Meningococcal Conjugate Vaccine Protocol for Use During the Midwestern Outbreak - July 2015 (Word)
advertisement

Meningococcal Conjugate Vaccine Protocol for Use During the
Midwestern Outbreak - July 2015
1. CONDITION FOR PROTOCOL: To reduce incidence of morbidity and mortality of Neisseria meningitidis disease during the
Midwestern outbreak caused by serotype C in persons at risk for meningitis.
2. POLICY OF PROTOCOL: The [nurse/pharmacist] will implement this protocol for MCV4 vaccination during the Midwestern
outbreak.
3. CONDITION-SPECIFIC CRITERIA AND PRESCRIBED ACTIONS:
For persons adopting these protocols: The criteria below lists indications, contraindications, and precautions that are
necessary to implement the vaccine protocol. The prescribed actions include examples shown in [ ] but may not suit your
institution’s clinical situation and may not include all possible actions. A licensed prescriber must review the criteria and
actions and determine the appropriate action to be prescribed. (Delete this paragraph before version is signed.)
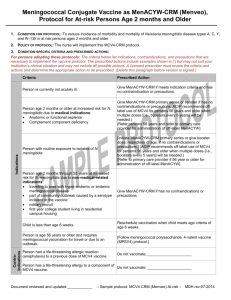
Criteria
Prescribed Action
Precaution
Contraindication
Indication
Currently healthy person with or without HIV
Give meningococcal vaccine per prescription and indication
infection who has requested vaccination due to the
table if meets remaining criteria.
current outbreak.
Person is 56 years of age or older
1) Give MCV4 vaccine off-label per MDH recommendation
and guidelines and per prescription and indication table if
meets remaining criteria.
OR
2)Do not give, refer person to clinic that provides off-label
MCV4 vaccine.
Person has previous history of meningococcal
disease.
Give meningococcal vaccine per prescription and indication
table if meets remaining criteria.
Person previously received MCV4 or MPSV4.
Give meningococcal vaccine, if indicated per indication
table and if meets remaining criteria.
Person had a life-threatening allergic reaction
(anaphylaxis) to a previous dose of MCV4 vaccine.
Do not vaccinate; _____________________
Person has a life-threatening allergy to a
component of MCV4 vaccine.
Do not vaccinate; _____________________
Person has a mild illness defined as temperature
less than ____°F/°C with symptoms such as: {to be Not a contraindication; give MCV4.
determined by medical prescriber}
Person has a moderate to severe illness defined as
[Refer to health care provider for evaluation of symptoms
temperature ____°F/°C or higher with symptoms
and determination of whether to vaccinate.]
such as: {to be determined by medical prescriber}
4. PRESCRIPTION:
For quadrivalent conjugate meningococcal A, C, Y, W-135 (MCV4) indications, give either:
Menactra (MenACYW-D) 0.5 ml, IM.
Or
Menveo (MenACYW-CRM) 0.5 ml, IM.
Document reviewed & updated:____________
– Sample: Meningococcal vaccination in special situation –
MDH guidance: 07/2015
Give vaccine according to following indication table.
Previous vaccination
status
Never vaccinated
Received 1 dose in the
last 5 years
Not HIV Infected
HIV infected
1 dose MCV4
(Menactra or Menveo)
2 doses MCV4,
8 weeks apart
No dose necessary
1 dose MCV4
Received 2 doses in the
past 5 years
No dose necessary
Received 1 or more
doses more than 5
years ago
1 dose MCV4
5. MEDICAL EMERGENCY OR ANAPHYLAXIS: [Depending on clinic staffing, include one of the two options below.]
In the event of a medical emergency related to the administration of a vaccine. RN will apply protocols as described in
____________________________________________________________________________________________.
In the event of an onset of symptoms of anaphylaxis including:
o rash
o itchiness of throat
o difficulty breathing
o bodily collapse
o swollen tongue or throat
LPN or unlicensed assistive personnel (MA) will immediately contact the RN in order to implement the
____________________________________________________________________________________________.
6. QUESTIONS OR CONCERNS:
In the event of questions or concerns, call Dr. ____________________________at _____________________________.
This protocol shall remain in effect for all patients of ______________________________until rescinded or until
_____________________________________.
Name of prescriber: _______________________________________________________________________________
Signature: ________________________________________________________________________________________
Date: ___________________________
Document reviewed & updated:____________
– Sample: Meningococcal vaccination in special situation –
MDH guidance: 07/2015