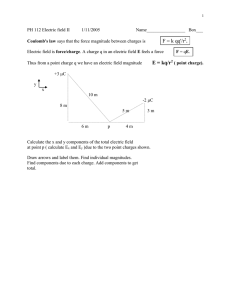
Electric Force
advertisement

Electric Force • One of the four fundamental forces • Responsible for much of our technology • Governs chemistry which deals with interactions of the outer electrons between atoms or groups of atoms • Interesting topic because it also involves the concept of magnetism Electric Force • We are all familiar with static electricity • When the humidity is low, we can walk across a carpet wearing leather-soled shoes and give someone a big awakening • What is happening here? • Transferring some entity into our fingertips Electric Charge • We now know that elementary particles like the electron and proton have a quality known as charge. • Objects that accumulate charge have either gained or lost electrons • These objects experience large forces as a result of being “charged” Electric Charge • There are two kinds of charges which we have called positive and negative • We have defined the charge of the electron to be negative and the charge of the proton to be positive • What it is interesting is that “Like charges repel and unlike charges attract!” Electric Charge • Earlier we studied some conservation laws that turned out to be very important • The conservation of energy, linear momentum and angular momentum have major consequences for the way the world behaves • Electric charge is also a conserved quantity and for every positive charge there seems to be a corresponding negative charge Conservation of Charge • In any process, the net amount of electric charge produced is zero! • This means that if we add up all the positive charges and all the negative charges moving around in a process, the total charge will sum to zero • So we can neither create nor destroy charge Electric Charge • Charged objects are composed of atoms, a few of which have either gained or lost an electron • Those special atoms are called ions • On a macroscopic level, ions eventually gain or lose an electron from contact with something else and return to neutral status Insulators and Conductors Insulators and Conductors • Materials such as metals conduct electricity • This means that charges can move freely through the material (electrons do the moving) • Other materials, such as wood or glass do not conduct electricity • Electrons cannot move freely in these materials Insulators and Conductors • A few materials fall somewhere between these two extremes and are called semiconductors • Examples are silicon and germanium • The key is whether or not the electrons in the material are tightly bound to individual atoms Insulators and Conductors • In metals, not all the electrons are required for bonding the atoms into a lattice • At least one electron per atom is free to move around at will in the material • It is almost like the molecules of air can move freely anywhere in a room Insulators and Conductors • Since the metals have free electrons, they can easily allow charges to move • In insulators, the electrons are tightly bound to individual atoms and are not free to move • The result is they do not conduct electricity • In semiconductors, only a small fraction of the electrons are able to move. We’ll learn more next semester Induced Charge Touch a neutral metal rod with a charged metal rod and you will transfer some charges to the neutral rod Induced Charge We have induced charges at the ends of the neutral rod. Bring a charged metal rod close to a neutral metal rod and you can separate charges on the neutral rod. Note charge conservation is at work. Induced Charge The earth is large and can conduct. So, when you separate the charges and allow the electrons to flow to the earth, you can leave a charged object behind. Coulomb’s Law By sharing charges between two objects, Coulomb was able to make charges that were 1/2, 1/4, etc. of the original charge on a rod. Q1Q2 F k 2 r He then could measure the forces between two charged objects. Coulomb’s Law • The standard unit of charge is the Coulomb • We’ll define it later in terms of magnetic fields, but for now all we need is the constant k in SI units. • We want force in Newtons, distance in meters and charge in Coulombs • K = 8.988 x 109 N m2/C2 Coulomb’s Law • The Coulomb is a HUGE charge • The electron has a charge 1.6 x 10-19 C and is designated as e. • The net charge on any object is some multiple of this fundamental charge • We say charge is quantized because there is a smallest value, namely e Coulomb’s Law • Strictly speaking, Coulomb’s Law applies only to point charges (no finite extent in space) • However, for spheres, can consider all the charge concentrated at a point in the center • Just like gravity applies to a sphere (like the earth) Coulomb’s Law • Compare electrical to gravitational force in a hydrogen atom 1.6 10 8.2 10 0.5310 19 Felectrical 9 10 9 Fgravity 6.67 10 2 8 10 2 27 31 1.67 10 9.1110 11 0.5310 10 2 3.6110 47 Coulomb’s Law • The electric force is a vector! • Superposition of forces Fnet = F1 + F2 + … Coulomb’s Law • The electric force is a vector! • Superposition of forces Fnet = F1 + F2 + …