IRB Online Submission 2015
advertisement

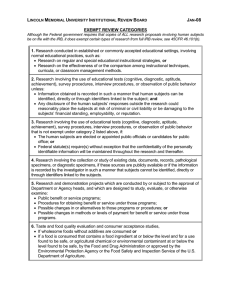
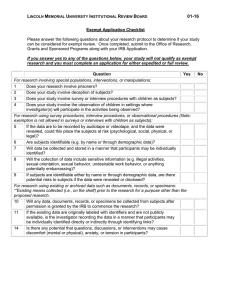
IRB Online Submission 2015 Q1 IRB Online Submission for Review Name) Primary Investigator's Name (Last Name, First Q2 E-mail address of primary investigator Q3 Nature of Project Faculty Research Project Student Research Project Q4 Faculty research supervisor Note: The faculty research supervisor listed must approve your project prior to IRB review and approval. Name Email (name@methodist.edu) Q5 If there is more than one faculty researcher, then enter co-researchers information (Name, e-mail address) Q6 Project Title Q7 In a brief statement, complete the following statement: The purpose of this study is: Q8 Describe the characteristics of the group you are studying (Check all that apply) MU students Individuals under 18 / minors Individuals with mental disabilities Pregnant Women Illegal Behaviors Incarcerated Individuals Fetuses Educationally disadvantaged individuals New drugs or medical devices Economically disadvantaged individuals Abortuses Vertebrae animals Other ____________________ Q9 Does your project involve or use a survey? Yes No Q10 How will you collect your survey data? (check any that apply) Qualtrics Paper E-mail Other ____________________ Q11 If someone begins your survey and does not complete it or does not answer all of the questions, what will you do with the data? Include what has been inputted Delete the data Do not know Q12 Where are you getting the e-mail addresses for your survey ? Q13 Proposed Data Collection Start Date (month, day, year 00/00/00 format) Q14 Proposed Completion of Data Collection Q15 Funding Source External Grant MU Grant Non-Funded Project Q16 Methods and Procedures Describe in detail the study design and all procedures (sequentially) to be applied to participants Q17 What in your estimation is the nature of the risk to individuals participating in your study? Minimal risk - meaning that the risk of harm anticipated in the proposed research is not more likely than those risks encountered in daily life, or during routine physical or psychological examinations/tests More than minimal risk - risk exists when the possibility of physical or psychological harm or harm related to breach of confidentiality or invasion of privacy is greater than what is typically encountered in everyday life. Q18 Describe any protections against the risks you propose to implement including how you ensure data security Q19 Describe any potential benefits expected from this research project. Benefits can be directly to the participant and or community Q20 Describe any monetary or other forms of compensation which will be provided to participants, and any conditions which must be fulfilled to receive compensation Q21 Describe any alternatives to participation in the study which might be advantageous to the participant. If the participants are to receive academic credit for research participation, describe the alternatives available to earn equivalent academic credit Q22 Identify the nature of any information to be purposely withheld from participants, and provide justification for the non-disclosure Q23 Describe the procedure for post-study debriefing of participants as applicable. Q24 Describe explicitly how confidentiality of data will be maintained. If any information with participants identifiers will be released, specify the participants. Include a statement that all data will be retained for at least three years in compliance with federal regulations Q25 Do you have a consent form for this project? Yes No Q26 Some projects may be exempt from ongoing review by the IRB. Exempt DOES NOT exempt the project from the review process only that the IRB does not follow up annually with these projects.Exempt categories DO NOT apply to research involving deception of participants (i.e., where the researcher deceives the participant with regard to the purpose of the research and/or the results of the participant's actions in the study), OR projects involving sensitive behavioral research, OR research involving pregnant women, prisoners, mentally disabled people, and other participant populations determined to be vulnerable.Based upon reading the categories below, does your research fall into one of the six exempt categories? (Check all that apply) Research conducted in established or commonly accepted educational settings, involving normal educational practices provided all seven components are met as outlined in the MU Institutional Review Board Policies and Procedures Manual. Research involving the use of educational tests (cognitive, diagnostic, aptitude, achievement), survey procedures, interview procedures or observation of public behavior, unless: (a) information obtained is recorded in such a manner that human participants and animal can be identified, directly, or through identifiers linked to the paricipants; and (b) any disclosure of the human and animal participants' responses outside the research could reasonably place the participants at risk for criminal or civil liability or be damaging to the participants' financial standing, employability, or reputation. Sensitive survey research is not exempt. Research involving the collection or study of existing data, documents, records, pathological specimens, or diagnostic specimens, if these sources are publicly available or if the information is recorded by the investigator in such a manner that subjects cannot be identified directly or through identifiers linked to the subjects Research involving the use of educational tests (cognitive, diagnostic, aptitude, achievement), survey procedures, interview procedures, or observation of public behavior that is not exempt under paragraph 2 of this section, if: (a) the participants are elected or appointed public officials or candidates for public office; or (b) federal statute(s) require(s) without exception that the confidentiality of the personally identifiable information is maintained throughout the research and thereafter Research and demonstration projects which are conducted by or subject to the approval of department or agency heads, and which are designed to study, evaluate, or otherwise examine: (a) public benefit or service programs; (b) procedures for obtaining benefits or services under those programs; (c) possible changes in or alternatives to those programs or procedures; and/or (d) possible changes in methods or levels of payment for benefits or services under those programs Taste and food quality evaluation and consumer acceptance studies: (a) if wholesome foods without additives are consumed; or (b) if a food is consumed that contains a food ingredient at or below the level and for a use found to be safe, or agricultural chemical or environmental contaminant at or below the level found to be safe, by the Food and Drug Administration or approved by the environmental Protection Agency or the Food Safety and Inspection Service of the U.S. Department of Agriculture None of these apply Q27 Studies may also fall into expedited review categories. Please read the descriptors below and select any of the review categories that apply to your project. Minor modifications or additions to existing approved studies Research on individual or group behavior or characteristics of individuals, such as studies of perception, cognition, game theory, or test development, where the investigator does not manipulate subjects' behavior and the research will not involve stress to subjects The study of existing data, documents, records, pathological specimens, or diagnostic specimens Voice recordings made for research purposes such as investigations of speech defects Moderate exercise by healthy volunteers Collection of blood samples by venipuncture, in amounts not exceeding 450 milliliters in an eight-week period and no more often than two times per week, from subjects 18 years of age or older who are in good health and not pregnant Collection (in a non-disfiguring manner) of hair, nail clippings, and deciduous teeth; and permanent teeth if patient care indicates a need for extraction Collection for analysis of excreta and external secretions including sweat, uncannulated saliva, placenta removed at delivery, and amniotic fluid at the time of rupture of the membrane prior to or during labor Recording of data from subjects 18 years of age or older using noninvasive procedures routinely employed in clinical practice. This includes the use of physical sensors that are applied either to the surface of the body or at distance and do not involve input of matter or significant amounts of energy into the subject or an invasion of the subject's privacy. (These procedures include weighing, testing sensory acuity, electrocardiogram, electroencephalogram, thermography, detection of naturally occurring radioactivity, diagnostic echography, and electroretinography. It does not include exposure to electromagnetic radiation outside the visible range, i.e., x-rays, microwaves. Collection of both supra- and subgingival dental plaque and calculus, provided the procedure is not more invasive than routine prophylactic scaling of the teeth and the process is accomplished in accordance with accepted prophylactic techniques Research on drugs or devices for which an investigational new drug exemption or an investigational device exemption is not required None of these apply Q28 Date of Submission Q29 Please attach a copy of your consent form Q30 If using Qualtrics, please "collaborate"/share your survey with the IRB Chair (found under "IRB CHAIR") and your instructor. If not, please attach a copy of your survey. Q31 Please attach a copy of brochures, flyers, or other material that you intend to use to recruit participants. This includes any letters, and/or email messages.