Unexpected-adverse-event-report-form-March-2016.docx (43.9 KB)
advertisement

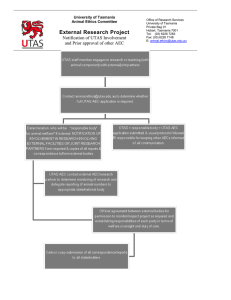
Research Services University of Tasmania Private Bag 01 Hobart, Tasmania 7001 Tel: (03) 6226 7283 Fax: (03) 6226 7148 University of Tasmania Animal Ethics Committee UNEXPECTED ADVERSE EVENT REPORTING FORM animal.ethics@utas.edu.au An unexpected adverse event impacts on the welfare of the animal and was not considered in the initial application. It must be immediately reported to the University Veterinarian and the Animal Ethics Committee 1 - Details of the project (Please type in the boxes provided). Project No: Title of approved project: Responsible investigator: Original approval date of the project: 2 - Please re-state the aims of the project in plain English: 3 – Date and time of the unexpected adverse event: Date of the event: Enter date. Date of notification to the University Veterinarian Enter date. Time of the event: Time of notification to the University Veterinarian Enter Time Enter Time 4 - Where did the event occur? Building/Site Room/Area Other (e.g. location of fieldwork) Did the event occur at a UTAS facility? Yes / No 5 – Details of the event Briefly outline the event and the circumstances leading up to the event: Probable cause of the event: Impact of the event: (both the immediate impact and any long term impact on research) Animal Ethics Committee Version March 2016 Page 1 of 3 Total number of mortalities or injuries which incurred out of the total population: (include species details) Autopsy details and findings: (including autopsy personnel) Remedial action taken: How has this event been communicated to members of the Research team and others? What procedures/practices need to be modified as a result of the event? (Note: If a modification is necessary, an Application to modify an approved project form must be submitted) Please sign and complete the declaration below, before emailing it to: animal.ethics@utas.edu.au Hard Copies are not required DECLARATION The outlined account represents a true and accurate description of the event as they relate to this approved protocol. Responsible Investigator or Nominee Name of Investigator or Nominee: Signature: ......................................................................... Date: University Veterinarian Name of University Veterinarian: Signature: .......................................................................... Date: Comments: Animal Ethics Committee Version March 2016 Page 2 of 3 (Following table for completion by the Animal Ethics Committee) Cause (tick all that apply) Human Error Equipment/Facility Failure Experimental Procedure (includes adverse drug reactions but not incorrect doses) Uncontrollable event (e.g. adverse weather). Please provide details. Action taken (tick all that apply): Project Halted Retraining of staff/investigators Equipment/Facility Repair Change to SOP Change to Experimental Procedure Other (please detail) Severity Low Medium High Catastrophic Animal Ethics Committee Version March 2016 Page 3 of 3