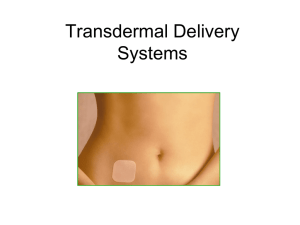
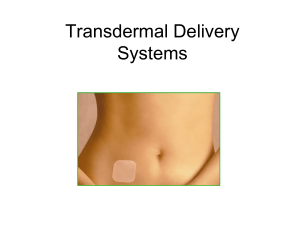
Medical Converting and Packaging for Transdermal Delivery
advertisement

cosmeceuticals Medical Converting and Packaging for Transdermal Delivery of Cosmeceuticals, Nutraceuticals, and Aromatherapy The market for cosmeceuticals, nutraceuticals, and aromatherapy is rapidly expanding as the ability to deliver these items using transdermal patches is further refined. From patches for anti-aging products, to essential oils for homeopathic remedies, to vitamins and nutrients for soldiers in the battlefield, transdermal patch technology has blossomed since the introduction of the first nicotine patches decades ago. Cosmeceuticals, Nutraceuticals, and Aromatherapy Transdermal delivery became more popular in areas where design and manufacturing complexity and regulatory monitoring were less intense: cosmeceuticals, nutraceuticals, and aromatherapy. Transdermal delivery technology is becoming popular for cosmeceuticals, nutraceuticals, and aromatherapies. Cosmeceuticals What are cosmeceuticals? Most often, a cosmeceutical delivers some active ingredient through the skin, while cosmetics are more “topical” in nature. Some industry experts would like to define them as products that don’t require a prescription, but are only available from professionals such as doctors, aestheticians, healthcare professionals, and technicians. Today’s most popular cosmeceuticals include: • Teeth whiteners • Lip protection • Skincare, especially anti-aging products • Haircare • Scarless wound healing, or anti-scarring treatments www.fabrico.com Backing Nutraceuticals Nutraceuticals can include everything from nutritional supplements to complete emergency nutritional delivery systems for those without access to meals for a period of time, for example soldiers in the field, firefighters, miners, and astronauts. Drug/Adhesive Release Liner Cleavage Load Dietary and nutritional supplements benefit from transdermal delivery: • Instant and continuous delivery to the blood stream; • Time-release delivery; • Delivery of the optimal dosage strength; • No breakdown in the harsh, acidic digestive system; • Convenient dosage. Aromatherapy Aromatherapy has become increasingly popular for: • Relaxation and stress relief; • Increased circulation to warm muscles; • Mood enhancements. The use of a transdermal delivery patch can speed delivery of essential oils, allow for greater effectiveness, as in applications where magnesium is delivered using a thermal patch, and deliver therapeutic benefits in conjunction with other homeopathic treatments. Elements in Patch Designs Transdermal patch designs contain a liner, formula/drug, adhesive, membrane, and backing: • Liner – protects the patch during storage and is removed prior to use; • Formula – supplement, oil, or medication in direct contact with the liner; • Adhesive – keeps the components of the patch together, keeps the patch adhered to the skin and, in new designs, may include the formula/drug; • Membrane – in some designs, it controls the release of the formula/drug from the reservoir; • Backing – protects the patch from the environment. Drug-in-adhesive transdermal patch designs contain a liner, adhesive, and backing. Types of Transdermal Patch Designs The basic functional requirements for any transdermal delivery system include adhesion to the skin, both the initial tack and the duration, delivery of the drug, and eventual removal from the skin without causing unnecessary irritation. A transdermal patch design will meet these basic functional requirements using a custom combination of components. There are several types of transdermal patch designs: reservoir, matrix, drug-in-adhesive (DIA), and multi-laminate DIA. Patch design must consider the properties of the drug, cosmeceutical, nutraceutical, or aromatherapy, the desired delivery profile, and the target patient group. The reservoir type patch contains the drug solution in a liquid reservoir compartment, separated from a release liner by a semi-permeable membrane and an adhesive. The membrane controls the timed release of the liquid drug. Closely related, the matrix design has a drug that is a semi-solid. In the matrix design, there is no membrane layer. Drug-in-adhesive (DIA) systems add the drug directly to the adhesive that contacts the skin. DIA designs can be single layer or multi-laminate designs that may add a membrane between two distinct DIA layers, or multiple DIA layers under a single backing. The Role of Adhesives in Transdermal Patches Selection of the correct adhesive for a transdermal patch is critical. It affects both drug delivery and the adhesion of the patch to the skin for the required time period and, ultimately, its easy and pain-free removal when the therapy is completed. The right adhesive will work with the formula/drug, the patient’s skin, and body movements that occur during normal wear. Custom formulations are often required to meet the specifics for a transdermal patch. Acrylic, acrylic-rubber hybrids, polyisobutylene, and styrenic rubber solutions are used for their good adhesion and ability to control the rate at which the formula/drug enters the body. New polymer adhesives are also being modified to improve solubility and drug diffusion while keeping favorable adhesive and cohesive properties. These polymers include hydrophilic pressure-sensitive adhesives (PSAs) that can be applied wet and still provide good adhesion. Other types of polymer adhesives include synthetic polymers, such as poly acrylic acid (PAA) and poly methyl acrylate derivatives, as well as hyaluronic acid and chitosan. Copolymerization of different polymers is often used to achieve the correct balance of drug delivery, adhesion, and release properties. Putting the Package Together with the Help of a Medical Converter Fabrico helps OEMs and contract manufacturers with everything from initial patch design to selection of the appropriate adhesive formulation, materials for the backing and release liner, laminating the materials and adhesives together, selecting the appropriate cutting technique, and assembling and kitting the final transdermal patch product. The choice of backing materials influences the delivery profile, adhesion, and wearability. The backing might include the lamination of several layers of materials such as polyethylene and ethyl vinyl acetate. The backing protects and contains the drug/formulation, prevents the unwanted absorption of the drug before application, and controls the evaporation of moisture through the patch. The release liner must also be carefully Fabrico offers three Class 10,000 clean rooms, as well as ISO 13485 certification. selected to protect the adhesive, which may contain the formulation, until application without interacting with other components in the design. Fabrico can also suggest the best packaging for the transdermal patch for shipment and storage until use. Fabrico provides slitting, layering, laminating, precision die-cutting, and packaging of the finished product. The converting process might entail clean room production; for example, Fabrico offers three Class 10,000 clean rooms in its facilities, as well as ISO 13485 certification. Fabrico can select from servo driven rotary die-cutting, CNC die-cutting, laser cutting, and water jet cutting to meet the complex specifications of medical components. For example, a servo driven rotary die-cutter can maintain tight tolerances ranging from 0.015” to +/-0.005” at speeds up to 500 fpm, and is ideal for the complex, multi-layer die-cutting, and lamination that a transdermal patch might require. For complex foam tape converting, water jet technology provides clean edges with no distortion. Laser cutting, kiss-cutting, slitting, and laminating can also be used in converting for medical and cosmeceutical applications. About Fabrico Fabrico’s materials converting capabilities include: custom design solutions for applications that require slitting, laminating, and die-cutting. Laser cutting, kiss-cutting, and water jet cutting are available depending on the application and materials being used. Fabrico has a fully-equipped in-house test lab to ensure that materials and adhesives meet design specifications. Fabrico has a fully equipped test laboratory to ensure that customer materials meet designed-in specifications before they move to the factory floor, often eliminating the need to test materials at the customer’s facility. The test lab offers: • Accurate and precise part dimension measurement and verification; • Adhesive/release liner testing to determine converting properties and high speed application characteristics; • Material strength measurement to ensure that material meets application requirements; • Static shear testing to measure the cohesive strength of the adhesive to withstand a fixed load over time; • Material weight measurement to determine adhesive coating weight; • Microscopic imaging to determine differences between adhesive and material over time. Fabrico Headquarters 4175 Royal Drive, Suite 800, Kennesaw, GA 30144 Phone: 678-202-2700 | Fax: 678-202-2702 Toll Free: 800-351-8273 | E-mail: info@fabrico.com With more than 30 years of materials experience, Fabrico engineers also understand the impact of material selection on the overall manufacturing process, and design material systems that optimize production efficiency and improve overall cost-effectiveness. Material Partners Fabrico has strategic relationships with world-class materials and adhesives suppliers, such as 3M, Adhesives Research, Dow Chemical, NuSil Technology, Polymer Technology Corporation, and Saint-Gobain. Fabrico helps its customers in selecting the best materials and adhesives for the application. Fabrico’s extensive network of suppliers helps expedite materials and adhesives sourcing. All critical material and adhesive properties are considered in any Fabrico project. www.fabrico.com Fabrico is a trademark of EIS, Inc.; 3M is a trademark of the 3M Company. Loctite is a Registered Trademark of Henkel AG & Co. KGaA.