Help Determining whether an AE is an Unanticipated problem
advertisement

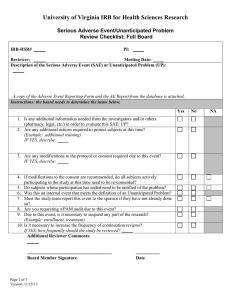
AE Worksheet How to determine whether problems that occur during research are “unanticipated problems” (UPIRSOs) Is this incident, experience, or outcome an adverse event? Definition - An adverse event is any untoward or unfavorable medical occurrence in a human subject, including any abnormal sign (for example, abnormal physical exam or laboratory finding), symptom, or disease, temporally associated with the subject’s participation in the research, whether or not considered related to the subject’s participation in the research. (Encompasses both physical and psychological harms.) No If no, go to the Non-AE Worksheet for determining whether a Non-Adverse Event is an unanticipated problem. Yes If yes, use the three criteria below to determine whether the adverse event is an unanticipated problem (UPRISO). Criterion 1: Is this adverse event UNEXPECTED? Definition - An unexpected adverse event is any adverse event, the nature, severity, or frequency of which is not consistent with either: (1) the known or foreseeable risk of adverse events associated with the procedures involved in the research that are described in (a) the protocol related documents, such as the IRB-approved research protocol, any applicable investigator brochure, and the current IRB-approved informed consent document, and (b) other relevant sources of information, such as product labeling and package inserts; or (2) the expected natural progression of any underlying disease, disorder, or condition of the subject(s) experiencing the adverse event and the subject’s predisposing risk factor profile for the adverse event? To determine whether an adverse event is unexpected, answer the following 3 questions. Yes No Questions Q1. Is the nature of the AE a known or foreseeable risk? Yes No (e.g., liver failure due to diffuse hepatic necrosis occurring in a subject without any underlying liver (go to Q2) This is an disease would be an unexpected adverse event (by Unexpected virtue of its unexpected nature) if the protocolAE related documents and other relevant sources of (skip Q2&3 and information did not identify liver disease as a continue to potential adverse event) Criterion 2) Q2. Is the severity of the AE greater than anticipated? Yes No (e.g., liver failure due to diffuse hepatic necrosis occurring in a subject without any underlying liver This is an (go to Q3) disease would be an unexpected adverse event (by Unexpected virtue of its unexpected greater severity) if the AE protocol-related documents and other relevant (skip Q3 and sources of information only referred to elevated continue to hepatic enzymes or hepatitis as potential adverse Criterion 2) events related to the procedures involved in the research) Q3. Is the frequency of the AE greater than anticipated? Yes No [Note: It may be difficult to determine whether an individual AE is unexpected by virtue of an This is an This is not unexpected higher frequency; this determination Unexpected an Unknown at this time This is not an can only be done through an analysis of appropriate data on all or most subjects. This question can be answered as unknown at the present time, as appropriate] (e.g., liver failure due to diffuse hepatic necrosis in a group of subjects without any underlying liver disease if the occurrence is in a higher percentage of subjects than referenced in the protocol-related documents and other relevant sources of information) AE (continue to Criterion 2) Unexpected AE Unexpected AE at this time Prompt Report Not Needed Prompt Report Not Needed Criterion 2: Is this unexpected adverse event RELATED OR PROBABLY RELATED to participation in research? Definition – There is at least a reasonable possibility that the unexpected adverse event may have been caused by the procedures involved in the research. To determine whether an unexpected adverse event is related or probably related, choose which statement is accurate (you should only select one). The unexpected adverse event is at least partially caused by the procedures involved in the research. The unexpected adverse event is caused solely by either: an underlying disease, disorder, or condition of the subject; or other circumstances unrelated to either the research or any underlying disease, disorder, or condition of the subject. The unexpected AE is related, (continue to Criterion 3) The unexpected AE is not related Prompt Report Not Needed Criterion 3: Does this unexpected adverse event, that is related or probably related to participation in research suggests that the research places subjects or others at a greater risk of harm than was previously known or recognized? This is a three step process Step 1 - Is this unexpected adverse event, that is related or probably related to participation in research, SERIOUS? Yes No Is this an AE that: results in death is life-threatening (places the subject at immediate risk of death from the event as it occurred) results in inpatient hospitalization or prolongation of existing hospitalization results in a persistent or significant disability/incapacity results in a congenital anomaly/birth defect based upon appropriate medical judgment, may jeopardize the subject’s health and may require medical or surgical intervention to prevent one of the other outcomes listed in previous five statements. If you answered yes to If you answered no to ANY, then the AE is ALL, the AE is not serious, serious, (skip Step 2 and (continue to continue to Step 2) Step 3 check 3.a.) Step 2 – Is this not serious, unexpected adverse event that is at least probably related suggest that the research places subjects or others at a greater risk of physical or psychological harm than was previously known or recognized? YES NO If yes, go to Step 3, check 3.b. If no, this AE is not an “unanticipated problem” Prompt Report Not Needed Step 3 – Unanticipated Problems 3a. You should promptly report this serious, unexpected adverse event that is at least probably related to the research as an unanticipated problem because such events always suggest that the research places subjects or others at a greater risk of physical or psychological harm than was previously known or recognized. 3b. You should promptly report this not serious, unexpected adverse event that is at least probably related to the research as an unanticipated problem because it was determined that the event suggests that the research places subjects or others at a greater risk of physical or psychological harm than was previously known or recognized.