Introduction
advertisement

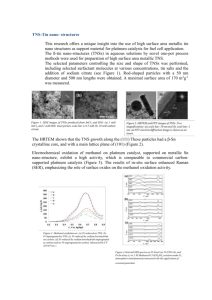
Vimal Parekh*, Ian Lennon,2 James Ramsden2 and Martin Wills1 1. Department of Chemistry, The University of Warwick, Coventry, CV4 7AL, UK 2. CPS Chirotech, Dr Reddy’s Laboratories, Unit 162 Cambridge Science Park, Cambridge, CB4 OGH, UK Introduction Synthesis of the catalyst Asymmetric reduction of C=N bonds represents a powerful method for the asymmetric formation of chiral amines.1 Whilst many methods exist for the asymmetric reduction of isolated C=N groups, their reduction when they are a part of an aromatic ring represents a more challenging objective. Tetrahydroquinoline derivatives have attracted considerable attention owing to their importance as: The studies carried out showed that the 4C ‘tethered’ link is required for rapid conversion and functionality on the aromatic ring might be essential for a high ee, which was why the catalyst shown in Scheme 3 was synthesized. -Synthetic intermediates for drugs, -Agrochemicals, and -Dyes2 A number of reports on the pressure hydrogenation of quinolines have been published3 mostly using Ir based catalysts. Reductions using Ru based catalysts have also been reported using pressure hydrogenation, but Ir based catalysts are by far more active for quinoline reductions. My poster presentation will describe the use of asymmetric transfer hydrogenation (ATH) of quinolines using tethered and untethered Ru (II) catalysts. Scheme 1. General scheme for the asymmetric transfer hydrogenation of quinolines. Results and Discussion Catalyst (9) was tested out with the conditions used previously (Table 3), and the reaction proved to be unsuccessful as only 29% conversion was obtained after 24hrs, with an ee of 42%. In this project our aim was to carry out ATH of quinolines using ‘tethered’ and untethered Ru (II) catalysts. The results obtained from preliminary studies showed that the 4C ‘tethered’ catalyst (7) was far more active than the untethered catalyst (3) for the reduction of quinolines, which was why optimization of conditions to give high e.e. and conversion was carried out using catalyst (7) as a starting point along with the least bulkier quinoline, 2-methylquinoline (1). (Scheme 1) Scheme 3. Shows the reaction scheme for the formation of the 4C ‘tethered’ catalyst (9) with added functionality on the aromatic ring about the Ru. Reductions using different solvents ATH of quinolines was carried out using different solvents, and from the results obtained it was quite clear that overall methanol gave the best conversion and ee. Entry Solvent Temp (°C) Catalyst HCO2H: Et3N ratio Substrate: catalyst ratio Time (hrs) Conversion (%) Enantiomeric excess (%) Configuration 1 Methanol 28 (7) 5:2 400:1 24 96 46 R 2 Acetonitrile 28 (7) 5:2 400:1 24 79 36 R 3 Water 28 (7) 5:2 400:1 24 23 32 R 4 Ethanol 28 (7) 5:2 400:1 24 96 37 R 5 Dichloromethane 28 (7) 5:2 400:1 24 92 25 R 6 Diethyl ether 28 (7) 5:2 400:1 24 98 17 R 7 Acetone 28 (7) 5:2 400:1 24 8 8 R 8 Toluene 28 (7) 5:2 400:1 24 73 22 R 9 2-Propanol 28 (7) 5:2 400:1 24 98 31 R 10 Ethyl acetate 28 (7) 5:2 400:1 24 74 18 R Reductions using different Ru (II) catalysts The graph clearly shows that the 4C ‘tethered’ catalyst (7) proves to be the best Ru (II) catalyst for ATH of quinolines (Figure 2). Figure 2. Shows the conversion vs time at 60°C for different catalysts. Reductions carried out on various quinoline substrates using catalyst (7) and (10) Table 1. Shows the reduction of 2-methylquinoline using different solvents. Reductions carried out at different temperatures Having determined the best solvent, the next task was to see whether changing the temperature had any effect on the conversion and ee. Entry Solvent Temp (°C) Catalyst HCO2H: Et3N ratio Substrate: catalyst ratio Time (hrs) Conversion (%) Enantiomeric excess (%) Configuration 1 Methanol 28 (7) 5:2 400:1 24 96 46 R 2 Methanol 40 (7) 5:2 400:1 24 94 44 R 3 Methanol 50 (7) 5:2 400:1 24 94 43 R 4 Methanol 60 (7) 5:2 400:1 24 96 43 R Table 2. Shows the reduction of 2-methylquinoline at different temperatures. The results clearly show that the fastest conversion was obtained at 60°C with only a drop of 3% ee. The next task was to carry out ATH reductions at 60°C using both untethered and ‘tethered’ Ru (II) catalysts. Reductions using different Ru (II) catalysts Figure 1. Shows the different Ru (II) and Rh (III) catalysts used. 1 2 3 4 5 6 7 Methanol Methanol Methanol Methanol Methanol Methanol Methanol Temp (°C) 60 60 60 60 60 60 60 Catalyst (7) dimer (3) monomer (4) monomer (8) dimer (6) dimer (5) monomer (6) monomer HCO2H: Et3N ratio 5:2 5:2 5:2 5:2 5:2 5:2 5:2 Substrate: catalyst ratio 400:1 200:1 200:1 400:1 400:1 200:1 200:1 Imine Solvent 1 12 13 14 15 16 17 18 19 Methanol Methanol Methanol Methanol Methanol Methanol Methanol Dichloromethane Methanol Temp (°C) 28 28 28 28 28 28 28 28 28 HCO2H: Et3N ratio 5:2 5:2 5:2 5:2 5:2 5:2 5:2 5:2 5:2 Substrate: catalyst ratio 400:1 400:1 400:1 400:1 400:1 400:1 400:1 400:1 400:1 Time (hrs) 24 168 48 30 144 144 48 48 48 Conversion (%) 96 68 57 95 94 93 90 86 93 Enantiomeric excess (%) 46 73 0 41 42 41 50 47 67 Configuration R S R R R R R R Table 4. Shows the reduction of substrates (1, 12-19) using Ru (II) catalyst (7). ATH of quinolines was carried out using both untethered and ‘tethered’ catalysts (Figure 1), and results obtained clearly showed that the 4C ‘tethered’ link (7) is important for the rapid conversion of quinoline to tetrahydroquinolines, and functionality on the aromatic ring above the Ru on the untethered catalyst (3) is vital for obtaining a high ee. Entry Solvent Figure 3. Shows the different quinoline substrates used for ATH. The reduction of a series of further quinolines, 12-19 (Figure 3) was examined using tethered catalyst 7 (Table 4). As this proved to be the most successful catalyst for conversion of quinolines to tetrahydroquinolines. Of these, the phenyl-substituted substrate 12 was reduced in the highest ee of the series, whilst the tBu derivative 13 was successfully reduced but only in racemic form. Other isoquinolines were reduced in high conversion but only moderategood ee. Time (hrs) 24 24 24 24 24 24 24 Conversion (%) 96 17 66 87 62 27 59 Enantiomeric Configuration excess (%) 43 R 80 R 29 R 44 R 43 S 68 R 43 R Table 3. Shows the reduction of 2-methylquinoline at 60°C using different catalysts. References 1. (a) Noyori, R. Adv. Synth. Catal., 2003, 345, 15-32. (b) Noyori, R.; Sandoval, C. A.; Muniz, K.; Ohkuma, T. Phil. Trans. R. Soc. A 2005, 363, 901-912. (c) Noyori, R.; Kitamura, M.; Ohkuma, T. Proc. Nat. Acad. Sci. 2004, 101, 5356-5362. (d) Noyori, R.; Ohkuma, T. Angew. Chem. Int. Ed. 2006, 40, 40-73. 2. Katritzky, A. R.; Rachwal, S.; Rachwal, B. Tetrahedron 1996, 52, 15031, and references cited therein. 3. (a) Xu, L.; Lam, K. H.; Ji, J.; Wu, J.; Fan, Q.-H.; Lo, W.-H. Chem. Commun. 2005, 13901392. (b) Zhou, H.; Li, Z.; Wang, Z.; Wang, T.; Xu, L.; He, Y.; Fan, Q.-H.; Pan, J.; Gu, L.; Chan, A. S. C. Angew. Chem. Int. Edn. 2008, 47, 8464-8467. 4. Hayes, A. M.; Morris, D. J.; Clarkson, G. J.; Wills, M. J. Am. Chem. Soc. 2005, 127, 7318. 5. Parekh, V.; Ramsden, J. A.; Wills, M. Tetrahedron: Asymmetry, 2010, 21, 1549-1556. Better results, in terms of enantioselectivity, in several cases exceeding 90% ee, were achieved using the rhodium tethered catalyst 10, which has previous been used for ketone and imine reduction (Table 5). Using 0.5 mol% of 10, the reactions did not go to full conversion after 48 hours, although the use of a higher loading (2 mol%)** of catalyst increased the conversions in most cases. Further work is required to optimize the reductions by these catalysts. Imine Solvent 1 12 13 14 15 16 17 18 19 Methanol Methanol Methanol Methanol Methanol Methanol Methanol Dichloromethane Methanol Temp (°C) 28 28 28 28 28 28 28 28 28 HCO2H: Et3N ratio 5:2 5:2 5:2 5:2 5:2 5:2 5:2 5:2 5:2 Substrate: catalyst ratio 200:1 200:1 200:1 200:1 200:1 200:1 200:1 200:1 200:1 Time (hrs) 24 48 48 48 48 48 48 48 48 Conversion (%)** 68 (85) 30 (35) 16 (43) 67 (76) 65 (73) 64 (76) 57 (65) 30 (29) 58 (69) Enantiomeric excess (%) 93 86 0 91 90 92 93 81 94 Configuration R S R R R R R R Table 5. Shows the reduction ofsubstrates (1, 12-19) using Rh (III) catalyst (10). Conclusion In conclusion, we have demonstated that tethered Ru(II) and Rh(III) complexes are effective catalysts for the ATH of substituted isoquinolines, which are generally regarded as challenging substrates for this application. To the best of our knowledge, this is the first report5 of the use of such catalysts in a solution of formic acid/triethylamine/methanol. As has been observed in ketone reduction, the increased reactivity of tethered complexes over the untethered ones appears to be key to their capacity to work as effective catalysts in this application. Acknowledgements I would like to thank Martin Wills and the Wills group for their support and encouragement during this project. I would also like to thank my industrial supervisors James Ramsden and Ian Lennon and the EPSRC and Dr Reddy’s for the financial support.