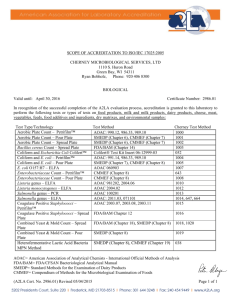
SCOPE OF ACCREDITATION TO ISO/IEC 17025:2005 5205 Quincy Street
advertisement

SCOPE OF ACCREDITATION TO ISO/IEC 17025:2005 EUROFINS MICROBIOLOGY LABORATORIES, INC. – MOUNDS VIEW 5205 Quincy Street Mounds View, MN 55112 Peter Dragasakis Phone: 515 631 9529 BIOLOGICAL Valid To: January 31, 2018 Certificate Number: 3329.02 In recognition of the successful completion of the A2LA evaluation process (including an assessment of the laboratory's compliance with the A2LA Food Testing Program Requirements, containing the 2010 “AOAC International Guidelines for Laboratories Performing Microbiological and Chemical Analyses of Food and Pharmaceuticals”), accreditation is granted to this laboratory to perform the following tests on commodities, foods, dietary supplements, feeds, and pet foods. Test Type/Technology Aerobic Plate Count Aerobic Plate Count (Petrifilm) Coliform & Escherichia coli (Petrifilm) Coliform & E. coli (MPN) Listeria species Dupont Qualicon BAX 24E Listeria species bioMerieux VIDAS Test Kit Listeria species 3M Molecular Detection System Listeria species bioMerieux VIDAS LPT Test Kit Listeria Confirmation - BAM Salmonella Dupont Qualicon BAX Salmonella bioMerieux VIDAS Test Kit Salmonella bioMerieux VIDAS SPT Test Kit Salmonella 3M Molecular Detection System (A2LA Cert. No. 3329.02) 01/04/2016 Test Method AOAC 966.23 BAM Chapter 3 SMEDP 6.020 AOAC 990.12 AOAC 989.10 AOAC 991.14 AOAC 989.10 BAM Chapter 4 AOAC 966.24 AOAC-RI 050903 AOAC 2004.06 AOAC-RI 981202 AOAC-RI 081203 AOAC 2013.10 BAM Chapter 10 AOAC 2003.09 AOAC-RI 100201 AOAC 2011.03 AOAC 2013.01 AOAC-RI 031208 Page 1 of 2 Test Type/Technology Staphylococcus aureus (Petrifilm) Staphylococcus aureus Yeast and Mold Count (A2LA Cert. No. 3329.02) 01/04/2016 Test Method AOAC 2003.07 AOAC 2003.08 AOAC 2003.11 BAM Chapter 12 AOAC 975.55 BAM Chapter 18 Page 2 of 2 Accredited Laboratory A2LA has accredited EUROFINS MICROBIOLOGY LABORATORIES, INC. MOUNDS VIEW Mounds View, MN for technical competence in the field of Biological Testing This laboratory is accredited in accordance with the recognized International Standard ISO/IEC 17025:2005 General requirements for the competence of testing and calibration laboratories. This accreditation demonstrates technical competence for a defined scope and the operation of a laboratory quality management system (refer to joint ISO-ILAC-IAF Communiqué dated 8 January 2009). Presented this 4th day of January 2016. _______________________ Senior Director, Quality and Communications For the Accreditation Council Certificate Number 3329.02 Valid to January 31, 2018 For the tests to which this accreditation applies, please refer to the laboratory’s Biological Scope of Accreditation.