Lesson 2 - Fill in the blanks notes
advertisement

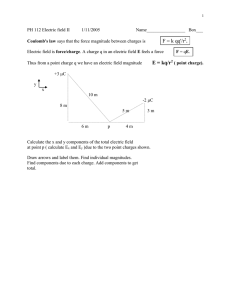
Lesson 6: Coulomb’s Law Part 1: Introduction to Coulomb’s Law Electrostatic Forces and the Law of Charges: Coulomb’s Law allows us to calculate the magnitude of force between two charges: Charles-Augustin de Coulomb Born on June 14, 1736, in Angoulême, France, Charles-Augustin de Coulomb studied engineering and plied his trade with the military before winning accolades for his work in torsion balances. He offered pioneering theories in the force found between electrical charges, as well as magnetic attraction and repulsion. The unit of measurement known as the coulomb is named in his honor. He died in Paris on August 23, 1806. Note: Coulomb’s law gives you the magnitude of the force only. It is up to you to use the law of charges to determine the direction. Example: What is the force on the +3.0 µC charge? -2.0 µC 0.15 m +3.0 µC Part 2: Coulomb’s Law and Graphing There are three common graph shapes that you will encounter in Physics 30 Linear Parabolic Inverse Square Coulomb’s law belongs to a family of formulas called inverse square laws Universal Gravitation Coulomb’s Law In this lesson, we will use curve straightening techniques to determine Coulomb’s constant using graphical data. Example: The electrostatic force on a 10 µC charged pith ball is measured as a 10 µC charged rod is brought near. Curve straightening: turn the inverse square graph into a linear grap Part 3: Coulomb’s Law and Vector Addition If there are more than two charges in a problem, the electrostatic forces combine to produce a net force. Since Force is a vector, vector addition techniques must be used to combine the forces. Example: Calculate the force on charge 3 2 1 1.5x10-7 C -2.3x10-7 C 1.4 m Example: What is the force on charge 2? 1 - 7.0 C 4.0 m 2 -2.0 C 3 12 m 3.4 C 3 1.7m -3.5x10-7 C