Honors Biology Ch.7 Photosynthesis Notes
advertisement

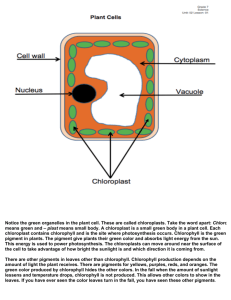
Honors Biology Ch.7 Photosynthesis Notes Opening Essay: Explain how plants can be used as a renewable energy source. Explain why this is better than burning fossil fuels. Renewable: Plants remove CO2 from the atmosphere Plants add oxygen to the atmosphere Plants reduce the greenhouse effect Plants can be grown Fossil Fuels: Fossil fuels (ancient plant remains) are running out Fossil fuels are not renewable Burning fossil fuels releases stored CO2 into the atmosphere causing the greenhouse effect. Biofuel: A better energy source because the CO2 added to the atmosphere by burning biofuel was taken out of the atmosphere by growing the plants. This keeps the atmospheric CO2 in balance. An Overview of Photosynthesis 7.1 Define autotrophs, producers, and photoautotrophs. Autotrophs: “self-feeders” They make their own food and sustain themselves without other organisms. Producers: Using the energy from the sun and chlorophyll, these organisms make their own glucose which is used by other organisms as food. Producers are always the base of the food chain. Photoautotrophs: All organisms that produce organic molecules from inorganic molecules using the energy of light. Examples: Prokaryotes: cyanobacteria (aquatic) Eukaryotes: Plants (both aquatic and terrestrial) protists (like euglena, kelp, green algae) (aquatic: both marine and fresh water) 7.2 Describe the structure of chloroplasts and their location in a leaf. Identify specifically where most light energy is converted to chemical energy. Describe the structure of chloroplasts Double membrane organelle Thylakoid membrane encloses a space called “thylakoid space” A stack of thylakoids is called a granum or granal stack Space surrounding thylakoids is the stroma Chlorophyll and photosynthetic enzymes are embedded in thylakoid membrane the site of the conversion of light into chemical energy. Essentially; it is two spaces separated by a membrane. Mrs. Loyd cloyd@waukee.k12.ia.us Page 1 of 4 http://loydbiology.weebly.com 10/20/11 http://www.mybiology.com Describe the location of chloroplasts in a leaf Chloroplasts are located in mesophyll cell (means: “middle leaf”) Mesophyll: Palisade mesophyll: most photosynthesis Spongy mesophyll: surrounds stoma How do the reactant molecules of photosynthesis (CO2 + H2O) reach the chloroplasts in leaves? How does leaf structure promote this process as well as light absorption? 7.3 Explain how plants produce oxygen Plants produce oxygen when water (H2O) is split in the stroma. The hydrogen is used to make sugar and the excess oxygen diffuses out of the leaf. 7.5 Compare the reactants and products of the light reactions and the Calvin cycle. Explain how photosynthesis relates to these reactions. Light Rxn Calvin Reactants H2O NADP+ ADP + P light energy CO2 ATP NADPH (e-s) Products O2 ATP NADPH (e-s) NADP+ ADP + P glucose Explain how photosynthesis relates to these reactions. The light reactions convert light energy into the chemical energy of ATP. The Calvin cycle uses the ATP as the energy to build 6CO2 + 6H2O into C6H12O6. The Light Reactions: Converting Solar Energy to Chemical Energy 7.6 Describe the properties and functions of the different photosynthetic pigments. LIGHT ENERGY Electromagnetic Spectrum of Energy: Gamma – X-rays – UV – visible – Infrared – Microwaves – Radiowaves Visible light spectrum: Red Orange Yellow G.reen Blue Indigo Violet Infrared – ROY G. BIV – Ultraviolet (UV) (heat lamp) (sunburn/skin cancer) Plant Pigments Why are plants green? Reflected wavelengths (color) reach our eyes and we see green. Mrs. Loyd cloyd@waukee.k12.ia.us Page 2 of 4 http://loydbiology.weebly.com 10/20/11 http://www.mybiology.com Is green light important to plants? No, green light is reflected or “thrown away.” Pigment properties and functions Pigments absorb light of different wavelengths Chloroplasts contain several kinds of pigments o Chlorophyll a: absorbs blue-violet, looks grass green o Chlorophyll b: absorbs blue and orange, looks yellow-green o Carotenoids: absorb , look yellow to orange More kinds of pigments broadens the spectrum of colors that can drive photosynthesis. All pigments must pass energy absorbed to chlorophyll a, which serves as the reaction center of photosystems. Why do leaves change color and drop in autumn? 1. The change to cold weather causes the most sensitive and abundant pigment, chlorophyll a, to break down and disappear from the mesophyll cells in leaves. 2. The colors of the other pigments particularly the carotenoids, previously masked by chlorophyll a, are revealed. 3. Bright reds, oranges, and yellows are typical in the fall. 4. Once the temperature falls more drastically, the remaining pigments breakdown and the leaves turn brown. 5. Abscisic acid is released by the tree to cause the leaves to abscise or drop thus greatly reducing surface area and water loss during winter. Photosynthesis, Solar Radiation, and Earth’s Atmosphere 7.13 Describe the greenhouse effect. Explain how deforestation and the use of fossil fuels contribute to global warming. Greenhouse Effect: Sunlight warms Earth’s surface, Radiates heat to the atmosphere, CO2 and other gases absorb and radiate some heat back to Earth. The higher the concentration of greenhouse gases (CO2, water - H2O and methane - CH4), the higher the global temperature. History: Before 1850 CO2, = < 0.03% of air we breathe After industrial revolution, increased to 40% Due mostly to combustion of fossil fuels Deforestation: Coincided with industrial revolution Timber cleared for fuel and agriculture Rise in population increased demand for fuel Trees acting as a carbon “sink” were cut down Mrs. Loyd cloyd@waukee.k12.ia.us Page 3 of 4 http://loydbiology.weebly.com 10/20/11 http://www.mybiology.com 7.14 Explain how the ozone layer forms, how human activities have damaged it, and the consequences of the destruction of the ozone layer. Ozone: Photosynthesis produces O2. Upper atmosphere converts O2 into O3. O3 acts as shield from UV radiation UV can mutate and destroy DNA. 1974 CFCs were identified as destroying ozone o Cholorfluorocarbons (CFCs) used as: o Refrigerants o propellants in spray cans o solvents o plastic manufacturing. 1985 ozone hole identified over Antarctica Mrs. Loyd cloyd@waukee.k12.ia.us Page 4 of 4 http://loydbiology.weebly.com 10/20/11 http://www.mybiology.com