abnormal in 34 patients (79%). Maximum interictal spike activity was concordant with the location of FCD lesions in 31 patients (72%). Ictal discharges recorded on surface EEG had localizing value in 24 patients (56%). Invasive monitoring using stereotactic implanted depth electrodes (SEEG) was performed in nine patients because EEG data and imaging did not sufficiently converge to plan the resection. In all of them SEEG demonstrated a very active, at times continuous interictal epileptic activity and focal electrographic changes at seizure onset in the electrodes close to or in the lesion. Patients were compared to 21 age-matched healthy individuals (7 males, mean age 23 9 years). The Ethics Board of the Montreal Neurological Institute and Hospital approved the study and written informed consent was obtained from all participants. Twenty-nine patients (29/43 = 67%) underwent resective surgery and the FCD was confirmed by histopathology in all. Mean post-operative follow-up was 6.4 years (median = 7.0 years). Twenty-one patients (21/29 = 72%) became free of disabling seizures (Engel’s class I) (Engel et al., 1993), four had rare disabling seizures (class II), three a worthwhile improvement (class III) and one no worthwhile improvement (class IV). Histological features of the resected tissue were graded as proposed by Palmini et al. (2004). In this scheme, type I dysplasias are characterized by isolated architectural abnormalities (Ia) 3247 or by an association of cortical dyslamination and giant or immature neurons (Ib). Type II dysplasias consist of architectural abnormalities with dysmorphic neurons, but without balloon cells (IIa) or with dysmorphic neurons and balloon cells (IIb). According to this classification, 20 of our patients had FCD IIb, six FCD IIa and three had FCD Ib. Three patients await operation. Three patients refused surgery and one with normal neurological examination was not operated because the lesion encroached on the dominant sensorimotor hand area. Two more patients were operated in a centre abroad and were lost for followup. In five others, seizures were controlled with antiepileptic medication. The FCD lesions were located in the frontal lobe in 20/43 (47%) patients, the central area in 10/43 (23%), the parietal lobe in 8/43 (19%), the Sylvian region in 2/43 (5%), the occipital lobe in 1/43 (2%), in the posterior temporal lobe in 1/43 (2%) and in the insular cortex in 1/43 (2%). MRI acquisition and image pre-processing MR images were acquired on a 1.5 T Gyroscan (Philips Medical Systems, Eindhoven, Netherlands) using a 3D T1-fast field echo sequence (TR = 18; TE = 10; NEX = 1; flip angle = 30; matrix size = 256 256; FOV = 256; slice thickness = 1 mm), providing an isotropic voxel size of 1 1 1 mm3. Each image underwent automated correction for intensity non-uniformity and intensity standardization (Sled et al., 1998). Images were then linearly registered into a standardized stereotaxic space based on the Talairach atlas (Talairach and Tournoux, 1988; Collins et al., 1994). We chose the ICBM 152 T1-weighted target as template for linear registration and subsequent tricubic resampling (Mazziotta et al., 1995). As part of our clinical protocol, coronal and axial protondensity (PD) and T2-weighted images (thickness 3.0–5.0 mm, gap 0.3 mm, TR 2100 ms, TE 20, 78 ms) were obtained in all patients and showed an increase in signal within the lesion in 26 of them. Coronal fluid attenuation inversion recovery images (FLAIR, slice thickness 3.0 mm, inter-slice gap 0.3 mm, TR 6000 ms, TE 150 ms, TI 1900 ms, FOV 230 mm) were available for analysis in 37 patients and showed increased signal in 17 of them. A linear or funnel-shaped abnormal signal intensity extending from the cortical-white matter junction to the margin of the lateral ventricular surface, i.e. transmantle dysplasia (Barkovich et al., 1997), was seen in five patients. A lesion was defined as ‘overlooked’ if it had not been identified during the initial routine clinical assessment of conventional MRI. Overlooked lesions were subsequently identified with the help of our image processing tools, including curvilinear reformatting (Bastos et al., 1999), texture analysis based on FCD computational models (Bernasconi et al., 2001), voxel-based morphometry (Colliot et al., 2005), and co-registration of multispectral images (Mahvash et al., 2006). We previously demonstrated that these techniques yield a significantly increased sensitivity in detecting FCD lesions relative to conventional MRI by maintaining high specificity. Automatic sulcal extraction and labelling Figure 1 illustrates the main steps involved in the automated sulcal extraction and labelling as described below. Downloaded from http://brain.oxfordjournals.org/ by guest on September 30, 2016 of the affected cortex and subjacent white matter. In two patients in whom no macroscopic abnormality was found, the authors indicated that: ‘the crown of the affected gyrus was spared, the anomalous neurons being concentrated around the base of the sulcus’. More recent MRI-based clinical observations also suggest that FCD may be located in the depth of a sulcus (Bastos et al., 1999; Urbach et al., 2002; Barkovich and Raybaud, 2004b). To date, however, no study has evaluated systematically the exact relationship between FCD lesions and brain sulci. The visual identification of morphologic sulcal characteristics is difficult on orthogonal planes obtained in conventional MRI. This disadvantage is bypassed when using precise and robust computer-based identification algorithms that allow automatic labelling on the brain surface (Mangin et al., 1995; Riviere et al., 2002) and spatial relationship between lesions and brain sulci. Moreover, they allow quantified comparison of inter-subject sulcal morphometry. Using these techniques, we were able to quantify sulcal patterns of the basal temporal lobe and demonstrated that a single-branched, unbroken collateral sulcus is the predominant folding pattern found in patients with temporal lobe epilepsy (Kim et al., 2008). In the present study, we sought to elucidate and quantify the spatial relationship between FCD lesions and brain sulci using automated sulcal extraction and morphometry. Brain (2008), 131, 3246 ^3255 3248 Brain (2008), 131, 3246 ^3255 Generation of brain surfaces and sulcal models For sulcal extraction, images were processed using BrainVISA. This image analysis software allows to reconstruct the surfaces corresponding to GM–WM and GM–CSF interfaces and to extract the brain sulci (Riviere et al., 2002). To compute accurate cortical surfaces and brain sulcal folds, image processing includes the following steps: (i) brain segmentation; (ii) classification of WM, GM and CSF generating separate maps in each hemisphere; (iii) reconstruction of the surfaces corresponding to the GM–WM and GM–CSF interfaces using the above classification maps and (iv) extraction of the sulcal folds by segmenting the skeletonized GM–CSF interface into simple surfaces. Sulcal labelling To place the threshold, we used entropy, a measure of homogeneity. This enabled us to maximize the ‘information gain’, which is the gain in homogeneity after thresholding. The entropy of our dataset S is defined by: EntropyðSÞ ¼ p log2 p ð1 p Þlog2 ð1 p Þ where p = number of lesions seen on MRI divided by total number of lesions. By setting a threshold T, our dataset was divided into two sets, S1 and S2. S1 grouped lesions of volume smaller than T and S2 grouped larger lesions. The information gain is given by: GainðS,T Þ ¼ Entropy ðSÞ Entropy ðS1 Þ þ Entropy ðS2 Þ The threshold for which the gain is maximized ensures that the sum of entropies of S1 and S2 is minimized. In other words, the probabilities of the events ‘lesion is overlooked if smaller than T ’ and ‘lesion is seen if larger than T ’ are maximized simultaneously. Criteria to categorize FCD position with respect to sulci Spatial correspondence between an FCD lesion and sulci related to it was assessed by synchronized visual inspection of 2D MR images and 3D surface rendering of that image. An FCD lesion was considered to be located at the bottom of a given sulcus (from now on identified as the FCD sulcus) if centred at the highest curvature of its sulcal pial surface and spreading along its walls. Criteria to separate small from large FCD lesions First, an expert observer manually segmented all FCD lesions as previously described (Colliot et al., 2006b). In 18 patients, two observes had segmented the same lesions to assess accuracy. Details about similarity indices, which showed good agreement between raters, are published elsewhere (Colliot et al., 2006b). We then separated small and large FCD lesions by placing a threshold along the volume continuum based on quantitative and qualitative criteria. The small lesion group was defined to include lesions with a volume below the threshold (as defined below) and to have high rate of overlooked lesions. Measure of sulcal depth To calculate the depth of a given sulcus, we first obtained the sulcal bottom (‘fundus’) line, which is defined by the inner edge of the sulcus (Cachier et al., 2001) as shown in Fig. 2. The depth of each point along this line was determined by calculating the shortest distance computed using the Chamfer’s distance transform (Borgefors, 1996) from the outer cortical surface (Smith, 2002). For controls and patients, we evaluated mean and maximum sulcal depth (Fig. 2B). Mean depth was calculated by averaging the depth of all points along the sulcal bottom (i.e. fundus) line, Fig. 1 Automated sulcal identification and labelling (see methods for details). (A) Grey matter (GM) and white matter extraction overlaid on the T1-weighted MRI; (B) skeletonization of the GM^ CSF union; (C) automatically colour-labelled sulci are shown on the 3D MRI surface rendering of a healthy control subject. The right central sulcus (red) is extracted for better visualization. Downloaded from http://brain.oxfordjournals.org/ by guest on September 30, 2016 After extraction, sulci are automatically labelled using a congregation of neural networks trained on a database of manually identified sulci by maximizing similarity of sulci features and sulci relations (Rivière et al., 2000). In each healthy control, we identified the sulcus that matched the one related to the FCD in each patient. Moreover, by simultaneously inspecting individuals’ sulcal morphology and labelling, we ensured the best possible sulcal equivalence between subjects. The sulcal nomenclature provided by Brain-VISA in the labelling step was verified on the atlas of Ono et al. (1990). P. Besson et al. Bottom of a sulcus cortical dysplasia Brain (2008), 131, 3246 ^3255 3249 which is defined by the inner edge of a given sulcus. The deepest point of the sulcal bottom line determined the sulcal maximum depth. Our purpose was to generate parameters for global and local morphology providing complementary information about sulcal depth. Indeed, for a given sulcus, depending on its shape, high value of maximum depth does not necessarily imply high mean sulcal depth value, and conversely. Thus, these two measurements are not directly comparable to each other. We also calculated the average depth of the portion of the sulcal bottom line located in the neighbourhood of the lesion, as determined by a 4-mm dilation of the FCD label (Fig. 2C). Statistical analysis We compared mean depth in patients to mean depths in controls using a one-tailed Wilcoxon signed-rank test. Similarly, maximum sulcal depth in patients was compared to maximum sulcal depth in controls. In addition, the sulcal portion within neighbourhood of the lesion was compared to the mean depth of the whole sulcus. EEG data of patients with small and large FCD lesions were compared using two-tailed Fisher exact test. Results The FCD volumes ranged from 128 mm3 to 94620 mm3 (mean SD: 7731 14891 mm3). Based on the entropy measurement described above, small lesions were defined as those having a volume smaller than T = 3093 mm3 (Fig. 3). Using this threshold, 21/43 (49%) FCD lesions were considered to be small (range: 128–3093 mm3; mean: 1282 852 mm3). Separating groups according to the size of FCD lesions was an efficient method, as demonstrated by the maximization of the information gain that allowed us to form two uniform groups: 17/21 (81%) of small lesions had been overlooked versus 0% of large lesions. Details about sulcal depth values in controls and patients are presented in Table 1. 18/21 (86%) small FCD lesions were located at the bottom of a sulcus. Among them, one lesion was at the bottom of two neighbouring sulci, giving a total of 19 FCD sulci. Fig. 3 Plot showing information gain as function of threshold (T) location. Small lesions were defined as those having a volume below T = 3093 mm3 (red line). To identify potential outliers in healthy controls, we used a leave-one-out procedure and compared the mean and maximum depths of all sulci corresponding to those harbouring the FCD in patients using a two-tailed Wilcoxon signed-rank test. This procedure revealed that sulci were deeper only in one healthy control (P = 0.02) and shallower in another (P = 0.02). Mean and maximum depth values of the FCD sulci were higher than those of the corresponding sulci in healthy controls (P50.008). Moreover, the sulcal portion within the neighbourhood of the lesion had larger mean depth than that of the whole sulcus (P50.0002). Figure 4 shows three representative examples of small, histologically proven FCD lesions located at the bottom of deep sulci. Although the proportion of patients with interictal EEG spikes concordant with the FCD location was lower in patients with small than large lesions, the difference was not significant (57% versus 86%, P = 0.5). Similarly, we found Downloaded from http://brain.oxfordjournals.org/ by guest on September 30, 2016 Fig. 2 Sulcal depth measurements. (A) Three-dimensional surface rendering of the MRI of a patient with right frontal focal cortical dysplasia. The intermediate right frontal sulcus (turquoise) and the lesion (red) are shown in semi-transparent. (B) The sulcal bottom line (yellow) was used to calculate mean and maximum depth (see methods for details). (C) We calculated the average depth of the portion of the sulcal bottom line located in the lesional neighbourhood (purple), as determined by a 4 mm dilation of the FCD lesion label (red wireframe in B and C). 3250 Brain (2008), 131, 3246 ^3255 P. Besson et al. Table 1 Mean and maximum depth (in millimetres), and z-scores (in parenthesis) of FCD sulci presented by lobes and regions Patients Frontal lobe Inferior frontal, L Inferior pre-central, L Intermediate frontal, L Pre-central L Intermediate frontal, RA Pre-central, R Superior frontal, R Central, RC Parietal lobe Intraparietal, L Intraparietal, R Sylvian region Sylvian fissure, L Insular region Insular, R Mean Max Neighbourhood Mean SD Max SD 8.6 (0.6) 9.9 (0.5) 11.7 (2.0) 6.0 (1.1) 12.1 (1.8) 18.1 (3.2) 11.2 (0.5) 12.6 (0.7) 11.5 (0.2) 10.5 (0.2) 11.8 (0.4) 16.9 (0.6) 20.7 (0.6) 17.6 (1.1) 11.8 (2.1) 20.3 (2.6) 22.2 (2.1) 19.7 (1.2) 20.1 (1.0) 19.9 (0.9) 17.9 (0.2) 20.3 (1.1) 13.6 (2.6) 18.9 (2.6) 10.4 (1.0) 7.7 (2.1) 18.1 (5.4) 20.2 (4.0) 14.2 (1.7) 18.3 (3.0) 13.9 (1.2) 11.1 (0.1) 15.0 (1.6) 9.5 1.6 11.3 2.9 9.1 1.3 4.3 1.6 9.0 1.7 9.9 2.6 18.9 3.1 18.1 4.3 15.6 1.8 7.1 2.2 14.9 2.1 16.2 2.9 10.9 2.5 17.5 2.6 11.5 (1.2) 12.2 (0.7) 14.0 (0.6) 13.6 (0.2) 21.1 (0.6) 21.6 (0.3) 24.0 (1.4) 23.3 (0.9) 11.8 (1.0) 18.2 (3.6) 18.6 (5.2) 15.7 (2.3) 13.2 1.4 22.1 1.6 13.4 1.0 22.1 1.4 18.6 (3.4) 13.3 (1.1) 32.1 (3.6) 21.7 (1.8) 27.3 (5.7) 20.3 (6.9) 6.1 3.7 12.0 1.2 10.4 6.0 25.2 2.0 11.7 (0.4) 20.8 (0.7) 16.3 (1.4) 9.9 4.5 16.4 6.7 15.6 (0.6) 32.1 (1.5) 12.3 (0.1) 12.7 5.2 20.1 7.8 Note that the FCD lesion in one patient was related to the bottom of two sulci (intraparietal and Sylvian). Average depth of the portion of the sulcal bottom line in the neighbourhood of the FCD lesion (see Methods section for details) is also shown. Mean and maximum ( SD) depths for the same sulcus in controls are also shown. Letters in superscript (A^ C) correspond to the three examples shown in Fig 4. Fig. 4 Histologically proven focal cortical dysplasia at the bottom of a sulcus in three patients (A^C). Top row. Three-dimensional surface rendering of the MRI. The FCD sulcus (turquoise) and the lesion (red) are shown in semi-transparent. Sulci related to the FCD are magnified. Bottom rows: T1-weighted MR images (sagittal, coronal and axial) with and without overlaid FCD label. Details about sulcal depth are given in Table 1. no difference in the proportion of patients with ictal activity concordant with the FCD between small and large lesions (48% versus 64%, P = 0.6). There was a tendency to perform more invasive SEEG recordings in patients with small than large FCD lesions (33% versus 5%; P = 0.05). Discussion Clinical observations have prompted the recent addition of the ‘bottom-of-sulcus dysplasia’ as a subtype of FCD with balloon cells to the classification of malformations of cortical development (Barkovich et al., 2005). Here, we present Downloaded from http://brain.oxfordjournals.org/ by guest on September 30, 2016 Central area Central, LB Controls Bottom of a sulcus cortical dysplasia Bottom-of-a-sulcus dysplasia: potential pathophysiological mechanisms During the initial phase of brain development, the surface of the hemispheres is smooth. Most sulci and gyri develop during the third trimester, and the primary and secondary fissures are visible at birth (Chi et al., 1977; Ono et al., 1990 Armstrong et al., 1995). The deepest parts of sulci form early (Welker, 1990) and their appearance is less variable than superficial folds that form later (Lohmann et al., 2008). Importantly, sulcal bottoms are believed to play a pivotal role in the ontogeny of human gyrification (Armstrong et al., 1995; Ochiai et al., 2004; Regis et al., 2005; Lohmann et al., 2008). Typically, the sulci continue to develop after birth (Jernigan et al., 1991) and during early childhood the degree of gyrification stabilizes (Zilles et al., 1988). A variety of non-genetic factors, including intrauterine environment (Bartley et al., 1997; Mohr et al., 2004), modulate hemispheric sulcal morphology, while data from morphological analysis of individual sulci suggest genetic encoding (Le Goualher et al., 2000). Sulcal morphology and patterns are thought to be linked to the underlying cytoarchitecture, and to be an effect of the expansion of cortical grey matter and the development of interconnecting circuits (Armstrong et al., 1995; Van Essen, 1997; Hilgetag and Barbas, 2005). In these models, 3251 gyrification is influenced mainly by three factors: forces generated by growing and migrating cells, local mechanical properties of tissue and extrinsic factors. More specifically, it has been postulated that cortical folding involves forces resulting from different tension of growth between early cortical strata (Caviness, 1975), tension along axons connecting cortical areas (Van Essen, 1997) and changes in subcortical connectivity (Rakic, 1988). The axonal tension hypothesis (Van Essen, 1997) posits that the shape of the brain reflects the viscoelastic tension exerted by fibres. The convoluted cortical landscape is produced by the global competition and resulting minimization of axonal tension. In this scenario, gyri form between strongly connected areas as axons draw them together, and sulci arise between weakly linked or unconnected areas. Recent studies based on the analysis of architectonic and connection data of primate prefrontal cortices (Hilgetag and Barbas, 2005) provide quantitative evidence consistent with a key role of these mechanical factors in the formation of cortical convolutions through axonal tension. The exact mechanisms responsible for the differential vulnerability of the sulcal bottom to host small FCD lesions are unclear. Van Essen’s theory (Van Essen, 1997) postulates that weaker local interconnections may lead to deep sulci due to the predominant traction exerted by the long connections (i.e. thalamo–cortical or cortico–cortical). This is supported by experimental data indicating that early disruption in connectivity can lead to the emergence of anomalous sulcation (Rakic, 1988). Thus, it is plausible that the pattern observed in our patients may in part result from a disturbed neuronal connectivity in areas adjacent or distant to the FCD lesion. Indeed, dysplasias have been shown to be associated with abnormal connectivity (Sisodiya et al., 1995; Sisodiya and Free, 1997; Matsuda et al., 2001; Lee et al., 2004) and decreased white matter organization (Lee et al., 2005a). Cytoarchitectonic differentiation, selective neuronal death and progressive myelination are among the ‘micromechanic’ factors that also influence gyrification (Welker, 1990). Such factors are modified in large part by nongenetic influences (Lohmann et al., 1999). Neuronal density has been shown to be decreased within the FCD lesion compared to the adjacent unaffected cortex, possibly reflecting neuropil expansion, local failure of neuronal migration, proliferation or secondary neuronal loss (Thom et al., 2005). Decreased neuronal density and the presence of abnormal cytoarchitecture, including increased cellular volume and abnormal dendrites (Alonso-Nanclares et al., 2005), are likely to lead to changes in mechanical properties, such as increased elasticity (Lu et al., 2006; Smith et al., 2007) and reduced anisotropy (Van Essen, 1997) of the dysplastic cortex. In a recent morphogenetic computational model, convolutions were found to be deeper in the more elastic part of the cortical surface (Toro and Burnod, 2005). Thus, the co-occurrence of incomplete maturation, decreased neuronal density and abnormal connectivity may Downloaded from http://brain.oxfordjournals.org/ by guest on September 30, 2016 for the first time a systematic evaluation of morphological characteristics of Taylor-type FCD and the sulci harbouring them. Our high-resolution MRI protocol combined with advanced image processing enabled us to perform 3D models of sulcal structure and shape, and the quantification of their spatial characteristics. The results of this analysis indicate that small FCD lesions are often located at the bottom of a sulcus. Furthermore, the morphometric analysis revealed that this sulcus is usually deeper than its equivalent in healthy controls. Accurate review of FCD images examples presented in the literature shows that lesions may be located in the inferior portion of a sulcus (Barkovich et al., 1997; Chan et al., 1998; Bastos et al., 1999; Matsuda et al., 2001; Usui et al., 2001; Urbach et al., 2002; Chassoux, 2003; Colombo et al., 2003; Barkovich et al., 2005; Focke et al., 2008). However, only one report on 24 FCD lesions explicitly indicated that 4/19 (21%) of the rather small dysplasias were located in the depth of a sulcus (Urbach et al., 2002). This low incidence is most likely explained by the use of low-resolution MR images with orthogonal views, potentially leading to incorrect determination of sulcal depth, connective patterns and interruptions. A small lesion that affects the cortex in the depth of a sulcus, however, is not easy to detect, mainly because of the complexity of the brain’s convolution. Mathematical models of sulcal structure and shape can quantify cortical characteristics, and thus give diagnostic information not easily available on visual observation. Brain (2008), 131, 3246 ^3255 3252 Brain (2008), 131, 3246 ^3255 create a local weakness within the developing cortical mantle, particularly in the sulcal bottom. Fragility of the fundi in neurodevelopmental disorders is also suggested by quantitative cytoarchitectonic studies in schizophrenia that have demonstrated reduced cell density in the bottom of temporal lobe sulci (Chance et al., 2004). Moreover, we previously demonstrated an abnormally deep collateral sulcus in epileptic patients with developmental malformations of the hippocampus (Bernasconi et al., 2005). Preferential location of small FCD lesions: relevance to pharmacologically intractable epilepsy removal (Bailey and Von Bonin, 1951; Sanides, 1962). In our patients, sulci harbouring small FCD lesions at their bottom were easily identified macroscopically. In the patient with a small lesion located at the crown of a gyrus, neither careful visual inspection nor the automatic sulcal extraction revealed the presence of microscopic gyration at this location. Partial volume effects due to the relatively low resolution of our 1 mm isotropic images most likely preclude the distinction of microscopic sulci from the surrounding grey matter. High-field MR imaging combined with multiple phased arrays yielding images at submillimetre resolution may resolve such small-scale features and thus allow exploring the subgyral architecture and its relation to developmental lesions. This may be of particular interest in patients with so-called non-lesional epilepsy. In our cohort, active epileptic abnormalities were less frequent in patients with small compared to those with large FCD lesions, although the difference did not reach statistical significance. This finding reflects the well-known intrinsic epileptogenicity of dysplastic human cortex (Avoli et al., 1999). The fact that scalp EEG enables the detection of abnormal electrical activity even in some subtle deep lesions suggests that the epileptogenic network in FCD involves the peri-lesional areas (Raymond and Fish, 1996). A more detailed evaluation of the relationship between FCD location and EEG patterns would entail the exploration of the geometric correspondence between the folded cortical surface and the rectilinear trajectory of depth electrodes, a complex task that needs to be addressed in future prospective studies using sophisticated EEG–MRI co-registration methods and extensive SEEG coverage (Munari, 1985; Munari et al., 1994). Information about lesion localization is important when planning invasive EEG recordings (Sisodiya, 2000). Subdural electrodes are frequently used for evaluating extra-temporal seizures (Jeha et al., 2007; Widdess-Walsh et al., 2007) because they cover a large area of cortex and allow functional mapping (Goldring and Gregorie, 1984; Najm et al., 2002). However, the discrete ictal activity of dysplastic lesions buried in the depth of a sulcus is more likely to be detected when using depth electrodes (Talairach et al., 1992; Chassoux et al., 2000; Privitera et al., 2000; McGonigal et al., 2007), particularly when guided with advanced imaging. Accurate knowledge about the spatial location of subtle FCD lesions may promote minimally invasive approaches (Mikuni and Hashimoto, 2006) and is likely to increase the sensitivity of novel intra-operative tools such as highresolution ultrasound to detect and delineate the surgical target (Miller et al., 2008). Funding Canadian Institutes of Health Research (grant #203707MOP 57840). Downloaded from http://brain.oxfordjournals.org/ by guest on September 30, 2016 Our approach based on a quantitative threshold in a consecutive group of patients allowed us to determine that FCD measuring 53 cm3 represent about 50% of cases, indicating that small dysplastic lesions are indeed prevalent in patients undergoing pre-surgical evaluation for intractable seizures of extra-temporal lobe origin. The identification of FCD on MRI during presurgical evaluation is of crucial clinical importance since the absence of an identifiable lesion is likely to lead to a poor outcome (Sisodiya, 2000). Although high-resolution MRI has made it possible to visually identify malformations of cortical development in an increasing number of patients with neocortical epilepsy (Bernasconi, 2005), the identification of small FCD lesions remains a difficult task, as shown by the low detection rate (19%) on routine clinical imaging reported in this study. Indeed, in recent retrospective series, about 40% of patients with subtle, histopathologically proven Taylor-type FCD had an unrevealing MRI (Matsuda et al., 2001; Hong et al., 2002; Tassi et al., 2002; Colombo et al., 2003; Cossu et al., 2005; Lee et al., 2005b; Jeha et al., 2007; McGonigal et al., 2007; Nobili et al., 2007). Such cases are usually referred to as ‘MRI-negative’. FCDs with balloon cells are not necessarily associated with increased T2-signal (Chan et al., 1998). Moreover, 525% of our patients with small FCD lesions at the bottom of a sulcus had signs of transmantle dysplasia (Barkovich et al., 1997). Therefore, we believe that prior knowledge of preferential location of small FCD at the bottom of a deep sulcus can direct the search for developmental abnormalities, particularly in patients in whom large-scale MRI features are only mildly abnormal or absent. Moreover, this information should be kept in mind when evaluating MRI data of infants, since signal changes related to some Taylor-type FCD lesions may almost completely regress with completion of myelination (Eltze et al., 2005). A detailed histological examination of surgically resected tissue of patients with pharmacoresistant epilepsy has shown that major gyri may be composed of microscopic gyri with corresponding sulci (Kaufmann et al., 1996). These features, imperceptible by gross inspection, have been described in anatomical atlases that paid particular attention to cortical surface characteristics after meningeal P. Besson et al. Bottom of a sulcus cortical dysplasia 3253 cortex in the normal brain and schizophrenia: lamina thickness and cell density. Neurosci Lett 2004; 367: 384–8. Chassoux F. Stereo-EEG: the Sainte-Anne experience in focal cortical dysplasias. Epileptic Disord 2003; 5 (Suppl 2): S95–103. Chassoux F, Devaux B, Landre E, Turak B, Nataf F, Varlet P, et al. Stereoelectroencephalography in focal cortical dysplasia: a 3D approach to delineating the dysplastic cortex. Brain 2000; 123: 1733–51. Chi JG, Dooling EC, Gilles FH. Gyral development of the human brain. Ann Neurol 1977; 1: 86–93. Collins DL, Neelin P, Peters TM, Evans AC. Automatic 3D intersubject registration of MR volumetric data in standardized Talairach space. J Comput Assist Tomogr 1994; 18: 192–205. Colliot O, Antel SB, Naessens VB, Bernasconi N, Bernasconi A. In vivo profiling of focal cortical dysplasia on high-resolution MRI with computational models. Epilepsia 2006a; 47: 134–42. Colliot O, Bernasconi N, Khalili N, Antel SB, Naessens V, Bernasconi N. Individual voxel-based analysis of gray matter in focal cortical dysplasia. Neuroimage 2006; 29: 162–71. Colliot O, Mansi T, Bernasconi N, Naessens V, Klironomos D, Bernasconi A. Segmentation of focal cortical dysplasia lesions on MRI using level set evolution. Neuroimage 2006b; 32: 1621–30. Colombo N, Tassi L, Galli C, Citterio A, Lo Russo G, Scialfa G, et al. Focal cortical dysplasias: MR imaging, histopathologic, and clinical correlations in surgically treated patients with epilepsy. AJNR Am J Neuroradiol 2003; 24: 724–33. Cossu M, Cardinale F, Castana L, Citterio A, Francione S, Tassi L, et al. Stereoelectroencephalography in the presurgical evaluation of focal epilepsy: a retrospective analysis of 215 procedures. Neurosurgery 2005; 57: 706–18; discussion 706–18. Eltze CM, Chong WK, Bhate S, Harding B, Neville BG, Cross JH. Taylor-type focal cortical dysplasia in infants: some MRI lesions almost disappear with maturation of myelination. Epilepsia 2005; 46: 1988–92. Engel J Jr, Van Ness PC, Rasmussen T, Ojemann LM. Outcome with respect to epileptic seizures. In: Engel J Jr, editor. Surgical treatment of the epilepsies. New York: Raven; 1993. p. 609–21. Focke NK, Symms MR, Burdett JL, Duncan JS. Voxel-based analysis of whole brain FLAIR at 3T detects focal cortical dysplasia. Epilepsia 2008; 49: 783–93. Frater JL, Prayson RA, Morris IH, Bingaman WE. Surgical pathologic findings of extratemporal-based intractable epilepsy: a study of 133 consecutive resections. Arch Pathol Lab Med 2000; 124: 545–9. Goldring S, Gregorie EM. Surgical management of epilepsy using epidural recordings to localize the seizure focus. Review of 100 cases. J Neurosurg 1984; 60: 457–66. Hilgetag CC, Barbas H. Developmental mechanics of the primate cerebral cortex. Anat Embryol (Berl) 2005; 210: 411–7. Hong KS, Lee SK, Kim JY, Lee DS, Chung CK. Pre-surgical evaluation and surgical outcome of 41 patients with non-lesional neocortical epilepsy. Seizure 2002; 11: 184–92. Jeha LE, Najm I, Bingaman W, Dinner D, Widdess-Walsh P, Luders H. Surgical outcome and prognostic factors of frontal lobe epilepsy surgery. Brain 2007; 130: 574–84. Jernigan TL, Trauner DA, Hesselink JR, Tallal PA. Maturation of human cerebrum observed in vivo during adolescence. Brain 1991; 114: 2037–49. Kaufmann WE, Krauss GL, Uematsu S, Lesser RP. Treatment of epilepsy with multiple subpial transections: an acute histologic analysis in human subjects [see comments]. Epilepsia 1996; 37: 342–52. Kim H, Bernasconi N, Bernhardt B, Colliot O, Bernasconi A. Basal temporal sulcal morphology in healthy controls and patients with temporal lobe epilepsy. Neurology 2008; 70: 2159–65. Le Goualher G, Argenti AM, Duyme M, Baare WF, Hulshoff Pol HE, Boomsma DI, et al. Statistical sulcal shape comparisons: application to the detection of genetic encoding of the central sulcus shape. Neuroimage 2000; 11: 564–74. Lee SK, Kim DI, Kim J, Kim DJ, Kim HD, Kim DS, et al. Diffusion-tensor MR imaging and fiber tractography: a new method of describing Downloaded from http://brain.oxfordjournals.org/ by guest on September 30, 2016 References Alonso-Nanclares L, Garbelli R, Sola RG, Pastor J, Tassi L, Spreafico R, et al. Microanatomy of the dysplastic neocortex from epileptic patients. Brain 2005; 128: 158–73. Armstrong E, Schleicher A, Omran H, Curtis M, Zilles K. The ontogeny of human gyrification. Cereb Cortex 1995; 5: 56–63. Avoli M, Bernasconi A, Mattia D, Olivier A, Hwa GG. Epileptiform discharges in the human dysplastic neocortex: in vitro physiology and pharmacology. Ann Neurol 1999; 46: 816–26. Bailey P, Von Bonin G. The isocortex of Man. Urbana: University of Illinois Press; 1951. p. 301. Barkovich AJ, Kuzniecky RI. Neuroimaging of focal malformations of cortical development. J Clin Neurophysiol 1996; 13: 481–94. Barkovich AJ, Raybaud CA. Malformations of cortical development. Neuroimaging Clin N Am 2004a; 14: 401–23. Barkovich AJ, Raybaud CA. Neuroimaging in disorders of cortical development. Neuroimaging Clin N Am 2004b; 14: 231–54, viii. Barkovich AJ, Kuzniecky RI, Bollen AW, Grant PE. Focal transmantle dysplasia: a specific malformation of cortical development. Neurology 1997; 49: 1148–52. Barkovich AJ, Kuzniecky RI, Jackson GD, Guerrini R, Dobyns WB. A developmental and genetic classification for malformations of cortical development. Neurology 2005; 65: 1873–87. Bartley AJ, Jones DW, Weinberger DR. Genetic variability of human brain size and cortical gyral patterns. Brain 1997; 120: 257–69. Bastos AC, Comeau RM, Andermann F, Melanson D, Cendes F, Dubeau F, et al. Diagnosis of subtle focal dysplastic lesions: curvilinear reformatting from three-dimensional magnetic resonance imaging. Ann Neurol 1999; 46: 88–94. Bernasconi A. Quantitative MR imaging of the neocortex. Neuroimaging Clin N Am 2004; 14: 425–36. Bernasconi A. Magnetic resonance imaging in intractable epilepsy: Focus on structural image analysis. In: Blume W, Carlen PL, Wiebe S, Young AB, Padjen A, Starrveld E, editors. Advances in neurology. Philadelphia, Pennsylvania: Lippincott-Williams; 2005. p. 273–8. Bernasconi A, Antel SB, Collins DL, Bernasconi N, Olivier A, Dubeau F, et al. Texture analysis and morphological processing of magnetic resonance imaging assist detection of focal cortical dysplasia in extratemporal partial epilepsy. Ann Neurol 2001; 49: 770–5. Bernasconi N, Kinay D, Andermann F, Antel S, Bernasconi A. Analysis of shape and positioning of the hippocampal formation: an MRI study in patients with partial epilepsy and healthy controls. Brain 2005; 128: 2442–52. Borgefors G. On digital distance transforms in three dimensions. computer vision and image understanding. Comput Vis Image Underst 1996; 64: 368–76. Bronen RA, Spencer DD, Fulbright RK. Cerebrospinal fluid cleft with cortical dimple: MR imaging marker for focal cortical dysgenesis. Radiology 2000; 214: 657–63. Bronen RA, Vives KP, Kim JH, Fulbright RK, Spencer SS, Spencer DD. Focal cortical dysplasia of Taylor, balloon cell subtype: MR differentiation from low-grade tumors. AJNR Am J Neuroradiol 1997; 18: 1141–51. Cachier P, Mangin J-F, Pennec X, Rivière D, Papadopoulos-Orfanos D, Régis J, et al. Multisubject non-rigid registration of brain MRI using intensity and geometric features. In: Proceedings of Medical Image Computing and Computer-Assisted Intervention – MICCAI 2001, 4th International Conference Utrecht, The Netherlands, October 14–17 London, UK: Springer Verlag; 2001. p. 734. Caviness VS Jr. Mechanical model of brain convolutional development. Science 1975; 189: 18–21. Chan S, Chin SS, Nordli DR, Goodman RR, DeLaPaz RL, Pedley TA. Prospective magnetic resonance imaging identification of focal cortical dysplasia, including the non-balloon cell subtype. Ann Neurol 1998; 44: 749–57. Chance SA, Tzotzoli PM, Vitelli A, Esiri MM, Crow TJ. The cytoarchitecture of sulcal folding in Heschl’s sulcus and the temporal Brain (2008), 131, 3246 ^3255 3254 Brain (2008), 131, 3246 ^3255 Privitera MD, Yeh HS, Blisard K, Sanchez N. Detection of epileptogenic focal cortical dysplasia by depth, not subdural electrodes. Neurosurg Rev 2000; 23: 49–51. Rakic P. Specification of cerebral cortical areas. Science 1988; 241: 170–6. Raymond AA, Fish DR. EEG features of focal malformations of cortical development. J Clin Neurophysiol 1996; 13: 495–506. Raymond AA, Fish DR, Sisodiya SM, Alsanjari N, Stevens JM, Shorvon SD. Abnormalities of gyration, heterotopias, tuberous sclerosis, focal cortical dysplasia, microdysgenesis, dysembryoplastic neuroepithelial tumor and dysgenesis of archicortex in epilepsy. Clinical, EEG and neuroimaging features in 100 adult patients. Brain 1995; 118: 629–60. Regis J, Mangin JF, Ochiai T, Frouin V, Riviere D, Cachia A, et al. "Sulcal root" generic model: a hypothesis to overcome the variability of the human cortex folding patterns. Neurol Med Chir (Tokyo) 2005; 45: 1–17. Rivière D, Mangin J-F, Papadopoulos-Orfanos D, Martinez J-M, Frouin V, Régis J. Automatic recognition of cortical sulci using a congregation of neural networks. In: Proceedings of Medical Image Computing and Computer-assisted Intervention – MICCAI. London, UK: Springer Verlag; 2000. pp. 40–9. Rivière D, Mangin JF, Papadopoulos-Orfanos D, Martinez JM, Frouin V, Régis J. Automatic recognition of cortical sulci of the human brain using a congregation of neural networks. Med Image Anal 2002; 6: 77–92. Sanides F. Die Architektonik des Menschlichen Stirnhirns. Berlin: Springer; 1962. Sisodiya SM. Surgery for malformations of cortical development causing epilepsy. Brain 2000; 123: 1075–91. Sisodiya SM. Surgery for focal cortical dysplasia. Brain 2004; 127: 2383–4. Sisodiya SM, Free SL. Disproportion of cerebral surface areas and volumes in cerebral dysgenesis. MRI-based evidence for connectional abnormalities. Brain 1997; 120: 271–81. Sisodiya SM, Free S, Stevens JM, Fish DR, Shorvon SD. Widespread cerebral structural changes in patients with cortical dysgenesis and epilepsy. Brain 1995; 118: 1039–50. Sled JG, Zijdenbos AP, Evans AC. A nonparametric method for automatic correction of intensity nonuniformity in MRI data. IEEE Trans Med Imaging 1998; 17: 87–97. Smith BA, Roy H, De Koninck P, Grutter P, De Koninck Y. Dendritic spine viscoelasticity and soft-glassy nature: balancing dynamic remodeling with structural stability. Biophys J 2007; 92: 1419–30. Smith SM. Fast robust automated brain extraction. Hum Brain Mapp 2002; 17: 143–55. Talairach J, Tournoux P. Co-planar stereotaxic atlas of the human brain. New York: Thieme Medical; 1988. Talairach J, Tournoux P, Musolino A, Missir O. Stereotaxic exploration in frontal epilepsy. Adv Neurol 1992; 57: 651–88. Tassi L, Colombo N, Garbelli R, Francione S, Lo RG, Mai R, et al. Focal cortical dysplasia: neuropathological subtypes, EEG, neuroimaging and surgical outcome. Brain 2002; 125: 1719–32. Taylor DC, Falconer MA, Bruton CJ, Corsellis JAN. Focal dysplasia of the cerebral cortex in epilepsy. J Neurol Neurosurg Psychiatry 1971; 34: 369–87. Thom M, Martinian L, Sen A, Cross J, Harding B, Sisodiya S. Cortical neuronal densities and lamination in focal cortical dysplasia. Acta Neuropathologica 2005; 110: 383–92. Toro R, Burnod Y. A morphogenetic model for the development of cortical convolutions. Cereb Cortex 2005; 15: 1900–13. Urbach H, Scheffler B, Heinrichsmeier T, von Oertzen J, Kral T, Wellmer J, et al. Focal cortical dysplasia of Taylor’s balloon cell type: a clinicopathological entity with characteristic neuroimaging and histopathological features, and favorable postsurgical outcome. Epilepsia 2002; 43: 33–40. Usui N, Matsuda K, Mihara T, Tottori T, Ohtsubo T, Baba K, et al. MRI of cortical dysplasia–correlation with pathological findings. Neuroradiology 2001; 43: 830–7. Van Essen DC. A tension-based theory of morphogenesis and compact wiring in the central nervous system. Nature 1997; 385: 313–8. Downloaded from http://brain.oxfordjournals.org/ by guest on September 30, 2016 aberrant fiber connections in developmental CNS anomalies. Radiographics 2005a; 25: 53–65; discussion 66–8. Lee SK, Kim DI, Mori S, Kim J, Kim HD, Heo K, et al. Diffusion tensor MRI visualizes decreased subcortical fiber connectivity in focal cortical dysplasia. Neuroimage 2004; 22: 1826–9. Lee SK, Lee SY, Kim KK, Hong KS, Lee DS, Chung CK. Surgical outcome and prognostic factors of cryptogenic neocortical epilepsy. Ann Neurol 2005b; 58: 525–32. Lohmann G, von Cramon DY, Colchester AC. Deep sulcal landmarks provide an organizing framework for human cortical folding. Cereb Cortex 2008; 18: 1415–20. Lohmann G, von Cramon DY, Steinmetz H. Sulcal variability of twins. Cereb Cortex 1999; 9: 754–63. Lu YB, Franze K, Seifert G, Steinhauser C, Kirchhoff F, Wolburg H, et al. Viscoelastic properties of individual glial cells and neurons in the CNS. Proc Natl Acad Sci USA 2006; 103: 17759–64. Mahvash M, Konig R, Urbach H, von Ortzen J, Meyer B, Schramm J, et al. FLAIR-/T1-/T2-co-registration for image-guided diagnostic and resective epilepsy surgery. Neurosurgery 2006; 58: ONS69–75; discussion ONS69–75. Mangin JF, Frouin V, Bloch I, Regis J, Lopez-Krahe J. From 3D magnetic resonance images to structural representations of the cortex topography using topology preserving deformations. J Math Imag and Vision 1995; 5: 297–318. Matsuda K, Mihara T, Tottori T, Otubo T, Usui N, Baba K, et al. Neuroradiologic findings in focal cortical dysplasia: histologic correlation with surgically resected specimens. Epilepsia 2001; 42 (Suppl 6): 29–36. Mazziotta JC, Toga AW, Evans A, Fox P, Lancaster J. A probabilistic atlas of the human brain: theory and rationale for its development. The International Consortium for Brain Mapping (ICBM). Neuroimage. 1995; 2: 89–101. McGonigal A, Bartolomei F, Regis J, Guye M, Gavaret M, Trebuchon-Da Fonseca A, et al. Stereoelectroencephalography in presurgical assessment of MRI-negative epilepsy. Brain 2007; 130: 3169–83. Mikuni N, Hashimoto N. A minimally invasive transsulcal approach to the paracentral inner lesion. Minim Invasive Neurosurg 2006; 49: 291–5. Miller D, Knake S, Bauer S, Krakow K, Pagenstecher A, Sure U, et al. Intraoperative ultrasound to define focal cortical dysplasia in epilepsy surgery. Epilepsia 2008; 49: 156–8. Mohr A, Weisbrod M, Schellinger P, Knauth M. The similarity of brain morphology in healthy monozygotic twins. Brain Res Cogn Brain Res 2004; 20: 106–10. Munari C. The role of stereo-EEG in the evaluation of partial epileptic seizures. In: Morselli PL, Porter RJ, editors. The epilepsies. London: Butterworths; 1985. p. 267–306. Munari C, Hoffmann D, Francione S, Kahane P, Tassi L, Lo Russo G, et al. Stereo-electroencephalography methodology: advantages and limits. Acta Neurol Scand Suppl 1994; 152: 56–67, discussion 68–9. Najm IM, Bingaman WE, Luders HO. The use of subdural grids in the management of focal malformations due to abnormal cortical development. Neurosurg Clin N Am 2002; 13: 87–92, viii–ix. Nobili L, Francione S, Mai R, Cardinale F, Castana L, Tassi L, et al. Surgical treatment of drug-resistant nocturnal frontal lobe epilepsy. Brain 2007; 130: 561–73. Ochiai T, Grimault S, Scavarda D, Roch G, Hori T, Riviere D, et al. Sulcal pattern and morphology of the superior temporal sulcus. Neuroimage 2004; 22: 706–19. Ono M, Kubik S, Abernethey CD. Atlas of the cerebral sulci. Stuttgart: Georg Thieme; 1990. Palmini A, Najm I, Avanzini G, Babb T, Guerrini R, Foldvary-Schaefer N, et al. Terminology and classification of the cortical dysplasias. Neurology 2004; 62: S2–8. Prayson RA, Spreafico R, Vinters HV. Pathologic characteristics of the cortical dysplasias. Neurosurg Clin N Am 2002; 13: 17–25. P. Besson et al. Bottom of a sulcus cortical dysplasia Welker W. Why does cerebral cortex fissure and fold? A review of determinants of gyri and sulci. In: Jones EG, Peters A, editors. Cerebral cortex. New York: Plenum Press; 1990. p. 3–136. Widdess-Walsh P, Jeha L, Nair D, Kotagal P, Bingaman W, Najm I. Subdural electrode analysis in focal cortical dysplasia: Predictors of surgical outcome. Neurology 2007; 69: 660–7. Brain (2008), 131, 3246 ^3255 3255 Yagishita A, Arai N, Maehara T, Shimizu H, Tokumaru AM, Oda M. Focal cortical dysplasia: appearance on MR images. Radiology 1997; 203: 553–9. Zilles K, Armstrong E, Schleicher A, Kretschmann HJ. The human pattern of gyrification in the cerebral cortex. Anat Embryol (Berl) 1988; 179: 173–9. Downloaded from http://brain.oxfordjournals.org/ by guest on September 30, 2016