Balancing Equations
advertisement

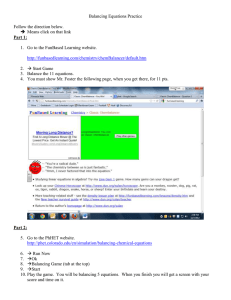
Balancing Equations Balancing a chemical equation refers to the skill (or art) of making sure that all atoms that are written on the left of a chemical reaction as reactants are also present on the right side of the equation as products. Since many of the problems you will see in this class require you to start with a balanced chemical equation, this is a skill you must learn now and will use over and over of the course of the year. Balancing Equations Balancing a chemical equation isn’t that hard to do, it is a simple trial trial-and-error and error process that is repeated for each element in the equation until all elements are balanced. After you get the hang of it it can even be kind of fun as you fi figure outt a way tto puzzle l th through h your problem. bl Balancing Equations Example 1. Balance the following chemical equation: FeCl3(aq) + AgNO3(aq) = AgCl(s) + Fe(NO3)3(aq) The (aq) and the (s) refer to the physical form of the chemical h i l and d we will ill talk t lk about b t that th t more in i chapter h t 4, 4 for f now the physical descriptors can be ignored. To balance the equation the only change you can make is to change the coefficient in front of a molecule. You cannot change any of the subscripts in a molecule because that changes the chemical to a different substance. ? FeCl3((aq) q) + ? AgNO g 3((aq) q) = ? AgCl(s) g ( ) + ? Fe(NO ( 3)3((aq) q) Fe 1 0 0 1 So pick an element and make sure you have the same number of atoms of that element on both sides of the equation. For instance, let’s start with the iron (Fe). How many Fe’s are there on each side of the equation? One Fe both sides of the equations so it is already balanced! That means the coefficients for FeCl3 and Fe(NO3)3 can be set to 1. 1 FeCl3((aq) q) + ? AgNO g 3((aq) q) = ? AgCl(s) g ( ) + 1 Fe(NO ( 3)3((aq) q) Fe 1 0 0 1 So pick an element and make sure you have the same number of atoms of that element on both sides of the equation. For instance, let’s start with the iron (Fe). How many Fe’s are there on each side of the equation? One Fe both sides of the equations so it is already balanced! That means the coefficients for FeCl3 and Fe(NO3)3 can be set to 1. 1 FeCl3((aq) q) + ? AgNO g 3((aq) q) = ? AgCl(s) g ( ) + 1 Fe(NO ( 3)3((aq) q) Fe Cl 1 3 0 0 0 1 1 0 Now let’s try the Cl Cl. (Notice I will continue with the table that I started on the last page) 3 Cl’s on the left, 1 Cl on the right. So we need to multiply p y the AgCl g on the right g hand side byy a coefficient of 3 to get 3 Cl’s on this side. 1 FeCl3((aq) q) + ? AgNO g 3((aq) q) = ? AgCl(s) g ( ) + 1 Fe(NO ( 3)3((aq) q) Fe Cl 1 3 0 0 0 3(1) 1 0 Now let’s try the Cl Cl. (Notice I will continue with the table that I started on the last page) 3 Cl’s on the left, 1 Cl on the right. So we need to multiply p y the AgCl g on the right g hand side byy a coefficient of 3 to get 3 Cl’s on this side. A d this And thi sets t the th coefficient ffi i t off AgCl A Cl to t 3 1 FeCl3((aq) q) + ? AgNO g 3((aq) q) = 3 AgCl(s) g ( ) + 1 Fe(NO ( 3)3((aq) q) Fe Cl 1 3 0 0 0 3(1) 1 0 Now let’s try the Cl Cl. (Notice I will continue with the table that I started on the last page) 3 Cl’s on the left, 1 Cl on the right. So we need to multiply p y the AgCl g on the right g hand side byy a coefficient of 3 to get 3 Cl’s on this side. A d this And thi sets t the th coefficient ffi i t off AgCl A Cl to t 3 1 FeCl3((aq) q) + ? AgNO g 3((aq) q) = 3 AgCl(s) g ( ) + 1 Fe(NO ( 3)3((aq) q) Fe Cl Ag 1 3 0 0 0 1 0 3(1) 3(1) 1 0 0 Now let’s try the Ag’s One on the left, three on the right, we will need to multiply The AgNO3 by three to make it all balance. balance 1 FeCl3((aq) q) + 3 AgNO g 3((aq) q) = 3 AgCl(s) g ( ) + 1 Fe(NO ( 3)3((aq) q) Fe Cl Ag N O 1 3 0 0 0 0 0 3(1) 3 9 0 3(1) 3(1) 0 0 1 0 0 3 9 Now let’s try the N’s… The N’s look good, I didn’t have to mess with any coefficients, so let’s let s finish with the O O’ss The O’s are balanced as well, so we must be done. The final balanced equation is: 1 FeCl3(aq) + 3 AgNO3(aq) = 3 AgCl(s) + 1 Fe(NO3)3(aq) Balancing equations As you balance a reactant and a product that both contain a given element in an equation, equation you try not to have to change the coefficient on either of those two compounds again. If you do, then you have to go back and start over. Notice how thi worked this k d in i the th last l t example. l Balancing equations Step 1 Fe FeCl3(aq) + AgNO3(aq) = AgCl(s) + Fe(NO3)3(aq) 1 0 0 1 Now you ttry nott tto mess with N ith either ith F FeCl Cl3 or Fe(NO F (NO3)3 from f this point onward. I will underline them remind myself to try not to touch these coefficients again. Balancing equations Step 2 Fe Cl FeCl3(aq) + AgNO3(aq) = 3AgCl(s) + Fe(NO3)3(aq) 1 0 0 1 3 0 3(1) 0 Now with the Cl, the AgCl coefficient is fixed, so I will underline that one as well. Balancing equations Step 3 Fe Cl A Ag FeCl3(aq) + 3AgNO3(aq) = 3AgCl(s) + Fe(NO3)3(aq) 1 0 0 1 3 0 3(1) 0 0 3(1) 3 0 Now with the Ag, the AgNO3 coefficient is fixed, so I will underline that one as well. Balancing equations Steps 4 and 5 q) + 3AgNO g 3((aq) q) = 3AgCl(s) g ( ) + Fe(NO ( 3)3((aq) q) FeCl3((aq) Fe 1 0 0 1 Cl 3 0 3(1) 0 Ag 0 (3)1 0 3 N 0 3 0 3 O 0 9 0 9 And fixing the Ag fixes the coefficient of the last compound, g that the N and O will come into so we cross our fingers balance without and further changes. Balancing Equations Now try this one: ( ) + H2S(g) (g) Au2S3((s)) + H2(g) = Au(s) ? Au2S3(s) + ? H2(g) = ? Au(s) + ? H2S(g) Au 2 0 1 0 Au first. So you have 1 Au2S3 on the left for a total of 2 Au’s, that means you need 2 Au(s) on the right. 1 Au2S3(s) + ? H2(g) = 2 Au(s) + ? H2S(g) Au 1(2) 0 2(1) 0 S 3 0 0 1 S next. So if you have 1 Au2S3 on the left, you’ll need 3 H2S on the right. 1 Au2S3(s) + ? H2(g) = 2 Au(s) + 3 H2S(g) Au 1(2) 0 2(1) 0 S 1(3) 0 0 H 0 2 0 3(1) 3(2) H next. So if you have 3 H2S on the left you’ll need 3 H2’s on the right. 1 Au2S3(s) + 3 H2(g) = 2 Au(s) + 3 H2S(g) Au 1(2) 0 2(1) 0 S 1(3) H 0 0 3(2) 0 3(1) 0 3(2) And it all worked, your final answer is: 1 Au A 2S3(s) ( ) + 3 H2(g) ( ) = 2 Au(s) A ( ) + 3 H2S(g) S( ) These were a couple of nice, easy examples. p Now look at the next tutorial for some harder problems.