Document 15561517
advertisement

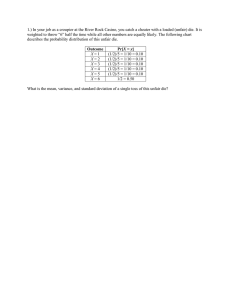
DEFINITION: Characterization of physical, chemical and mechanical properties of new drug molecule in order to develop safe, effective,and stable dosage form. GOAL OF PREFORMULATION: To formulate an elegant, safe, efficacious dosage form with good bioavailability. To formulate new dosage form of already existing drug. Determination of all the properties of drug and the best suitable dosage form for the drug molecule. Pharmaceutical factor mainly include those parameters of drug which affect the final dosage form manufacturing process like….. Flow property Density Compressibility Hygroscopicity Electrostatic charge Osmolarity Rheology Wettability Syringabilty 1)FLOW PROPERTY (A) Introduction Flow property is an important factor that determines the fate of drug molecule. Sufficient flow is required for uniformity of dosage form. So it is necessary to judge the flow of material in preformulation stage of the dosage form. However extreme increase in flow may improve weight uniformity but may reduce content uniformity through increased segregation. (B) Method of determination By Angle of repose By hopper flow rate By bulk density By angle of spatula By vibrational capillary method LATEST TECHNOLOGY TO DETERMINE FLOW PROPERTY REPOSOGRAPH: It is a stable instrument which at best can only indicate comparative flow properties. The formation of sharp cone would mean poor flow property while a good spread would indicate a superior flow property. FT (FREEMAN TECHNOLOGY) RHEOMETER: • An instrument for measuring flow property. • It can discriminate between the samples that differ by 1% Moisture. • Important for optimizing granulation because moisture variation have significant impact on final product quality. NEW MEASUREMENT SYSTEM TO EVALUATE POWDER FLOWABILITY BASED ON VIBRATIONAL CAPILLARY METHOD: Evaluates flowability of micrometer sizes particles under actual flow condition. The amplitude and frequency of vibration is controlled by computer and mass of powder discharged from vibrating capillary tube is measured by digital balance. The mass flow rate is measured by digital processing. [Chemical Abstract ,Jan. 2007, 146:9806] Angle of Repose Indirect method of quantifying powder flowability,because of their relationship with interparticle cohesion. It is a maximum angle between the surface of a pile of powder & horizontal plane. Angle of repose is measured by the equation: tanθ=h /r here, h=height of conical heap & r=radius of horizontal plane of powder DETERMINATION OF ANGLE OF REPOSE Static angle of repose Fixed height cone, Fixed base cone, Tilting table Dynamic angle of repose Rotating cylinder Rotating Drum Drained angle of repose Ledge type, Crater type RELATION BETWEEN ANGLE OF REPOSE & TYPE OF FLOW & TYPE OF POWDER Angle of repose <25 Type of flow Excellent Type of powder Non cohesive 25-30 Good Non cohesive 30-40 Passable Cohesive >40 Very poor Very Cohesive HAUSNER’S RATIO This is a simplex index that can be determined on small quantities of powder. Hausner-ratio= Tapped densityβ max) poured density(Pβ min) Hausner ratio Type of Flow <1.25 1.25-1.5 >1.5 Good flow Moderate Poor flow (C) FACTOR AFFECTING FLOW PROPERTY Particle size and Particle size distribution Particle shape and Surface roughness Density and Porosity Hygroscopicity Electrostatic charge (D) IMPROVEMENT OF FLOWABILITY By addition of glidant By addition of fine or by size reduction By wet granulation By removing static charge By densification with the help of slugging Using auger feed equipment By addition of flow activator. Eg. MgO By use silicon treated powder for Hygroscopic & moist powder. e.g. silicon coated talc or Nabicarbonate By altering process condition like vibration assisted hopper or forced feeder By use of spray drying : Advantose 100 maltose powder has improved flow property than MCC by using this process. 2. DENSITY (A) INTRODUCTION : The ratio of mass to volume is known as density. Density = Mass (gms.)/ Volume (ml.) TYPES OF DENSITY : (a) Bulk density (b)Tapped density (c)True density (d)Granule density :- may affect compressibility, tablet porosity, disintegration, dissolution (B) Method of Determination Parameter Method 1. Bulk density Measuring cylinder 2. Tapped Density Mechanical Device Mercury Displacement 3. True Density Helium densitometer (Helium Pycnometer) Mercury Instrution Porosimetry Bulk density measurement : It is determined by pouring presieved (40-mesh) bulk drug into a graduate cylinder via-a large funnel and measuring the volume and weight. Tapped density measurement: It is determined by placing a graduated cylinder containing an known mass of drug or formulation on a mechanical tapper apparatus, which is operated for a fixed numbers of taps(about-1000)untill the powder bed volume has reached a minimum. Measurement of True Density: True density can be determined using three methods: displacement of a liquid, displacement of a gas (pycnometry), or floatation in a liquid. The liquid displacement is tedious and tends to underestimate the true density. Displacement of a gas is more accurate, but needs relatively expensive instrumentation. Gas pycnometers rely on the measurement of pressure changes, as a reference volume of gas, typically helium, added to, or deleted from, the test cell. As an alternative, the floatation method is simple to use and inexpensive. An improved method for fast online measuring of density of solid substances: A densitometer for measuring of bulk density of solid & liquid consists of one vibrator means, to support pre-weighed samples to be tested in container with predefined shapes on this vibrator & atleast one ultrasonic sensor operatively connected to control unit such that sensor is adapted to transmit and receive reflected ultrasonic pulses to ascertain density of samples utilizing the said values of the fill levels of samples in containers. (chemical abstract ,October 2007) (C) CORRELATION WITH FLOWABILITY Carr’s index = Tapped density- Bulk density/ Tapped density Hausner ratio = Tapped density / Bulk density (D) IMPORTANCE In case of combination therapy or physical mixture ,if both drug or drug & excipients have different density then creates problem of segregation (demixing). Important in decide size & type of processing equipment. E.g. decide size of capsule formulation, Suppositories. Devereux et.al. compared GI transit time of multiple unit formulation of densities 2.8g/cm3 & 1.5g/cm3 & found significantly delayed gastric emptying of heavier pdt. (Review article, IJPS, sept-oct,2008) 3. COMPRESSIBILITY (A) INTRODUCTION Compressibility is the ability of powder to decrease in volume under pressure. %compressibility = Tapped density – bulk density * 100 Tapped density Neumann and Carr developed a simple test to evaluate flowability of a powder by comparing the poured (fluff) density (Pβmin)and tapped density (Pβmax) of a powder and the rate at which it packed down. Useful empirical guide is given by the Carr's compressibility index. here compressibility is misnomer since compression is not involved. Relationship between powder flowability and % compressibility SR. NO % COMPRESSIBILTY RANGE FLOW DESCRIPTIONS 1 5-15 Excellent (free flowing granules) 2 12-16 Good ( free flowing powder granules) 3 18-21 Fair to passable ( powder granules 4 23-28 Poor ( very fluid powder) 5 28-35 Poor ( fluid cohesive powder) 6 35-38 Very poor ( fluid cohesive powder) 7 >40 Extremely poor ( cohesive powder) (B) The characteristics Of material may be :1. PLASTICITY Plastic material are capable of permanent deformation, also exhibit a degree of brittleness (fragmentability) But plastic material will get bonding after Viscoelastic deformation. 2. FRAGMENTABILITY If material is fragmentable, neither lubricant mixing time nor dwell time affecting the tablet strength. 3. ELASTICITY E.g. paracetamol, acetyl salicylic acid If material is elastic, it rebound when compression force is released. Elastic material may lead to capping & lamination They require wet massing to induce plasticity or plastic tableting material. 4. PUNCH FILMING [STICKING]: This may lead to chipping of tablet. (C) METHOD OF IMPROVEMENT If plastic material add fragmentable excipient e.g.. Lactose . If Elastic material By plastic tableting material Wet granulation , Pre compression. If sticky material By change in salt form, By using high excipient ratio, By wet massing, By addition of Mg-stearate. NPTAB Technology: Innovative technology combining pellet coating with direct compression. Active drug is sprayed on carrier containing sugar sphere & these layered spheres are directly compressed. “ Multifunctional co-processesed excepients with improved compression properties are used for improved tabletting performance.” (CHEMICAL ABSTRACT, July 20,2009,151, No.3: 63288g) 4. HYGROSCOPICITY (A) INTRODUCTION Hygroscopicity: - It is the tendency of material to absorb moisture from atmosphere & be dynamic equilibrium with water in the atmosphere. Deliquescent: - It is the hygroscopic substance which absorb moisture from air and they can be liquefied by partially or wholly forming solution. Efflorescent: - a substance which loses water to form a lower hydrate or become anhydrous is term as efflorescent. List of examples: Hygroscopic & Deliquescent Ephedrine Hyoscymine Phenobarbital Pilocarpine Physostigmine Efflorescent atropine cocaine codeine scopolamine caffeine Glycerinated gelatin & PEG base of suppository are hygroscopic in nature. (B) METHOD OF DETERMINATION To carry out study, sample of compound are accurately weighed into container and placed at various humid condition for period of upto 2 weeks. If Weight gain – Deliquescent or Hygroscopic If Weight loss – Efflorescent Also determined by TGA, GC, & KF titration Versaperm has deviced a WVTR meter that can measure the permeability of package to moisture in as little as 30 min.so that humidity can be accurately controlled. (C) IMPORTANCE: It affects the flow property. It affects compression characteristic , granulation & hardness of final tablet. It also affects compaction. Important in aerosol. Affects chemical stability of hydrolysable drug. (E) METHODS OF IMPROVEMENT For granulation of hygroscopic material use nonaqueous solvent. For efflorescent material , use anhydrous salt. Add finely powdered adsorbents like MgO or Mg carbonate. Perform the entire tableting operation under controlled humidity condition. Store in desiccant, foil, blister, glass bottle. Use of Ion-exchange resins. Eg. Complexation of Ranitidine with Indion234. ( Journal of Pharmaceutical Research, vol.8.No.2, Apr 2009:112-115) Examples: Starch is hygroscopic ,but on pregelatinization it exhibits lower propensity for moisture, thus providing excellent stabilization for moisture sensitive active drugs. A new multifunctional excipient, Galen IQ. Problem associated with Hygroscopic material: Stick- slip mechanism of powder flow: It is pulsatile flow of granular material. It causes problem in die filling for tableting. “ The length of stick slip event increases with moisture content, increasing load, etc .” (Chemical Abstract vol.146:9803) 5. ELECTROSTATIC CHARGE INTRODUCTION: A) Electrostatic charges are the consequence of classic attraction & repulsion effect between the charges. Electrostatic charge is produced: By separation of positive & negative charge By mechanical impact By friction between two surface By rupturing of particle By separation of solid & liquid surface Pharmaceutical processing procedure such as mixing,micronizing, milling, sieving, rubbing, compressing, spray drying & congealing, pan coating & packaging can induce static charge. (B) METHOD OF DETERMINATION: INOSTAT, measures negative charge on the surface in volts/cm, when material is flowing from hopper. ELPI(13-stage Electrical Low Pressure Impactor), gives detailed charge profile of MDI aerosol particles. This has practical application on lung deposition of MDI aerosol. Electrostatic testers which consists of electrostatic voltage sensing probes. C) FACTORS AFFECTING STATIC CHARGE Effect of particle shape. e.g.- PCM Fine crystalline form >Crystalline form > Granules with EC > Granules with starch. Effect of tablet excipient. e.g.- Acetaminophen with mannitol(+++) > SDL(++) > Mg stearate(+) Effect of Particle size. Effect of moisture. (D) IMPORTANCE In preformulation of suspension . Affects flow property of powder. Affects mixing process. For thermal stability of emulsions. It may damage tablet machine. It may affect compression coating. E) METHOD OF REMOVAL OF STATIC CHARGE Addition of diluents or lubricant. Surface coating of particle with amphiphilic substance of o/w type. Eg Aerosol. Use crystallization method using more polar solvent. By granulation. Store under influence of air with sufficient humidity. Super critical fluid technology. Example: Poloxamer reduce electrostatic charge on the surface of polystyrene. 6. OSMOLARITY A) INTRODUCTION It is a colligative property DEFINITIONS: Osmoles : No. of osmotically active particles in solution. Osmolarity : osmoles or milliosmoles per liter of solution. Osmolality :osmoles or milliosmoles per kg of solvent. Isoosmotic: when two different solutions are separated by semipermiable membrane have same osmotic pressure so called as isoosmotic. Isotonic: when two different solutions are separated by biological membrane have same osmotic pressure so called as isotonic. (B) Method of Determination Osmolality should be measured carefully with a vapour pressure or freezing point osmometer or cryoscopic osmometer. Vapour pressure osmometer: Measures concentration of osmotically active particles that reduce V.P of solution. Membrane osmometer: This invention is directed to a membrane osmometer for direct measurement of osmotic pressures. ( United States Patent 4455864 ) Clifton nanolitre osmometer (C) IMPORTANCE Normal serum osmolality to be 285 mosmol/kg. Maintain osmolarity by 1%variation. It should be proper maintained in Oral nutrition fluid Peripheral infusion Parenteral product Ophthalmic preparation Administration of Paratonic solution can lead to crenulation or lysis of RBC. 7. RHEOLOGY (A) DEFINITION It describes flow of liquid and/or deformation of solid under stress. (B) TYPE OF FLOW: Newtonian flow Non Newtonian flow NEWTONIAN FLOW It is a flow in which a direct proportionality exists between shear stress and shear rate. E.g. water, simple organic liquid & dilute suspension , Glycerin. NON NEWTONIAN FLOW Where there no direct relation between shear stress and shear rate. There are three type (1) PLASTIC FLOW It is the Newtonian system at shear stress above yield value. Eg. Flocculated suspension. (2) PSEUDOPLASTIC FLOW Here yield value not associated .As applies shear stress increasing, viscosity decreases and disarranged molecules begin to align their long axes inline of molecules. Eg. Aq. Dispersion of tragacanth, Na-CMC, PVP. (3) DILATANT FLOW Opposite to pseudoplastic flow Increase in the shear rate, increasing in resistance to flow as viscosity increases. E.g. deflocculated suspension of Mg magma RHEOGRAM (C) DETERMINATION OF VISCOSITY Capillary viscometer Falling sphere viscometer Cup and bob viscometer Cone and plate viscometer Brook field viscometer Ultrasonic Shear Rheometer :- For analysing protein solution rheology. Instron Capillary Rheometer :- Measures viscosity as a function of rate of shear & temp at a high rate of shear. D)IMPORTANCE [1] FLUID For mixing For particle size reduction of disperse system Passing though orifice, pouring, packaging in bottle, passing though hypodermic needle. Flow though pipe Physical stability of disperse system [2] QUASISOLIDS Spreading and adherence to skin Removal from jar Capacity of solids to mix with liquid Release of drug from base [3] SOLID Flow of powder from hopper and into a die cavity in tableting or in encapsulation Packagability of powder or granules solids. [4] PROCESSING Production capacity of the equipment Processing efficiency THIXOTROPHY: In thixotropy apply shear stress convert gel – sol & remove shear stress convert sol – gel, means gel to sol to gel. Application :- for stability of suspension e.g. conc. Parental suspension containing 40-70% w/v of procaine penicillin G 8. WETTABILITY (A) INTRODUCTION Wettability of a solid is an important property with regards to formulation of solid dosage form. Adsorption at solid surface is involved in wetting & detergency. It may influence granulation of solid, penetration of dissolution fluid into tablet and granules & adhesion of coating material to tablet. (B) METHOD OF DETERMINATION : By contact angle: The contact angle is the angle between a liquid droplet and the surface over which it spreads. Contact angle – 00 – complete wetting. Contact angle – 1800 – No wetting . By Draves test: (C) IMPORTANCE: Crystal structure can influence the contact angle. Problems associated with Wettability of powder are poor dissolution rate & low adhesion of film coating. (C) IMPROVEMENT Mixing with hydrophilic excipient like Na CMC (water soluble) and bentonite, Al Mg silicate & colloidal silica (water insoluble). Use of wetting agent (HLB value 6-9) which acts by lowering contact angle. It displaces air & replace it with liquid phase. Wetting of powder by non aqueous solvent can be enhanced by certain lanolin derivative. 9. SYRINGABILTY It is more mechanical property rather than pharmaceutical property. This phenomenon happens when a liquid dosage form passes through a syringe. The flow of material is dependant on size & shape of crystals of material. Plates can easily move one over another .so, no friction observed & can easily pass through the syringe. While in case of needles or cubes or prisms , they can’t pass through the syringe easily. So, we can arrange the degree of syringabilty in following way: Plates > Needles > Cubes > Prisms REFERENCES Alfred Martin, physical Pharmacy, 4thedition, 1999, B.I. Waverly, New Delhi. Leon Lachman ,H.A. Lieberman , J.L.Kanig , the theory and practice of industrial pharmacy ,2nd edition Leon Lachman, H, A. Lieberman, Pharmaceutical dosage form –tablet volume 1 Leon Lachman, H, A. Lieberman, Pharmaceutical dosage form-parental Dosage form volume 1 Michael E. Aulton, Pharmaceutics: The science of dosage form design ELBS publication Remington, the science and practice of pharmacy, 21st Edition Encyclopedia of pharmaceutical technology vol. 14 Marcel Decker E.D.Summer et al. Journal of Pharmaceutical Science, 55:1441(1966) G.Gold et al, Journal of Pharmaceutical Science, 57:667(1968) F.Q. Danish et al, Journal of Pharmaceutical Science, 60:548(1971) S.Dawoodbhai C.T. Rhodes Drug and Industrial Pharmacy, 15: 1577(1989) Journal of pharmacuetical science( April 1999, vol. 88 no.4) G.Dold, B.T. Palermo Journal of Pharmaceutical Science, 54:311. Manufaccturing chemist( Jan 2005, Jan 2004 , Dec-Jan 2003) Chaquan Sun, Journal of Pharmaceutical Science, 93:646(2004). Journal of pharmacuetical science( Dec 2005 vol.94 ,no. 12) Indian Journal of pharmacuetical science (Sept-Oct 2008) Chemical Abstracts. Journal of Pharmaceutical Research,vol.8,No.2(Apr. 2009).