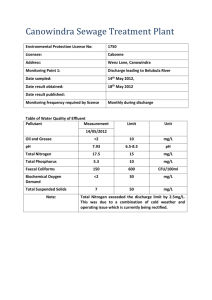
Calculation of Constant Power Lithium Battery Discharge Curves
advertisement

batteries
Communication
Calculation of Constant Power Lithium Battery
Discharge Curves
Lance W. Traub
Aerospace and Mechanical Engineering Department, Embry Riddle Aeronautical University, Prescott, AZ 86314,
USA; traubl@erau.edu; Tel.: +1-928-777-6683
Academic Editor: Andreas Jossen
Received: 22 February 2016; Accepted: 7 June 2016; Published: 11 June 2016
Abstract: Standard battery testing procedure consists of discharging the battery at constant current.
However, for battery powered aircraft application, consideration of the cruise portion of the flight
envelope suggests that power should be kept constant, implying that battery characterization should
occur over a constant power discharge. Consequently, to take advantage of existing battery discharge
curves it would be useful to have a methodology that can extract a constant power discharge curve
from a constant current discharge curve. The development of such a methodology for lithium
batteries is described in this article.
Keywords: battery discharge curve; lithium battery; constant power discharge curve; battery
powered aircraft
1. Introduction
Battery powered unmanned aerial vehicles have experienced consistent growth in use and
application. Like any other class of flight vehicle, accurate estimation of range and endurance is
paramount during the design and selection phase. Methods to estimate range and endurance for
battery powered aircraft [1–4] are not as well established as those for fossil fuel powered craft [5–7].
In [8], a method is developed to estimate the range and endurance of battery powered aircraft with
validation of the approach presented in [9]. To implement the method and approach of [8,9], battery
discharge curves are required at constant power, where the battery voltage and current vary. This is
atypical from the usual method of battery performance characterization, where the current is fixed and
power and voltage are variable. Consequently, to use the method in [8], battery data is required for
a constant power discharge, or a means is required to convert constant current discharge curves to
constant power discharge curves. Such a technique is the focus of this article.
2. Theoretical Development
Working with lead acid batteries, Peukert [10,11] established that their rate of discharge affected
their effective capacity; for discharge rates greater than a 1C, the capacity would reduce (and vice versa).
An increase in the discharge current of the battery may decrease the effective capacity due to a decline
of the reactivity of the battery’s active materials. Mathematically, this is expressed as:
K “ iP t
(1)
where P is the Peukert constant, i is current and K is a constant. For lithium batteries, which are
at present the “standard” due to their high power density, the impact of the Peukert effect is not
well established [12]. A value of P « 1.05 has been suggested [12]. The greatest impact on lithium
battery capacity stems from their sensitivity to temperature; low temperatures severely attenuate the
rated battery capacity C [13,14]. Lithium batteries are also sensitive to the number of charging and
Batteries 2016, 2, 17; doi:10.3390/batteries2020017
www.mdpi.com/journal/batteries
Batteries 2016, 2, 17
2 of 7
Batteries 2016,
2, 17 the greater the number of cycles the less the capacity due to a loss of active2material
of 7
discharging
cycles;
Batteries 2016, 2, 17
2 of 7
Batteries 2016, 2, 17
2 of 7
within
the cell and primarily loss of lithium inventory [15].
discharging cycles; the greater the number of cycles the less the capacity due to a loss of active
In
order to cycles;
transform
a constant
currentofdischarge
Figures
for of
examples
discharging
the greater
greater
the number
number
cycles the
the curve,
less the
thesee
capacity
due1a–3a
to a loss
active of
discharging
cycles;
the
the
of cycles
less
material
within
the cell
and primarily
loss of lithium
inventory
[15].capacity due to a loss of active
material
within
the
cell
and
primarily
loss
of
lithium
inventory
[15].
typical
discharge
profiles
[16,17],
it
would
be
useful
to
correlate
the
curves
by
establishing
material
within
cell and aprimarily
of lithium
inventory
In order
tothe
transform
constantloss
current
discharge
curve,[15].
see Figures 1a–3a for examples of their
In
order
to
transform
a
constant
current
discharge
curve,
seewas
Figures
1a–3a from
for examples
examples
of of
dependence
on
discharge
rate
or
current.
The
data
in
Figures
1–3
obtained
digitization
In discharge
order to transform
a constant
current
curve, see
Figures
1a–3a
for
of
typical
profiles [16,17],
it would
be discharge
useful to correlate
the
curves
by establishing
their
typical
discharge
profiles
[16,17],
it
would
be
useful
to
correlate
the
curves
by
establishing
their
the manufacturer’s
charts.
Higher
currents
to reduce
the voltage
during discharge
typical discharge
profiles
[16,17],
it would
bedata
useful
to seen
correlate
the obtained
curves
by
establishing
their
dependence
on discharge
rate
ordischarge
current.
The
inare
Figures
1–3
was
from
digitization
of
dependence on
on discharge
discharge rate
rate or
or current.
The data
data in
in Figures
Figures 1–3
1–3 was
was obtained
obtained from
from digitization
digitization of
of
dependence
The
because
the polarization
voltage
in current.
the electrode
reactions
[14].
It was
found
heuristically
the
manufacturer’s
charts.
Higher
discharge
currents
are seenincreases
to reduce
the voltage
during
discharge
the
manufacturer’s
charts.
Higher
discharge
currents
are
seen
to
reduce
the
voltage
during
discharge
thethe
manufacturer’s
charts.voltage
Higher
currents
are seen
to reduce
theItvoltage
during
discharge
because
the polarization
theconstant
electrode
reactions
increases
[14].
wasa found
heuristically
within
spirit
of Equation
(1)
thatindischarge
the
current
discharge
curves
for
given
battery
collapse
because the
the polarization
polarization voltage
voltage in
in the electrode
electrode reactions
reactions increases
increases [14].
[14]. It
It was
was found
found heuristically
heuristically
because
within
the
spirit
of
Equation
(1)
that
the
constant
current
discharge
curves
for
a
given
battery
collapse
whenwithin
the voltage
V
during
the
discharge
is
multiplied
by
the
current
raised
to
the
power
n
for
a given
the spirit
spirit of Equation
Equation (1)
(1) that
that the
the constant
constant current
current discharge
discharge curves
curves for
for aa given
given battery
battery collapse
collapse
withinthe
the
when
voltageof
V during
thecollapse
discharge
isseen
multiplied
bypower
the current
raised
to
the
powerapplication,
n for a given high
discharged
capacity
(while
this
is
for
high
cells
used
in
aircraft
when the
the voltage
voltage V
V during the
the discharge is
is multiplied
multiplied by
by the
the current
current raised
raised to the
the power
power nn for
for aa given
given
when
discharged
capacityduring
(while thisdischarge
collapse is seen
for high power
cells used into
aircraft
application,
high
energy
cells maycapacity
not show
such
correlation):
discharged
(while
thisa collapse
is seen for high power cells used in aircraft application, high
discharged
this collapse
is seen for high power cells used in aircraft application, high
energy
cellscapacity
may not(while
show such
a correlation):
energy cells
cells may
may not
not show
show such
such aa correlation):
correlation):
energy
i nnV ( D )“= ConstantpDq
Cons tan t ( D )
in VpDq
(2)
i V ( D ) = Cons tan t ( D )
(2)
i nV ( D ) = Cons tan t ( D )
(2)
5
5
4.55
4.5
4.54
4
3.54
5
5
4.55
4.5
4.54
3.5
3.53
n n Vin
ViVi
Voltage,
Voltage,
VV V
Voltage,
4
3.54
3.5
3.53
3
2.53
2.5A Discharge
2.5
2.52
2
1.52
1.5
1.51
1 0
1 0
0
500
2.5A
Discharge
5A
Discharge
2.5A Discharge
5A Discharge
10A
Discharge
5A Discharge
10ADischarge
Discharge
15A
10A Discharge
15ADischarge
Discharge
20A
15A Discharge
20A Discharge
20A Discharge
2.5
2.52
2
1.52
1.5
1.51
1000
500
500
2.5A Discharge
3
2.53
2.5A
Discharge
5A
Discharge
2.5A Discharge
5A Discharge
10A
Discharge
5A Discharge
10ADischarge
Discharge
15A
10A Discharge
15ADischarge
Discharge
20A
15A Discharge
20A Discharge
20A Discharge
1500
Discharged
mAh
1000 Capacity,
1500
1000
1500
Discharged Capacity, mAh
Discharged Capacity, mAh
2000
2500
2000
2000
2500
2500
1 0
1 0
0
500
500
500
1000
1500
2000
2500
1500
2000
2000
2500
2500
1500
2000
2500
2000
2000
2500
2500
Discharged
mAh
1000 Capacity,
1500
1000
Discharged Capacity, mAh
(a)
(b)Capacity, mAh
Discharged
(a)
(b)
(a)
(b)
Figure 1. INR 18650 [16]: (a) constant current and (b) correlated discharge curves. n = 0.05, and C =
Figure
1. INR
18650
(a)constant
constant
current
(b) correlated
discharge
Figure
1. INR
18650[16]:
[16]: (a)
current
and and
(b) correlated
discharge
curves. n curves.
= 0.05, andn C== 0.05,
Figure
1. INR 18650 [16]: (a) constant current and (b) correlated discharge curves. n = 0.05, and C =
2500
mAh.
and C2500
= 2500
mAh.
mAh.
2500 mAh.
5
5
5
4.5
4.5
4.5
4
4.5
4.5
4
4
4
3.5
4
4
3.5
3.5
3.5
3
3.5
3.5
3
n n Vin
ViVi
Voltage,
Voltage,
VV V
Voltage,
5
5
5
4.5
3
3
2.5
3
3
2.5
2.5
2.5
2
2.5
2.5
2
2.5A Discharge
10A
2.5ADischarge
Discharge
2.5A Discharge
20A
10ADischarge
Discharge
10A Discharge
20A Discharge
20A Discharge
2
2
1.5
1.5
1.5
1
1 0
1 0
0
500
1000
500
500
1500
Discharged Capacity, mAh
Discharged Capacity, mAh
(a)
(a)
(a)
10A
2.5ADischarge
Discharge
2.5A Discharge
20A
10ADischarge
Discharge
10A Discharge
20A Discharge
20A Discharge
1.5
1.5
1
1500
Discharged
mAh
1000 Capacity,
1500
1000
2.5A Discharge
2
2
1.5
2000
2500
2000
2000
2500
2500
1 0
1 0
0
500
500
500
1000
Discharged
mAh
1000 Capacity,
1500
1000
1500
Discharged Capacity, mAh
Discharged Capacity, mAh
(b)
(b)
(b)
Figure 2. LG18B50HE2 [16]: (a) constant current and (b) correlated discharge curves. n =
Figure 2.
2. LG18B50HE2
LG18B50HE2 [16]:
[16]: (a) constant
constant current
current and
and (b) correlated
correlated discharge
discharge curves.
curves. n =
Figure
0.05,
C = 2500 mAh.
Figure
2. and
LG18B50HE2
[16]: (a) (a)
constant current
and (b)(b)
correlated discharge
curves. nn == 0.05,
0.05,
and
C
=
2500
mAh.
andmAh.
C = 2500 mAh.
and C0.05,
= 2500
5
5
4.5
4.5
4.5
4
4.5
4.5
4
4
4
3.5
4
4
3.5
3.5
3.5
3
n n Vin
ViVi
5
5
5
4.5
Voltage,
Voltage,
VV V
Voltage,
5
3
3
2.5
2.5
2.5
2
2 0
2 0
0
25ADischarge
Discharge
50A
25A Discharge
50ADischarge
Discharge
75A
50A Discharge
75A Discharge
100A
Discharge
75A Discharge
100A Discharge
100A Discharge
1000
2000
1000
1000
25ADischarge
Discharge
50A
25A Discharge
50ADischarge
Discharge
75A
50A Discharge
75A Discharge
100A
Discharge
75A Discharge
100A Discharge
100A Discharge
3
3
2.5
2.5
2.5
2
3000
Discharged
mAh
2000 Capacity,
3000
2000
25A Discharge
3.5
3.5
3
25A Discharge
3000
Discharged Capacity, mAh
Discharged Capacity, mAh
4000
5000
4000
4000
5000
5000
2 0
2 0
0
1000
1000
1000
2000
3000
Discharged
mAh
2000 Capacity,
3000
2000
3000
Discharged Capacity, mAh
Discharged Capacity, mAh
4000
5000
4000
4000
5000
5000
(a)
(b)
(a)
(b)
(a)
(b)
Figure 3. Thunderpower Extreme [17]: (a) constant current and (b) correlated discharge curves. n =
Figure 3. Thunderpower Extreme [17]: (a) constant current and (b) correlated discharge curves. n =
Figure
3. C
Thunderpower
0.05,
and
= 5000 mAh. Extreme [17]: (a) constant current and (b) correlated discharge curves. n =
Figure
3. and
Thunderpower
0.05,
C = 5000 mAh. Extreme [17]: (a) constant current and (b) correlated discharge curves.
0.05, and C = 5000 mAh.
n = 0.05, and C = 5000 mAh.
(2)
Batteries 2016, 2, 17
3 of 7
The discharged capacity, D is the total charge drawn from the battery at the time instant in
which it is considered. This correlation is shown in Figures 1b–3b where data is presented for lithium
batteries from various manufacturers. It was found that values of n « 0.05 (determined using a
non-linear least squares minimization) showed the best collapse of the curves. As the discharge curves
can be correlated, they may then be used to estimate a constant power discharge via the following
procedural steps:
(1)
Using the available constant current discharge curves for the battery of interest, plot in V as a
function of the discharged capacity, D, see Figures 1b–3b for examples. Adjust n to afford the
best collapse of the curves. Alternatively, n may be established using a non-linear least square
minimization as follows. Let M = the number of test cases (discharges at different i for a given
battery). The least square criterion is implemented as:
Sum “
M“
ÿ
Vj i j n ´ Vin
‰2
(3)
j “1
(2)
where the overbar indicates an average for all j (for a discharge D). The counter j refers to the
individual data sets. A non-linear least square solver (found in MS Excel, or most mathematical
computational software) may be used to find the value of n that minimizes the summation.
Once n has been determined, the collapsed plots are curve fitted as a function of D. A function of
the form:
O
´
¯ ´
¯
Vin pDq “ a ` cD ` eD2
1 ` bD ` dD2 ` f D3
(4)
matches a battery discharge curve profile well and can be readily found using software such
as TableCurve, MathCad or most statistical curve fitting packages. Naturally, other curve fit
functions may also be used. Note that the non-linear least square minimization solver may be
used directly to solve Equations (3) and (4) with the form of the curve fit expressed by Equation (4)
prescribed. However, ill-conditioning and divergence may be an issue unless the initial estimates
of the coefficients in Equation (4) are close to the optimal values.
(3)
The voltage during the discharge may be found using Vj “
ij “
Pe
Vj
Vj “
in VpDq j´1
¸1{p1´nq
Pe n
(5)
note that for the calculations to not be circular, Vj cannot be a function of in V(D)j . It is thus
assumed that in V(D)j = in V(D)j´ 1 for each successive step, not for successive steps. What this
implies is that in V(D) does not change rapidly between each iteration. As long as the time step
is small relative to the total discharge time this is a reasonable assumption. If the required Preq
or mechanical power is known, then Pe = Preq /ηtot where ηtot is the efficiency of the propulsion
system including the propeller, electronic speed controller and motor.
The corresponding current at this time step is:
ij “
(5)
which after substitution of
yields:
˜
(4)
in V p Dq j´1
ij n
Pe
Vj
(6)
The discharged capacity then follows as:
D j “ i j ˆ p∆tq ` D j´1
(7)
Batteries 2016, 2, 17
4 of 7
Batteries 2016, 2, 17
4 of 7
(6)
(7)
Calculate
the voltage,
current
and discharged
capacity
at the at
next
t (t(h)
(6)
Calculate
the voltage,
current
and discharged
capacity
thetime
nextstep
time
step=t j/N
(t(h)where
= j/N
j = 1,where
2, 3, ... jand
N
is
an
arbitrary
constant
defining
the
time
step
increment.
In
this
article
180
= 1, 2, 3, ... and N is an arbitrary constant defining the time step increment.NIn= this
giving
timeNincrements
∆ttime
of 20increments
s). The time
small
the compared
discharge
article
= 180 giving
Δt step
of 20should
s). The be
time
stepcompared
should be to
small
duration
to
ensure
the
validity
of
Equation
(5).
to the discharge duration to ensure the validity of Equation (5).
Repeat
StepsSteps
3–5 until
the discharged
capacity
Dj is D
approximately
equalequal
to C. to C.
(7)
Repeat
3–5 until
the discharged
capacity
j is approximately
˝C
Note
Note that
that the
the discharge
discharge curves
curves in
in Figures
Figures 1–3
1–3 were
were determined
determined at
at temperatures
temperatures between
between 20
20 °C
˝
and
C. The
The validity
validity of
of Equation
Equation (2)
(2) at
at very
very low
and 25
25 °C.
low temperatures
temperatures needs
needs to
to be
be established.
established.
Consequently,
Figure
4
presents
data
for
a
lithium
polymer
battery
developed
at the Jet Propulsion
Consequently, Figure 4 presents data for a lithium polymer battery developed
at the Jet
˝ C at various
Laboratory
(JPL)
for
application
at
low
temperatures
[18].
The
cell
was
tested
at
´20
Propulsion Laboratory (JPL) for application at low temperatures [18]. The cell was tested at −20 °C at
discharge
rates. Asrates.
shown
Figure
a rate
on capacity
is clearly
evidentevident
with higher
various discharge
Asin
shown
in4a,
Figure
4a,dependence
a rate dependence
on capacity
is clearly
with
discharge
rates
reducing
the
effective
capacity.
Correlation
of
the
data
sets
using
Equation
(2)(2)
is
higher discharge rates reducing the effective capacity. Correlation of the data sets using Equation
presented
inin
Figure
4b.
is presented
Figure
4b.As
Asseen,
seen,Equation
Equation(2)
(2)only
onlyaffords
affordscorrelation
correlationof
ofthe
thethree
three curves
curves during
during the
the
initial
segment
of
the
discharge.
It
may
thus
be
inferred
that
Equation
(2)
is
not
representative
at very
very
initial segment of the discharge. It may thus be inferred that Equation (2) is not representative at
low
low temperatures.
temperatures.
5
5
25mA Discharge
4.5
50mA Discharge
100mA Discharge
4
100mA Discharge
Vin
Voltage, V
4
25mA Discharge
4.5
50mA Discharge
3.5
3.5
3
3
2.5
2.5
2
2
0
0.1
0.2
0.3
Discharged Capacity, Ah
(a)
0.4
0.5
0
0.1
0.2
0.3
0.4
0.5
Discharged Capacity, Ah
(b)
Figure 4.
4. Jet
Laboratory (JPL)/WGL
(JPL)/WGL [18]
[18] lithium-ion
lithium-ion cell
cell discharged
dischargedat
at´20
−20 °C:
˝ C: (a)
Figure
Jet Propulsion
Propulsion Laboratory
(a) constant
constant
current
and
(b)
correlated
discharge
curves.
n
=
0.05,
and
C
=
500
mAh.
current and (b) correlated discharge curves. n = 0.05, and C = 500 mAh.
3. Materials and Methods
3. Materials and Methods
To validate the procedure enumerated above, a test was undertaken using an Electrifly 1300 mAh
To validate the procedure enumerated above, a test was undertaken using an Electrifly 1300 mAh
30C lithium polymer battery (which consists of three cells in series and is rated at 11.1 V with a capacity
30C lithium polymer battery (which consists of three cells in series and is rated at 11.1 V with a
of 1300 mAh, Great Planes, Champaign, IL, USA). The battery was discharged at different constant
capacity of 1300 mAh, Great Planes, Champaign, IL, USA). The battery was discharged at different
current rates (1.45 A, 4.8 A and 7.5 A), while the voltage, power and current were monitored using a
constant current rates (1.45 A, 4.8 A and 7.5 A), while the voltage, power and current were monitored
Watt's Up Watt Meter and Power Analyzer (Denver, CO, USA). The ambient temperature during
using a Watt's Up Watt Meter and Power Analyzer (Denver, CO, USA). The ambient temperature
testing was maintained at 20 °C. Measurements from this Watt meter and power analyzer were
during testing was maintained at 20 ˝ C. Measurements from this Watt meter and power analyzer
compared against a Fluke 175 multi-meter (Everett, WA, USA). The difference between readings was
were compared against a Fluke 175 multi-meter (Everett, WA, USA). The difference between readings
within 0.3% for voltage and current (up to 10 A—limited by the range of the Fluke meter). After each
was within 0.3% for voltage and current (up to 10 A—limited by the range of the Fluke meter). After
test, the battery was allowed to cool for at least 2 h so that it was at ambient temperature before
each test, the battery was allowed to cool for at least 2 h so that it was at ambient temperature before
recharging. A Great Planes AC/DC Triton EQ Charger Balancer was used to recharge the battery.
recharging. A Great Planes AC/DC Triton EQ Charger Balancer was used to recharge the battery.
During discharge either power or current was held constant by monitoring the output of the Watt’s
During discharge either power or current was held constant by monitoring the output of the Watt’s Up
Up meter and manually adjusting the batteries power or current output. This method was viable
meter and manually adjusting the batteries power or current output. This method was viable without
without implementing a control system as changes in power or current were slow. Using this
implementing a control system as changes in power or current were slow. Using this approach, current
approach, current could be maintained within 0.06 A and power within 0.24 W. Consequently, the
could be maintained within 0.06 A and power within 0.24 W. Consequently, the worst case scenario for
worst case scenario for the presented data gives an error in the current of 2.4% and power of 0.7%
the presented data gives an error in the current of 2.4% and power of 0.7% respectively.
respectively.
4. Discussion
4. Discussion
The resulting discharge curves are shown in Figure 5a. For reference, also included in this plot is
The resulting
discharge
curves
shown
in Figure
5a.where
For reference,
also included
in this
plot
an experimental
constant
power
(Pe =are
34 W)
discharge
curve
both the voltage
and current
varied.
is an experimental constant power (Pe = 34 W) discharge curve where both the voltage and current
varied. Correlation (application of Step 1) of the constant current discharge curves in Figure 5a yields
Figure 5b with n = 0.05. As seen for the other data sets in Figures 1–3, collapse of the curves is good.
Batteries 2016, 2, 17
5 of 7
Correlation
of Step 1) of the constant current discharge curves in Figure 5a yields Figure
5b7
Batteries 2016, (application
2, 17
5 of
with n = 0.05. As seen for the other data sets in Figures 1–3, collapse of the curves is good. Data for
aData
7.5 Afor
discharge
included is
forincluded
completeness;
however, after
approximately
960 mAh discharge,
the
a 7.5 A isdischarge
for completeness;
however,
after approximately
960 mAh
battery
wasthe
no longer
maintaining
We fitted power.
the function
defined
Equation
(4) to
discharge,
batterycapable
was noof
longer
capablepower.
of maintaining
We fitted
theby
function
defined
nV as aa=function
the
collapsed (4)
curves
in Figure
5b then
defined
in V as5ba then
function
of D i(with
12.3063, of
b =D´0.000328,
by Equation
to the
collapsed
curves
in Figure
defined
(with a =
´
7
´
7
´
10
−7
−7
−10).
c12.3063,
= ´0.008112,
d = ´4.7809
10 , e = ´7.7835
ˆ 10 × ,10
and
Implementation
b = −0.000328,
c =ˆ−0.008112,
d = −4.7809
, ef = 1.4086
−7.7835ˆ×1010 ,).and
f = 1.4086 × of
10the
constant
power discharge
procedure
yielded
the estimate
shown
in Figure
5c, presented
inin
conjunction
Implementation
of the constant
power
discharge
procedure
yielded
the estimate
shown
Figure 5c,
with
the experimentally
measured
In order tomeasured
clarify thecurve.
application
procedure,
1 contains
presented
in conjunction
with the curve.
experimentally
In order
to clarifyTable
the application
data
for the Table
first two
iterations.
procedure,
1 contains
data for the first two iterations.
(a)
(b)
(c)
Figure5.5.Electrify
Electrify
lithium
polymer
(LiPo)
extreme:
(a) constant
and (b) correlated
Figure
lithium
polymer
(LiPo)
extreme:
(a) constant
current;current;
and (b) correlated
discharge
discharge
curves.
n
=
0.05,
and
C
=
1300
mAh.
(c)
Comparison
of
prediction
and
experiment
curves. n = 0.05, and C = 1300 mAh. (c) Comparison of prediction and experiment for 34 W discharge.
for 34 W discharge.
Table 1. Initial calculations for 34 W discharge.
Table 1. Initial calculations for 34 W discharge.
j
1
2
j
1
2
ij (A)
ij (A)
2.914
2.914
2.931
2.931
Vj (V)
Vj (V)
11.670
11.670
11.601
11.601
D (mAh)D (mAh) t (h)
16.202
32.498
16.202 0.0056
32.498 0.0112
t (h)
0.0056
0.0112
The steps
steps for
for populating
populating the
the table
table follow
follow as:
as:
The
•
The time increment is given by 1/180 = 0.0056 h (20 s);
‚ The time increment is given by 1/180 = 0.0056
(20 s);
1/ (1h
− 0.05)
12.3063
¯1{p1´0.05=
q 11.67 V; and
•
From Equation (5) with j = 1: V1 =´ 12.3063
0.05
‚ From Equation (5) with j = 1: V1 “ 3434
= 11.67 V; and
0.05
3434
A; A;
‚• Equation
(6)(6)
gives:
i1 “
i1 =11.67 = 2.914
Equation
gives:
= 2.914
11.67
‚ The discharged capacity follows from Equation (7) as: D1 = i1 ∆t + D0 = 2.914 ˆ 103 ˆ (0.00556)
• + 0The
discharged
= 16.202
mAh; capacity follows from Equation (7) as: D1 = i1Δt + D0 = 2.914 × 103 × (0.00556)
+ 0 = 16.202
‚ Advancing
j =mAh;
2 and evaluating Equations (5)–(7) yields V 2 = 11.601 V, i2 = 2.931 A and
• D Advancing
j = 2 and evaluating Equations (5)–(7) yields V2 = 11.601 V, i2 = 2.931 A and D2 =
2 = 32.498 mAh.
32.498 mAh.
It must be mentioned that published data for lithium batteries is not consistent in indicating the
presence of capacity dependence on the rate of discharge (especially for high power cell technology).
For the data sets presented in this article, with battery capacities and discharge rates consistent with
Batteries 2016, 2, 17
6 of 7
It must be mentioned that published data for lithium batteries is not consistent in indicating the
presence of capacity dependence on the rate of discharge (especially for high power cell technology).
For the data sets presented in this article, with battery capacities and discharge rates consistent with
that of a small unmanned aerial vehicle, significant rate dependence (which depends on the anode and
cathode material as well as the electrolyte in the battery) is not evident. This result was also noted by
Chang and Yu [19]. A rate dependency (or Peukert type effect) may only manifest at high discharge
current values that would not be used if flying for maximum range or endurance. High discharge
rates (>20C) may reduce the data collapse afforded by Equation (2). Consequently, if the application
requires discharge rates above 20C, the proposed method should be used cautiously as it may not
be representative.
5. Conclusions
Estimates of range and endurance for battery powered aircraft are less well established than
those for fossil fuel powered vehicles. A recently developed method to estimate these parameters
(range and endurance) requires the variation of battery voltage at constant power as the battery is
discharged. However, standard testing procedure for batteries involves discharge at constant current.
Consequently, a procedure is developed to estimate constant power discharge curves for lithium
batteries using information from constant current discharge data. The method is valid for high power
cells and may not be applicable for high energy cells. Constant current discharge curves are shown to
correlate when the voltage during the discharge is multiplied by the current raised to a power (with a
value of « 0.05). Curve fitting the correlation then allows for the determination of the voltage variation
during discharge for a set power. Comparison of a prediction made using the correlation method and
an experimental constant power discharge showed close accord. The correlation method is not shown
to be applicable at very low temperatures and does require that the effective capacity is not affected by
the discharge rate.
Conflicts of Interest: The author declares no conflict of interest.
Abbreviations
C
D
e
i
j
K
M
N
n
P
t
V
ηtot
Capacity
Discharged capacity
Electrical
Current
Counter
Constant
Number of test cases
Time step increment
Exponent
Power
Time
Voltage
Total efficiency of propulsion system
References
1.
2.
3.
4.
Lawrence, D.A.; Mohseni, K. Efficiency Analysis for Long-Duration Electric MAVs. In Proceedings of the
Infotech@Aerospace Conferences, Arlington, VA, USA, 26–29 September 2005. [CrossRef]
Gur, O.; Rosen, A. Optimizing electric propulsion systems for unmanned aerial vehicles. J. Aircr. 2009, 46,
1340–1353. [CrossRef]
Ostler, J.N. Flight Testing Small Electric Powered Unmanned Aerial Vehicles. Master’s Thesis,
Brigham Young University, Provo, UT, USA, April 2006.
Retana, E.R.; Rodrigue-Cortes, H. Basic Small Fixed Wing Aircraft Sizing Optimizing Endurance.
In Proceedings of the 4th International Conference on Electrical and Electronics Engineering (ICEEE),
Mexico City, Mexico, 5–7 September 2007; pp. 322–325.
Batteries 2016, 2, 17
5.
6.
7.
8.
9.
10.
11.
12.
13.
14.
15.
16.
17.
18.
19.
7 of 7
Anderson, J.D. Aircraft Performance, 1st ed.; McGraw Hill: Columbus, OH, USA, 1999; pp. 293–300.
McCormick, B.W. Aerodynamics, Aeronautics and Flight Mechanics; Wiley: New York, NY, USA, 1995;
pp. 378–385.
Hale, F. Introduction to Aircraft Performance, Selection and Design; Wiley: Hoboken, NJ, USA, 1984; pp. 123–129.
Traub, L.W. Range and endurance estimates for battery powered aircraft. J. Aircr. 2011, 48, 703–707.
[CrossRef]
Traub, L.W. Validation of endurance estimates for battery powered UAVs. Aeronaut. J. 2013, 117, 1155–1166.
[CrossRef]
Peukert, W. Über die Abhängigkeit der Kapazität von der Entladestromstärke bei Bleiakkumulatoren.
Elektrotech. Z. 1897, 20, 287–288. (In German)
Su, Y.; Liahng, H.; Wu, J. Multilevel Peukert Equations Based Residual Capacity Estimation Method for
Lead-Acid Batteries. In Proceedings of the 2008 IEEE International Conference on Sustainable Energy
Technologies, Singapore, 24–27 November 2008; pp. 101–105.
Andrea, D. Battery Management Systems for Large Lithium Ion Battery Packs, 1st ed.; Artech House: Norwood,
MA, USA, 2010.
Sony. Lithium Ion Rechargeable Batteries Technical Handbook. Available online: http://www.
dlnmh9ip6v2uc.cloudfront.net/datasheets/Prototyping/Lithium%20Ion%20Battery%20MSDS.pdf
(accessed on 15 September 2015).
Sanyo. Lithium Ion Handbook. Available online: http://www.rathboneenergy.com/articles/sanyo_lionT_E.
pdf (accessed on 15 September 2015).
Arora, P.; White, R.; Doyle, M. Capacity fade mechanisms and side reactions in lithium-ion batteries.
J. Electrochem. Soc. 1998, 145, 3647–3667. [CrossRef]
PowerStream. High Discharge Current 18650 Rechargeable Cylindrical Lithium Ion Batteries from
PowerStream, Sony VTC4 and VTC5, Samsung 25R and LG HE2, Guaranteed Genuine. Available online:
http://www.powerstream.com/18650-high-discharge-rate.htm (accessed on 20 September 2015).
Thunder Power Extreme.
Available online: http://www.thunderpowerrc.com (accessed on 20
September 2015).
Smart, M.C.; Huang, C.-K.; Ratnakumar, B.V.; Surampudi, S. Development of Advanced Lithium-Ion
Rechargeable Cells with Improved Low Temperature Performance. In Proceedings of the 32nd Intersociety
on the Energy Conversion Engineering Conference, Honolulu, HI, USA, 27 July–1 August 1997; pp. 52–57.
Chang, T.; Yu, H. Improving electric powered UAVs’ endurance by incorporating battery dumping concept.
Proc. Eng. 2015, 99, 168–179. [CrossRef]
© 2016 by the author; licensee MDPI, Basel, Switzerland. This article is an open access
article distributed under the terms and conditions of the Creative Commons Attribution
(CC-BY) license (http://creativecommons.org/licenses/by/4.0/).