Chapter 8 Reactions of Alcohols I. Oxidation and Reduction of Alcohols
advertisement

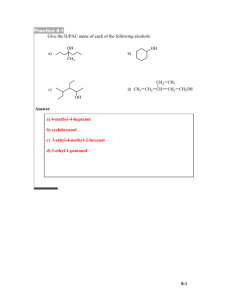
Chapter 8 Reactions of Alcohols I. Oxidation and Reduction of Alcohols A. B. Inorganic Oxidation and Reduction 1) Oxidation = loss of electrons: Cu+ 2) Reduction = gain of electrons: Zn2+ -2 e- +2 e- Cu3+ Zn0 Organic Oxidation and Reduction 1) Oxidation = addition of electronegative atoms (O, Cl, N) = removal of H O CH4 O 2) CO2 +2H CH3OH H2C O O HC OH -2H CO2 Reduction = removal of electronegative atoms (O, Cl, N) = addition of H O HC OH 3) -2H -O H2C O +2H CH3OH Relationship between alcohols and carbonyl groups 1) C==O = carbonyl -O CH4 2) Aldehyde is an oxidized Primary Alcohol (reduced aldehyde) O oxidation RCH2OH 3) reduction C R Ketone is an oxidized Secondary Alcohol (reduced ketone) O oxidation RR'CHOH C. H reduction C R R' Alcohol Synthesis by Reduction of Aldehydes and Ketones 1) Hydrogenation = adding H2 to a double bond H H H2 cat C C 2) 3) Catalyst = reactant that doesn’t get destroyed; it speeds up the reaction by lowering the Ea for the reaction a) Heterogeneous Catalyst = insoluble, reaction occurs at its surface i. Fine powder increases surface area (put cat. on fine charcoal) ii. Pd/C or Pt metal or Ni metal work as hydrogenation cat.’s b) Homogeneous Catalyst = soluble, reaction occurs in solution Hydrogenation of Carbonyls gives Alcohols C O H2 cat aldehyde R C O H H H R H R H C C 1o ROH C O R' ketone H2 cat R C O H R' 2o ROH D. Alcohol Synthesis by Hydride Reduction of Carbonyl’s 1) Carbonyl groups are polar - O - O C R + R' C + R' R C R' - 2) Hydride = H has several efficient sources for Organic Synthesis a) Na+H- or Li+HM+ + H- (not very organic soluble) b) NaBH4 Na+ + B(solvent) + 4 Hc) LiAlH4 Li+ + Al(solvent) + 4 H- 3) Reduction of Carbonyl’s to Alcohols R R O H R H NaBH4 C R O C O H 4) H C LiAlH4 O H R R' C O H R' NaBH4 Mechanism OH R' H3B H C O R H OR'' R' C H R + H3B OR'' B(OR)4 5) LiAlH4 is too reactive to use in Protic Solvents H3Al H H3Al H E. H2(gas) + Al(OH)4 H OH 6) LiAlH4 in Aprotic Organic Solvent R' R' x4 H2O C O R C O Al EtOEt "aqueous 4 R H workup" R' 4 R C OH + Al(OH)3 H Oxidation of Alcohols to Carbonyls 1) Reduction reactions can be reversed to give the aldehydes or ketones 2) Oxidizing Reagent is Cr(VI) a) (Na2Cr2O7 or K2Cr2O7 or CrO3) and H2SO4 and H2O R' CH OH R b) Na2Cr2O7 H2SO4, H2O R' C O R Primary alcohols can be overoxidized to carboxylic acids RCH2 OH Na2Cr2O7 H2SO4, H2O O R C OH 3) Do Primary alcohol oxidation without water NH+ CrO3Cl a) PCC = pyridinium chlorochromate = b) Anhydrous conditions and PCC don’t overoxidize primary alcohol R CH2 OH 4) O PCC CH2Cl2 R C Mechanism involves Chromic Ester O O RCH2 OH + VI VI HO Cr OH RCH2O Cr O RCHO Cr OH H O OH + H2O O Chromic Ester O H2O H Like E2 O RCH + H3O+ + CrVIO3H II. Organometallic Reagents = carbon-metal bonds A. Nucleophilic Carbon 1) H- is a nucleophilic hydrogen that gives new C—H bonds OH R' H3Al H C O R' R 2) R Formation of new C—C bonds is the key requirement in organic synthesis OH R' R''CH2- C O R B. C H R' C CH2R'' R Alkylmetal Reagents 1) Haloalkanes can be transformed into organometallic compounds a) Alkyllithium Synthesis (I > Br > Cl reactivity) ether or THF RBr + 2 Li 0 oC b) O RLi + LiBr Tetrahydrofuran Alkylmagnesium Synthesis (Grignard Reagent) ether or THF RI + Mg RMgI 0 oC 2) O R R- MgX+ + H OH Mg O R Alkylmetals are very strong Nucleophiles a) Use them as soon as you make them (…made in situ…) b) Air and water sensitive, must do reaction under N2 or Ar Hydrolysis RH + XMOH I 3) True structure involves coordinated solvent—require ether or THF 4) Very polar bond, metal is very electropositive a) Opposite of usual situation for carbon, as in Haloalkanes b) Treat the molecule like R- = Carbanion c) Resonance forms M + C 5) M C + M Alkylmetals are very basic a) Basicity: RCH2- > RNH- > RO- (electronegativity) b) Acidity: RCH3 < H2NR < ROH c) Leads to fast hydrolysis (see above) in protic solvents 6) Making Alkanes from Haloalkanes a) Use alkyl metal hydrolysis R- MgX+ + H OH RX + Mg b) Use hydride nucleophile Br c) LiAlH4 ether Useful for Deuterating compounds (D = deuterium = 2H) Br C. RH + XMOH LiAlHD4 ether D Alkylmetal reagents in Alkanol Synthesis 1) Formation of new C—C bond by using C Nucleophile 2) Nucleophilic attack of Haloalkane by alkylmetal reagent is too slow - + R' MgX slow + Br R' 3) Must use Carbonyl electrophile (C+==O-) a) Ketone gives tertiary alcohol product O RLi + ether or THF C R' O R' R' C OH + H , H2O C R' R' R R b) Aldehyde gives a secondary alcohol product O RLi + ether or THF C R' OH + H , H2O H H C R' R c) R' Formaldehyde (CH2O) gives primary alcohol product O RLi + ether or THF C H H + H , H2O OH H C R H III. Synthetic Strategies A. Use Mechanisms to predict the products of a reaction II1) I 2) B. Br F Br F CH3 Br CH3 Br2 I CH2Br Br2 Know all of the tools (reactions) you can use Nu, DMSO Br Mg, ether (CH3)3CO(CH3)3COH Nu H2O H2CO MgBr CH2OH O OH C. Synthesis of Complex Alcohols Using Alkylmetal Reagents 1. Useful Reaction Sequence CH4 Br2 CH3Br -OH CH3OH PCC CH2Cl2 H2C O + RMgBr H THF RCH2OH OH2 2. Retrosynthetic Analysis = Work Backwards from Target a) Complex molecules are made from simple parts b) Look for C—C bonds to break (form) OH CH3CH2CH2 CH2CH3 strategic disconnection CH3CH2CH2MgBr + CH3 CrO3, H2SO4, H2O O H+, H2O THF H3C H3C CH2CH3 OH CH2CH3 H H+, H2O THF O CH3Li + H CH2CH3 3) Avoid Synthetic Pitfalls a) Use fewest possible steps i. 2 steps at 90% yield = (0.9)(0.9) = 81% total yield ii. 4 steps at 95% yield = (0.95)(0.95)(0.95)(0.95)= 81% total yield b) Convergent Synthesis is better than Linear Synthesis A B C D (50% yield at each step) 80g 20g E 40g F 20g 10g 10g D 10g 20g c) G H 10g Don’t use functional groups that would interfere in the reaction O MgBr d) + HO Remember mechanistic limitations i. Br2 is very selective form tertiary H ii. SN2 can’t happen for tertiary electrophile Protic alcohl kills Grignard