2 Boyle's Law.doc
advertisement

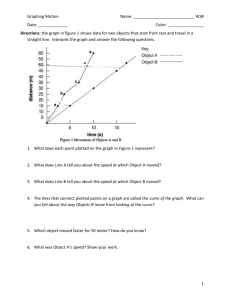
Boyle’s Law THEORY: Boyle’s Law states that the volume of a given quantity of an ideal gas varies inversely with the pressure at constant temperature. If P is pressure and V is volume, then the law can be expressed as: PV = k, where k is a constant. Similarly, the pressure and volume at two different pressures are related by: P1V1 = k = P2V2 By computing the pressure-volume product at constant temperature, Boyle’s Law may be verified. Plotting the pressure of the gas vs reciprocal volume will yield a straight line. EQUIPMENT: The Boyle’s Law apparatus consists of two tubes, the larger one to contain water and the smaller one to be used to measure the compression of the air column as a function of the depth the smaller tube is immersed upside down in the water. PROCEDURE: 1. Fill the larger cylinder about ¾ full of water. 2. Record barometric pressure, Po, and length of small cylinder, Lo, on data page. 3. Invert the small cylinder and immerse it into the water to a depth of 100 mm. Measure and record the depth of the small tube, D, and the depth of the bottom of the air column, H. 4. Repeat procedure 3 for depths of 200 mm through 800 mm in 100 mm increments. CALCULATIONS: 1. For each depth calculate the effective “volume” of the gas column, V = Lo - H 2. Calculate the reciprocal of each volume 3. For each depth calculate the absolute pressure in the air column, P = Po + (D – H)/13.55. The 13.55 converts the pressure due to the water depth into the pressure due to an equivalent depth of liquid mercury. 4. Plot a graph of Pressure vs Volume on the LabQuest: o Select the Table function, x|y o Enter your data into a table o Select Graph function o Select Analyze/Curve Fit/y/Choose Fit/?/OK 5. Record curve fit data 6. Print graph if possible 7. Plot a graph of Pressure vs 1/Volume on the LabQuest: o Select the Table function, x|y o Enter your data into a table o Select Graph function o Select Analyze/Curve Fit/y/Choose Fit/Linear/OK 8. Record curve fit data 9. Print graph if possible 10. Calculate PV for each depth 11. Find the mean deviation of the PV values. o Find the mean of the PV values o Find the deviation of each PV value from the mean by subtraction o Find the mean of the deviations. Conclusion 1. Discuss the overall purpose of this lab. 2. Briefly describe the equipment and general procedure. 3. Discuss and evaluate your results, including the shapes of the graphs and the mean deviation. 4. Make a comprehensive list of sources of measurement error. Data Page Barometric Pressure, Po = ______________________mm Hg Length of small tube, Lo = ______________________mm D(mm) H(mm) 100 200 300 400 500 600 700 800 Curve Fit Data: m = _____________ b = _____________ Correlation: ______________ Gas “Volume” Lo - H Pressure = Po + (D – H)/13.55 1/V