Development of a Nile-blue based chemodosimeter for Hg in
advertisement

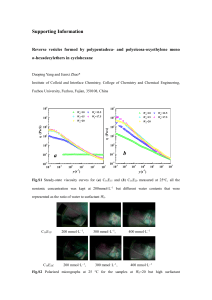
Electronic Supplementary Information For Development of a Nile-blue based chemodosimeter for Hg2+ in aqueous solution and its application in biological imaging Mingming Hu*, Jianhua Yin, Yahong Li and Xiaofang Zhao The Institute of Seawater desalination and Multipurpose Utilization, SOA(Tianjin), No.1 Keyan East Road, Nankai District, Tianjin 300192, P.R. China. Fax: 86-22-87898128; Tel: 86-22-87898127-829; Email: mmhu_sdmu@126.com Synthetic procedures Synthesis of compound 1: 8.7 g 3-diethylaminophenol (52.72 mmol) was dissolved in 30 mL concentrated hydrochloric acid and 10 mL water, dropped with 30 mL aqueous solution of sodium 1 nitrite 4.36 g (63.20 mmol) below 5oC, and the mixture was stirred for 1 h. The yellow solid was washed with 100 mL saturated sodium acetate solution, then recrystallizated with acetone to give red acicular crystal 1 (9.2 mg, yield: 89.2%), mp. 170°C. Synthesis of compound 2: To a solution of N-(1-naphthyl)ethylenediamine (2 g, 7.72 mmol) in CH3CN (50 ml), isothiocyanatobenzene (1.46 ml, 7.72 mmol) and triethylamine (5.38 ml, 38.58 mmol) were added slowly, and then refluxed for 1 h. The reaction mixture was concentrated under reduced pressure and dissolved in CH2Cl2 (100 ml). The organic layer was washed with water (3×300 ml), and dried over anhydrous MgSO4. The solution was evaporated under reduced pressure, the crude product was purified by column chromatography (silica, ethyl acetate/hexane, 1:4) to afford 2.35 g of 2 (95 % yield) as a colorless oil. Synthesis of NBHg-1: To a cold solution of 1 (0.20 mg, 1.11 mmol) in ethanol (2 ml), 2 (0.40, 1.11 mmol), concentrated hydrochloric acid (1 ml) was added. The mixture was refluxed for 4 h and monitored by TLC. The solvent was removed under reduced pressure and the crude mixture was purified by column chromatography (silica, methylene chloride/methanol, 10:1) to give NBHg-1 as a metalescent green solid (0.47 g, 85 %). 1H-NMR(400 MHz, CD3OD), H (ppm): 8.93(d, 1H, J=8 Hz), 8.37(d, 1H, J=8 Hz), 7.92 (t, 2H, J=8 Hz), 7.83(t, 1H, J=8 Hz), 7.31(t, 3H, J=8 Hz), 7.20(m, 4H, J=8 Hz), 6.93(s, 1H), 4.08(t, 2H, J=8 Hz), 3.99(t, 2H, J=8 Hz), 3.72(m, 4H, J=8 Hz), 3.60(m, 1H, J=8 Hz), 1.35(t, 6H, J=8 Hz), 1.18(t, 1H, J=8 Hz); C-NMR(100 MHz, CD3OD), C(ppm):181.83, 158.49, 13 154.42, 151.73, 148.48, 137.66, 131.57, 130.54, 129.44, 128.95, 124.41, 124.14, 123.23, 122.81, 115.57, 95.44, 93.31, 45.69, 44.42, 42.31, 11.57; TOF MS(ES):m/z Calcd for C29H30N5OS+:496.2171, Found: 496.2166. Experimental procedure 2 Figure S1. 1H NMR chart of NBHg-1 in CD3OD (400 MHz). Figure S2. 13C NMR chart of NBHg-1 in CD3OD (100 MHz). 3 Figure S3. TOF-MS of NBHg-1. Calc. C29H30N5OS+: 496.2171; found 496.2166. Figure S4. TOF-MS of the product of NBHg-1 recognition with Hg2+. Calc. C29H28N5O+: 462.2294, found 462.2279. 4