Memory of chirality
advertisement

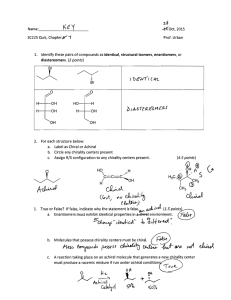
Memory of chirality Baran Group Meeting 6/21/2008 Florina Voica The concept of memory of chirality describes a phenomenon in which the "the chirality of a starting material having a chiral sp3 carbon is preserved in the reaction product even though the reaction proceeds at the chiral carbon as a reaction center through reactive intermediates such as carbanions, singlet monoradicals, biradicals, or carbenium ions". OL, 2002, 4, 1875. We all know that: Requirements for memory of chirality: Y X R1 R2 R2 R1 H chiral Y- R1 H R1 achiral intermediate + R2 - enantioselective formation of a conformationally chiral intermediate from a chiral starting material - slow rate of racemization of the conformationally chiral intermediate - enhanced reactivity of the chiral intermediate toward product formation R2 H H Y racemic Furthermore: H Ph (R)-A-H H NH2 Ph CO2H static chirality H very slow CO2H achiral H Ph CO2H H chirality transfer chirality transfer base fast (M)-Avery slow ΔG‡ (P)-AHO2C Ph H H gauche conformers with transient chirality (dynamic chirality) Therefore, we can hypothesize that upon erosion of an unique chiral center, a non-racemic product mixture may arise from a "chiral" intermediate. MeI fast (R)-A-Me very slow (S)-A-Me What would be the ideal characteristics of a conformationally chiral intermediate in order to generate enantioenriched products? No limitations on the type of the intermediate as long as it resits the forces of racemization: - assuming the racemization process to be an unimolecular phenomenon, the racemization rate constant can be calculated using the Eyring equation - best scenario: at -78 °C, for ΔG‡= 16 kcal/mol, t1/2 = 20h - chiral intermediates containing sp2-sp2 bonds which commonly have barriers to rotation higher that 16 kcal/mol, could be successfully used to achieve memory of chirality. for a review see: Synthesis 2005, 1. 1 Memory of chirality Baran Group Meeting 6/21/2008 Florina Voica 1. Enolate chemistry 1.1 Enantioselective alkylation of ketones Principle: O R1 Application: JACS 1991, 113, 9694 OM base R2 R2 R3 H Ph Ph R3 KO O MeO R1 OEt achiral-no static chirality R1 R2 OM A MO A B R1 R2 B dynamic chirality - axial chirality ii. M M R1 O O R1 R2 R3 R3 R2 KH,18-crown-6 THF, -78 °C to -20 °C EtO OMe 1' O MeI OEt EtO OEt However, under specific conditions: i. 1 Ph Me MeO 93% ee OEt chiral enolate with axial chirality 48% (66% ee) Experimental support for the formation of observed product via the supposed chiral enolate: Ph Ph Ph 1. 2. Me MeO O O OMe MeO KH,18-crown-6 MeO EtO OEt THF, -78 °C to -20 °C OEt EtO OEt OEt 96% ee isolated byproduct (optically active) dynamic chirality - planar chirality then MeI 51% (0% ee) 3. barrier to rotation across the 1,1' bond calculated to be 22. 6 kcal/mol at 21 °C. 1.2 α-Alkylation of amino-acids Principle: Chem-Eur. J. 1998, 4, 373 H R CO2R1 N R3 R2 base Dynamic chirality of the enolate: R1O R R1O MO MO NR2R3 achiral enolate R3 M R N R3 R2 axial chirality R2 M R1O N O R OR1 planar chirality O N R R2 R3 central chirality 2 Memory of chirality Baran Group Meeting 6/21/2008 Applications: * To rule out the formation of aggregates: A. Intermolecular alkylation Bn a) KHMDS CO2Et R H Boc R b) MeI, THF:toluene (1:4) - 78 °C N MOM Entry CO2Et Me Boc Yield (%) R N Boc 2 N N MOM CO2Et 93 N 94 CH2 MeO N Me Me b. MeI 80 80 Bn CO2Et Me + N Boc Boc a. LiTMP, THF -78 °C b. MeI (±) 30 % yield (S) Bn CO2Et Me N Boc Me Bn Me CO2nBu Me (S), 74% ee 26% yield CO2nBu + Bn N Boc + CO2nBu Me N Boc Me (±) 17% yield JACS 1994, 116, 10809 CH2 3 Boc Bn (±) 79 Boc a. LiTMP, THF -78 °C (±) (S) Me MOMO N Me ee (%) 83 CO2nBu + Boc Bn CH2 Bn CO2Et Me N 1 Florina Voica (S), 71% ee 24% yield * To explain the retained streochemistry of the product: MeO CH2 4 88 O O 76 N N Boc H AGIE 2000, 39, 2155 Mechanistic studies: * Possible sources of asymmetric induction: OtBu OEt Ph O Me N O OtBu Li O N EtO Me O N Ph OEt SM/enolate aggregates O Li Ph Me Boc EtO Boc N Ph base EtO N O Ph O CH2OMe MeI CO2tBu R Me Boc CO2Et N MOM chiral enolate product (S) most stable conformer (S) (molecular models ΔGrot(C-N bond) = 16 kcal/mol ( at -78 °C) calculations) AGIE 2000, 39, 2155 Li O OK O Me chiral enolate chiral enolate with central chirality with axial chirality * To certify the implication of an axially chiral enolate: same O CO2Et Bn CO2Et conditions Bn Bn and and Me N H Boc N Boc H BocN Synthesis, 2005, 1368 O Boc Boc O Bn Me BocN O racemic mixtures 3 Memory of chirality Baran Group Meeting 6/21/2008 B. Intramolecular α-alkylation RO2C R H RO2C NH2 R H Mechanistic considerations: X base RO2C R NH NH O O O O R Boc KHMDS or NaHMDS CO2Et N (CH)2-Br Entry 1 2 3 4 5 6 7 DMF -60 °C, 30 min R PhCH2 MeSCH2CH2 Me2CH Me PhCH2 PhCH2 PhCH2 n 3 3 3 3 2 4 5 Florina Voica EtO2C R M+R2N- N Ph Boc N (CH)3-Br OEt Ph O H Br R Br N OK A N Br R retention Br Yield (%) 94 92 78 91 61 84 31 ee (%) 98 (S) 97 94 95 (R) 95 97 83 (S) Ph O H N Boc Br O M+R2N(M = Li) THF, various temp EtO2C R Boc OEt Ph N O O O H Ph M N For the same R substituents, the yields are comparable with the ones observed in the previous study. The ees for are the inverted product in the 81--91% range. JACS 2006, 128, 15394 O OEt N OK ent-A OtBu R JACS 2006, 128, 15394 N inversion N retention Br O R LiTMP CO2Et O Alternatively, CO2Et N OtBu M (CH2)n JACS 2003, 125, 13012 R O OEt (M = Na, K) H NH Boc OtBu O Ph For improved conditions for the intramolecular alkylation with retention of configuration, see JACS 2008, 130, 4153. CO2Et Boc N inversion 4 Memory of chirality Baran Group Meeting 6/21/2008 Florina Voica 1.3 Alkylation of benzodiazepines 2. Radical chemistry 1,4-Benzodiazepin-2-ones are chiral, existing as (M)- or (P)-conformational isomers, 2.1 Memory of chirality due to spinisomers CO2Me Cl R O ΔG‡ R' N O R N Ph R1 O N Ph R1 N Me MeO2C OEt N Ts Cl naphtalene* PhH O (P) OEt N Ts ΔG‡ (kcal/mol) 12.3 18 21.1 > 24 Ph Cl N Me N R1 R1=Me R1=iPr CO2Et N For R or R'≠ H, the pseudoequatorial conformer is favored! 1. 1.2 eq. LDA 6 eq. HMPA, THF, -78 °C, 15min; 1.2 eq. nBuLi, 15 min 2. 10 eq. BnBr, -78°C 3. NH4Cl (aq.) OH MeO2C OH For R=R'=H, the equilibrium is influenced by the nature of R: O hν O R' (M) R1 H Me iPr tBu OH Me Me + N Ts 7% (94% ee) * Proposed mechanism: β Ph Cl N Bn Me O CO2Me OH Me OEt γ N Ts 72% (0% ee) 74% (97% ee) High enantioselectivities observed when other electrophiles (MeI, allylBr, 4-MeC6H4CH2Br) are used JACS 2003, 125, 11482 OL 2005, 7, 5305 (M) racemization N R1 O ΔG‡ MeO2C OH CO2Et N ΔGrot-β bond‡ = 5 kcal/mol B ΔG‡ = 2 kcal/mol CO2Et N O Me Ts HO Me EtO naphtalene is a triplet quencher MeO2C OH MeO2C Me Ts 40% (92% ee) α CO2Me CO2Et Ts (P) N Ts Me ent-B The observed ees are due to a slow racemization compared to the cyclization of the singlet diradical. AGIE 1999, 38, 2586 5 Memory of chirality Baran Group Meeting 6/21/2008 2.2 Memory of chirality due to radical conformers B. Intramolecular radical cyclization 1. oxalyl chloride 2. S OH N , DMAP A. Enantioselective radical quenching S Ph i. DIPDI, DMAP, N OH O ring inversion (racemization) > 97% ee O Ph ii. hν, 1M PhSH, -78 °C ΔG‡ PhSH 5 < ΔG‡ < 10 kcal/mol 93 7 O O Ph HO H Ph BnO2C H O Ph CN THF -78 °C JACS 1998, 120, 5589 JACS 2000, 122, 9386 e-, H+ CO2Bn PhMe, hν, -15 °C Ph H CO2Bn H CO2Bn racemization (chair-boat interconversion) CO2Bn CO2Bn H ΔG‡ = 15.5 kcal/mol H O CO2Bn Proposed mechanism: - Bu2SnH reacts slowly as a proton donor, leading to racemic mixtures - dilute concentrations of tBuSH or PhSH determine a decrease in ees - an increase in the temperature leads to low enantiomeric excess [R ] N 51% yield (68% ee) 92% yield (83% ee) Li (6.4M)/NH3 S H CO2Bn PhSH O CO2H Florina Voica H BnO2C CO2Bn Ph H CO2Bn 80% yield (90% ee) CO2Bn H H SPyr H SPyr CO2Bn H CO2Bn CO2Bn CO2Bn H H OL 2004, 6, 2713 6 Memory of chirality Baran Group Meeting 6/21/2008 3. Memory of chirality via carbocation intermediates C. Umpolung benzylic substitution via chiral organometallic intermediates EtO EtO Me Me Cr(CO)6 Cr(CO)3 THF, 145 °C Me i. LiDBB THF, -78 °C, then RX ii. air, sunlight (99% yield) Me RX: TMSCl BnBr Me2NC(O)Cl Initial result: O R N 72% yield (87% ee) 37% yield (87% ee) 67% yield (86% ee) O 2 F/mol NaOMe (OC)3Cr Me Me (17 VE) O N CO2H O Me R-X O Ph 80% ee O Proposed mechanism: Cr(CO)3 ΔG‡ = 13. 2 kcal/mol + e- OMe N H H Cr(CO)3 R ΔG‡ 69% yield, 39% ee O Pt cathode graphite anode MeOH Ph + e- OEt- O OL 2000, 2, 1689 CO2H O Me OMe N Improvement: N OEt O Pt cathode graphite anode MeOH O Me 2 F/mol NaOMe CO2H Me Mechanism: Florina Voica Nu N O Nu- O 90% N O retention Me H Cr(CO)3 Cr(CO)3 (18 VE) O NuOL 2002, 4, 1875 N O Nu 10% AGIE 1999, 38, 1620 inversion 7