Removal performance and mechanism for trace phenolic endocrine disruptors by... Andre Rodrigues dos Reis and Yutaka Sakakibara
advertisement
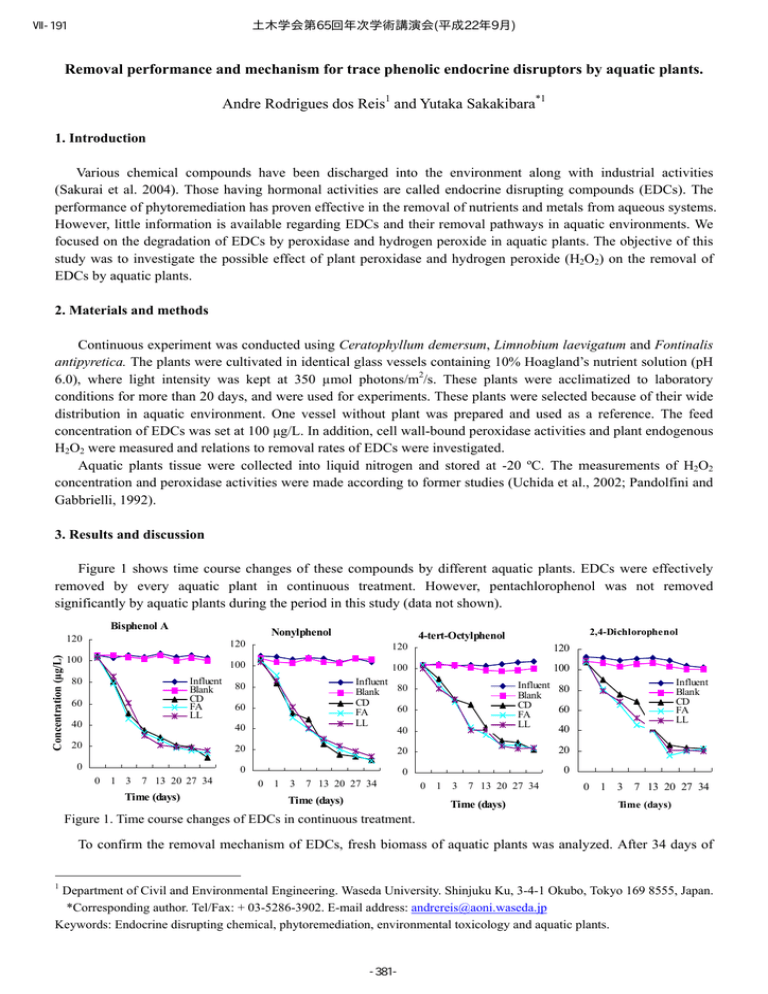
土木学会第65回年次学術講演会(平成22年9月) Ⅶ-191 Removal performance and mechanism for trace phenolic endocrine disruptors by aquatic plants. Andre Rodrigues dos Reis1 and Yutaka Sakakibara*1 1. Introduction Various chemical compounds have been discharged into the environment along with industrial activities (Sakurai et al. 2004). Those having hormonal activities are called endocrine disrupting compounds (EDCs). The performance of phytoremediation has proven effective in the removal of nutrients and metals from aqueous systems. However, little information is available regarding EDCs and their removal pathways in aquatic environments. We focused on the degradation of EDCs by peroxidase and hydrogen peroxide in aquatic plants. The objective of this study was to investigate the possible effect of plant peroxidase and hydrogen peroxide (H2O2) on the removal of EDCs by aquatic plants. 2. Materials and methods Continuous experiment was conducted using Ceratophyllum demersum, Limnobium laevigatum and Fontinalis antipyretica. The plants were cultivated in identical glass vessels containing 10% Hoagland’s nutrient solution (pH 6.0), where light intensity was kept at 350 µmol photons/m2/s. These plants were acclimatized to laboratory conditions for more than 20 days, and were used for experiments. These plants were selected because of their wide distribution in aquatic environment. One vessel without plant was prepared and used as a reference. The feed concentration of EDCs was set at 100 µg/L. In addition, cell wall-bound peroxidase activities and plant endogenous H2O2 were measured and relations to removal rates of EDCs were investigated. Aquatic plants tissue were collected into liquid nitrogen and stored at -20 ºC. The measurements of H2O2 concentration and peroxidase activities were made according to former studies (Uchida et al., 2002; Pandolfini and Gabbrielli, 1992). 3. Results and discussion Figure 1 shows time course changes of these compounds by different aquatic plants. EDCs were effectively removed by every aquatic plant in continuous treatment. However, pentachlorophenol was not removed significantly by aquatic plants during the period in this study (data not shown). Nonylphenol 80 Influent Blank CD FA LL 60 40 20 0 120 100 40 100 Influent 80 Blank CD 60 FA LL 40 20 20 Concentration (µg/L) Concentration (µg/L) Concentration (µg/L) 100 80 60 0 0 1 3 7 13 20 27 34 Time (days) 2,4-Dichlorophenol 4-tert-Octylphenol 120 120 Concentration (µg/L) Bisphenol A 120 1 3 7 13 20 27 34 Time (days) Influent Blank CD FA LL 40 20 0 0 0 100 Influent 80 Blank CD 60 FA LL 0 1 3 7 13 20 27 34 Time (days) 0 1 3 7 13 20 27 34 Time (days) Figure 1. Time course changes of EDCs in continuous treatment. To confirm the removal mechanism of EDCs, fresh biomass of aquatic plants was analyzed. After 34 days of 1 Department of Civil and Environmental Engineering. Waseda University. Shinjuku Ku, 3-4-1 Okubo, Tokyo 169 8555, Japan. *Corresponding author. Tel/Fax: + 03-5286-3902. E-mail address: andrereis@aoni.waseda.jp Keywords: Endocrine disrupting chemical, phytoremediation, environmental toxicology and aquatic plants. -381- 土木学会第65回年次学術講演会(平成22年9月) Ⅶ-191 2 treatment, EDCs was scarcely detected in the aquatic plant (data not shown). This result indicates that aquatic plants have no ability to absorb EDCs or accumulate it inside. These results suggest that these plants may possess enzymes such as peroxidases that specifically metabolize EDCs containing a phenolic group. According to Figure 1, the removal of EDCs did not occur in the absence of the plants (blank). Figure 2 shows time course changes of plant peroxidase activity, demonstrating the enzyme activity differed significantly among the plants. C. demersum had relatively larger soluble and ionically cell wall-bound peroxidase activities (IPO). On the other hand, F. antipyretica increased the activities of IPO and covalently cell wall-bound peroxidase (CPO) during the initial phase of experiment. Peroxidases are extremely versatile enzymes and best known for their ability to utilize H2O2 in the oxidation of phenols. Endogenous levels of H2O2 decreased with time. L. laevigatum and F. antipyretica showed relatively higher levels of H2O2 (Figure 2). In the presence of toxic substance or under stressed conditions, plants may increase the production of reactive oxygen species such as superoxide and H2O2, as a result of aerobic metabolism. It was thought that H2O2 might be consumed by a complex enzymatic antioxidant system including catalase and peroxidase. C. demersum L. laevigatum F. antipyretica SPO 180 IPO 160 CPO 140 120 100 80 60 40 20 0 200 150 100 50 0 0 5 10 20 Time (days) 30 0 5 10 20 700 H2O 2 (nmol/g FW) △ ABS470nm/mg prot 250 12 10 8 6 4 600 500 400 300 200 2 100 0 0 30 0 5 10 20 Time (days) Time (days) CD FA LL 800 14 △ ABS470nm/mg prot △ ABS470nm/mg prot 300 30 0 5 10 20 30 Time (days) Figure 2. Time course changes of peroxidase activity and hydrogen peroxide, where: Soluble peroxidase activity (SPO); Ionically cell wall-bound peroxidase activity (IPO); Covalently cell wall-bound peroxidase activity (CPO), Ceratophyllum demersum (CD), Limnobium laevigatum (LL) and Fontinalis antipyretica (FA), respectively. 4. Conclusion Experimental results demonstrated EDCs were effectively removed by different types of aquatic plants, except pentachlorophenol. In addition, it was thought the removal rate might be affected by endogenous H2O2 concentration and peroxidase activity. Further studies will be need to analyze the ability of aquatic plants to remove EDCs under different loading conditions. Acknowledgment This study was supported in part by a Research Found of JST Project on “Development for water reuse technology in tropical region”. References Pandolfini T., Gabrielli R. (1992). Nickel toxicity and peroxidase activity in seedling of Triticum aestrivum L. Plant Cell Environmental 15, 719-725. Sakurai A., Toyoda S., Masuda M., Sakakibara M. (2004). Removal of bisphenol A by peroxidase-catalyzed reaction using culture broth of Coprinus cinereus. Journal of Chemical Engineering of Japan, 37(2), 137-142. Uchida A., Jagendorf A.T., Hibino T., Takabe T. (2002). Effects of hydrogen peroxide and nitric oxide on both salt and heat stress tolerance in rice. Plant Science 163, 515-523. -382-