Bhattacharjee_Prussian Blue Analogue.ppt
advertisement

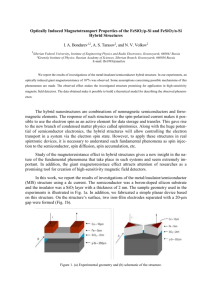
Metal-to-Metal Electron Transfer and Magnetic Interactions in a Mixed-Valence Prussian Blue Analogue A. Bhattacharjee, P. Gütlich et al. Department of Physics, Visva-Bharati University, Santiniketan 731235, India, E-mail: ashis@vbphysics.net.in Department of Chemistry, University of Mainz, 55099, Mainz, Germany, E-mail: guetlich@uni-mainz.de Prussian Blue (PB) Analogue The hexacyanometalate [B(CN)6]x- ions are well known building blocks used for fabrication of the hetero-metal assemblies exhibiting bulk magnetization, where reaction of the [B(CN)6]x- ions with metal ions gives rise to the so-called ‘Prussian Blue’ (PB) analogues - MA[B(CN)6].(solvent) (M = monovalent alkali metal ion, and A, B = diand trivalent transition metal ions). These materials exhibit various magnetic properties depending on their transition metal combinations, e.g., high TC magnet, magnetic pole reversal, spin glass behavior and photo-induced magnetic transition. The alkali-doped analogues are among the most extensively studied recent materials of the Prussian Blue family in regard to photo-induced and pressure-induced metal-to-metal electron transfer and magnetism. For further details see the follwoing references: - M. Verdaguer et al., Coord. Chem. Rev. 190-192 (1999) 1023; - T. Yokoyama, H. Tokoro, S.-i. Ohkoshi, and K. Hashimoto Phys. Rev. B 2002, 66, 184111; - A. Goujon, F. Varret, V. Escax, A. Bleuzen, M. Verdaguer Polyhedron, 2001, 20 (11-14), 1347-1354; - V. Ksenofontov, G. Levchenko, S. Reiman, P. Gütlich, A. Bleuzen, V. Escax, M. Verdaguer, Phys. Rev. B 2003, 68, 024415; - A. Bhattacharjee, S. Saha, S. Koner, V. Ksenofontov, S. Reiman, P. Gütlich, J. Magn. Magn. Mater. 2006, 302, 173-180; - A. Bhattacharjee, S. Saha, S. Koner, Y. Miyazaki J. Magn. Magn. Mater. 2007, 312, 435-442. K0.2MnII.66MnIII1.44[FeII0.2FeIII0.8(CN)6]O0.66(CH3COO)1.32]·7.6H2O Calorimetric study under magnetic field and field dependent magnetization studies of a new PB analogue - K0.2MnII.66MnIII1.44[FeII0.2FeIII0.8(CN)6]O0.66(CH3COO)1.32]·7.6H2O have indicated a ferrimagnetic phase transition around 8 K along with a ferromagnetic phase transition around 2 K. The compound exhibits metamagnetic transition around 3 K observed in the magnetic measurements. Furthermore, the compound exhibits a thermal anomaly around 185 K arising due to a glass transition. 15 3 8 8 kOe -1 7 6 1.5 5 0 1 7 kOe 2 2.0 2.5 3.0 T/K M / NAB 10 Cmag / JK mol -1 -1 Cmag / JK mol -1 9 kOe 6 kOe 5 kOe 4 kOe 3 kOe 1 2 kOe 1 kOe 0.6 kOe 0.2 kOe 11 21 T/K Magnetic Transitions 31 0 0 10 20 T/K Metamagnetic Transition Mössbauer Spectroscopy 100 II Fe 96 III Fe 300 K Mössbauer spectroscopic studies of this compound were done at various temperatures. 92 Rel. Transmission (%) 100 96 The Mössbauer spectra obtained at all the measuring temperatures exhibited the existence of both FeIII and FeII in low spin states. Thus, the compound exists in FeIII (low spin, t2g5, S = ½), FeII (low spin, t2g6, S = 0), MnIII (high spin, t2g3 eg1, S = 2) and MnII (high spin, t2g3 eg2, S = 5/2) mixed valence states. 11 K 92 100 96 92 10 K 100 96 5K 92 100 96 4.2 K 92 -5.0 -2.5 0.0 v /mm s 2.5 -1 5.0 The onset of magnetic ordering of the FeIII low spin species around 5 K is clearly seen by the broadening of the blue signal, which develops to a reasonably well resolved magnetic sextet at 4.2 K. Metal-to-Metal Electron Transfer Mössbauer spectroscopy successfully detects the phenomenon of metal to metal electron transfer between Mn and Fe ions possibly through the 12 [FeIII (t2g5, S = ½) –CN- MnII (t2g3 eg2, S =5/2)] to [FeII (t2g6, S = 0)–CN- MnIII (t2g3 eg1, S = 2)] process. III Fe /Fe II 9 At temperatures above the magnetic transition the compound exists as a mixture of 6 [FeIII(S = ½) –CN- MnII(S = 5/2)] and 3 0 100 200 300 T/K Temperature dependence of the population ratio of FeIII and FeII low spin species obtained from Mössbauer spectroscopy [FeII (S = 0) –CN- MnIII (S = 2)] states, whereas below the magnetic transition the former state predominates. Glass Transition 3.2 12 3.0 II 9 III Fe /Fe -2 (Cp / T) / JK mol -1 3.1 2.9 6 2.8 2.7 120 140 160 180 200 220 T/K From Calorimetry 240 3 0 100 200 300 T/K From Mössbauer spectroscopy A glass transition at 194 K has been observed in the heat capacity study due to freezing of the orientational motion of the H2O molecules present. This phenomenon is reflected in the temperature dependence of the estimated FeIII and FeII concentrations in the present material obtained through Mössbauer spectroscopy. Mössbauer spectroscopy being extremely sensitive to lattice dynamics is able to detect the effect of the glass transition due to the freezing of the orientational motion of the H2O molecules inducing non-rigid / dynamic character in the lattice on and around the glass transition temperature. Bhattacharjee, et al., J. Magn. Magn. Mater. 302 (2006) 173; J. Magn. Magn. Mater. 312 (2007) 435