Molecular Tagging Technique for the Simultaneous AIAA-2006-0041
advertisement

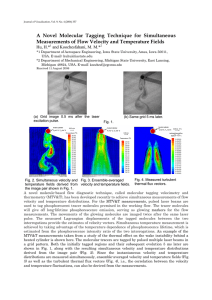
44th AIAA Aerospace Sciences Meeting and Exhibit AIAA-2006-0041 Jan 9 – 12, 2006, Reno, Nevada Molecular Tagging Technique for the Simultaneous Measurements of Flow Velocity and Temperature Fields Hui Hu* Department of Aerospace Engineering, Iowa State University, Ames, IA, 50011 U.S.A and Manoochehr M. Koochesfahani† Department of Mechanical Engineering, Michigan State University, East Lansing, MI, 48824, U.S.A We report recent improvements to the Molecular Tagging Velocimetry and Thermometry (MTV&T) technique for the simultaneous measurements of velocity and temperature fields in fluid flows. Instead of using tiny particles, specially-designed phosphorescent molecules, which can be turned into long-lasting glowing molecules upon excitation by photons of appropriate wavelength, are used as tracers for both velocity and temperature measurements. A pulsed laser is used to “tag” the tracer molecules in the regions of interest, and the movements of the tagged molecules are imaged at two successive times within the photoluminescence lifetime of the tracer molecules. The measured Lagrangian displacement of the tagged molecules between the two image acquisitions provides the estimate of the fluid velocity vector. The simultaneous temperature measurement is achieved by taking advantage of the temperature dependence of phosphorescence lifetime, which is estimated from the intensity ratio of the tagged molecules in the two images. The implementation and application of the MTV&T technique are demonstrated by conducting simultaneous velocity and temperature measurements in the wake region of a heated circular cylinder and a transient electroosmotic flow (EOF) inside a microchannel. I. Introduction V ELOCITY and temperature are the two most important parameters to study complex thermal-fluid problems. The simultaneous information revealed from velocity and temperature distributions could help us very much to improve our understanding about underlying physics of complex thermal-fluid phenomena, which, in turn, will greatly improve the performance of a large variety of heat-exchange devices used in our daily life. Recent advances in computer technology and turbulence modeling have resulted in different computational fluid dynamics (CFD) codes for various flows of practical interest. However, a general theory capable of predicting the behaviors of complex thermal flows with high heat fluxes and varying properties has not yet been established. Before computational methods can be applied with confidence to these thermal flows, they must be validated by comparing precise measurements of the turbulence characteristics of the flow velocity and temperature fields. Therefore, development of novel experimental techniques capable of simultaneously measuring whole-field velocity and temperature distributions is very important and essential for either optimum design of various heat-exchange devices or the development and validation of fundamental physical models. There is an extensive body of literature on the measurements of either velocity components or temperature in many types of flows. Studies involving simultaneous velocity and temperature measurements are far less common in the literature. Earlier work about the simultaneous velocity and temperature measurement in thermal flows was conducted by using intrusive probes such as cold-wire sensors mounted on X-wire probes1-2. The advent of optical diagnostics such as Laser Doppler Velocimetry (LDV) and Laser Induced Fluorescence (LIF) has presented new opportunities for the non-intrusive simultaneous acquisition of velocity and temperature measurements at interested points3-4. More recently, the advances in the development of whole-field flow diagnostic techniques such as Particle * † Assistant Professor, Department of Aerospace Engineering, Senior Member of AIAA, Email: huhui@iastate.edu. Professor, Department of Mechanical Engineering, Associate Fellow of AIAA, Email: koochesf@egr.msu.edu. 1 American Institute of Aeronautics and Astronautics 44th AIAA Aerospace Sciences Meeting and Exhibit AIAA-2006-0041 Jan 9 – 12, 2006, Reno, Nevada Image Velocimetry (PIV) and Planar Laser Induced Fluorescence (PLIF) have led to efforts at obtaining simultaneous maps of velocity and temperature distributions by using a PIV-PLIF combined system5-7. By using Thermochromic Liquid Crystal (TLC) encapsulating micro-spheres as tracer particles, a Digital Particle Image Velocimetry/Thermometry (DPIV/T) technique has also been developed8-9 recently to measure velocity and temperature fields simultaneously in turbulent thermal flows. It should be noted that all the optical velocimetry techniques mentioned above are particle-based velocimetry techniques, which measure the velocity of tracer particles, other than the velocity of working fluid directly. The velocity of the working fluid is deduced based on the assumption of that the tracer particles move with the same local velocity as the working fluid. The potential artifacts associated with the use of particles as tracers, such as the effects of inertia and buoyancy forces acting on the tracer particles, should be carefully considered for each experiment in order to make a physically meaningful measurement of fluid velocity. Even for the cases where tracer particles can track the flow perfectly, strong out-of-plane motions that bring tracer particles into and out of the laser sheet can affect the accuracy of in-plane velocity measurements in PIV10. For the temperature measurement with TLC micro-spheres8-9 or LIF dye infused particles7 mentioned above, additional considerations are also required about the thermal responses of the particle tracers. Using molecules as diagnostic tracers is expected to significantly mitigate, and perhaps even eliminate, these artifacts or implications. In the present paper, we report recent progress in the development of a molecule-based flow diagnostic technique, named as Molecular Tagging Velocimetry and Thermometry (MTV&T), for simultaneous measurements of velocity and temperature distributions in fluid flows. Instead of using tiny particles, the MTV&T technique uses specially-designed phosphorescent molecules as the tracers for both velocity and temperature measurements. The tracer molecules can be turned into long-lasting glowing tracers upon excitation by photons of an appropriate wavelength. The MTV&T technique is actually an extension of the Molecular Tagging Velocimetry (MTV) technique11-12. While the MTV technique can measure flow velocity only, the MTV&T technique described in the present study can map fluid temperature distributions simultaneously in addition to whole-field velocity measurements. In the sections that follow, a brief overview of MTV is given along with more details of lifetimebased Molecular Tagging Thermometry (MTT) and the related properties of the phosphorescent tracer used. Demonstrations of the application of this molecular-based approach are provided by carrying out simultaneous measurements of the velocity and temperature fields in the wake of a heated cylinder and a transient electroosmotic flow (EOF) inside a microchannel. II. Molecular Tagging Velocimetry and Themometry (MTV&T) technique Phosphorescent Molecular Tracers Supramolecules of phosphorescent triplex (1-BrNp⋅Gβ-CD⋅ROH) were used as the molecular tracers in the present study. The phosphorescent triplex (1-BrNp⋅Gβ-CD⋅ROH) is actually the mixture compound of three different chemicals, which are lumophore (indicated collectively by 1-BrNp), glucosyl-β-cyclodextrin (indicated collectively by Gβ-CD) and alcohols (indicated collectively by ROH). Upon the pulse excitation of a UV laser, the phosphorescence lifetime of the phosphorescent triplex (1-BrNp⋅Gβ-CD⋅ROH) molecules in an aqueous solution can be as long as 5ms at room temperature. Further information about the chemical and photoluminescent properties of the phosphorescent triplex (1-BrNp⋅Gβ-CD⋅ROH) is available at Ref. 13-14. Since the chemical composition of the phosphorescent triplex (1-BrNp⋅Gβ-CD⋅ROH) affects the intensity and lifetime of the phosphorescence emission significantly, we used a concentration of 2×10−4 M for Gβ-CD, a saturated (approximately 1×10−5 M) solution of 1BrNp and a concentration of 0.06 M for the alcohol (ROH) in the present study, as suggested by Gendrich et al.11. Flow Velocity Measurement The methodology of the MTV&T technique is actually an extension of Molecular Tagging Velocimetry (MTV) technique11-12, which can be thought as the molecular counterpart of the Particle Imaging Velocimetry (PIV) technique for flow velocity field measurements. Compared with PIV, MTV offers advantages in situations in which the use of seed particles is either not desirable or may lead to complications. Figure 1 illustrates one implementation of the technique. A planar grid of intersecting laser beams, formed from a pulsed UV excimer laser (20 ns pulse, 308 nm wavelength), turns on the luminescence of the 1-BrNp⋅Gβ-CD⋅ROH supramolecules that are premixed in a water flow of a vortex ring approaching a solid wall at normal incidence11. The displacement of the tagged regions is determined, in this case, using a direct spatial correlation method. The conventional planar imaging shown in Figure 1 provides information on two components of the velocity vector, the projection onto the viewed plane. Stereo 2 American Institute of Aeronautics and Astronautics 44th AIAA Aerospace Sciences Meeting and Exhibit AIAA-2006-0041 Jan 9 – 12, 2006, Reno, Nevada imaging can produce the complete three components of the velocity vector15. Various advances in MTV technique in terms of available molecular tracers, methods of tagging, detection/imaging and data processing can be found in several review articles 16-19, in addition to a special issue of Measurement Science and Technology on this topic20. (a) (b) (c) Figure 1. Typical MTV image pairs and the resultant two-component velocity field (Gendrich et al. 1997). The flow show is from a vortex ring impacting on a flat wall at normal incidence. The axis of symmetry is indicated by the dashed lines: (a) The grid imaged 1 μs after the laser pulse. (b) The same grid imaged 8 ms later. (c) The velocity field derived from (a) and (b). Tagging the molecular tracers along single or multiple parallel lines is perhaps the simplest method of tagging and has been utilized in a large fraction of studies to date. It is clear that line tagging allows the measurement of only one component of velocity, that normal to the tagged line. In addition, the estimate of this velocity component has an inherent error associated with it, which is connected with the ambiguity in the unique determination of the displacements of various portions of a (continuous) tagged line. This ambiguity can also cause significant errors in the temperature inferred from MTT in three-dimensional flows with non-uniform temperature field. In order to unambiguously measure two components of the velocity in a plane, the luminescence intensity field from a tagged region must have spatial gradients in two, preferably orthogonal, directions. For single-point velocimetry, this is easily achieved using a pair of crossing laser beams; a grid of intersecting laser lines allows multi-point velocity measurements as shown in Figure 1. As already mentioned, stereo imaging would allow the recovery of the third, out-of-plane, velocity component as well15. In the original work of Gendrich et al.11, for each laser pulse the MTV image pairs were acquired by a pair of aligned image detectors viewing the same region in the flow. In the current work, the two detectors are replaced by a single intensified CCD camera (PCO DiCam-Pro) operating in the dual-frame mode, which allows the acquisition of two images of the tagged regions with a programmable time delay between them. The displacement of the tagged regions is determined by a direct digital spatial correlation technique21. A small window, referred to as the source window, is selected from a tagged region in the earlier image, and it is spatially correlated with a larger roam window in the second image. A well-defined correlation peak occurs at the location corresponding to the displacement of the tagged region by the flow; the displacement peak is located to sub-pixel accuracy using a multidimensional polynomial fit21. For velocity measurement, MTV utilizes the information about the spatial distribution of the photoluminescence of the tagged molecules within a region to determine the displacement and, therefore, the flow velocity in the tagged region. As described in the following section, monitoring the phosphorescence intensity decay rate (i.e. emission lifetime) within the tagged regions provides information on the fluid temperature within those regions simultaneously along with velocity information. Flow Temperature Measurement According to quantum theory22, the intensity of phosphorescence emission decays exponentially. For a dilute solution and unsaturated laser excitation, the collected phosphorescence signal (Sp) by using a gated imaging detector with integration starting at a delay time to after the laser pulse and a gate period of δ t can be given by 23-24: 3 American Institute of Aeronautics and Astronautics 44th AIAA Aerospace Sciences Meeting and Exhibit AIAA-2006-0041 Jan 9 – 12, 2006, Reno, Nevada ( S p = AI i Cε Φ p 1 − e −δ t / τ )e −to / τ (1) Photoluminescence where A is a parameter representing the detection collection efficiency, Ii is the local incident laser intensity, C is the concentration of the phosphorescent dye (the tagged molecular tracer), ε is the absorption coefficient, and Φp is the phosphorescence quantum efficiency. The emission lifetime τ refers to the time at which the intensity drops to 37% (i.e. 1/e) of the initial intensity. In general, the absorption coefficient ε , quantum yield Φp, and the emission lifetime τ are temperature dependent25, resulting in a temperature-dependent phosphorescence signal (Sp). Thus, in principle, the collected phosphorescence signal (Sp) may be used to measure fluid temperature if the incident laser intensity and the concentration of the phosphorescent dye remain constant (or are known) in the region of interest. Actually, a thermometry method has been suggested by imaging the laser-induced phosphorescence intensity of 1-BrNp⋅GβCD⋅ROH molecules26. The method is the phosphorescence version of the conventional LIF technique, where the acquired phosphorescence intensity is used directly to refer to fluid temperature. It should be noted that the collected phosphorescence signal (Sp) is also the function of incident laser intensity (Ii) and the concentration of the phosphorescent dye (C). Therefore, the spatial and temporal variations of the incident laser intensity and the nonuniformity of the phosphorescent dye in the region of interest would have to be corrected separately in order to Laser excitation pulse derive quantitative temperature data from the acquired phosphorescence images. In practice, however, it is very −δ t /τ ⎞ ⎛ difficult, if not impossible, to ensure a non-varying S P1 = AIi Cε Φ p ⎜ 1 − e 1 ⎟ e−t o /τ ⎝ ⎠ incident laser intensity distribution, especially for unsteady thermal flows with varying index of refraction. ⎛ −δt /τ ⎞ −(t +Δt)/τ SP2 = AIi Cε Φp ⎜ 1−e 2 ⎟ e o This will cause significant error in the fluid temperature ⎝ ⎠ measurements. To overcome this problem, Hu and 23 Koochesfahani have recently developed a novel SP1 SP2 lifetime-based thermometry that can eliminate the effects of incident laser intensity and concentration of t0 δt1 phosphorescent dye on fluid temperature measurements. Δt δt2 time Following is a brief description of the technical basis of the lifetime-based thermometry; further details are Figure 2. Schematic of the timing chart for the available at Ref. 23-24: As illustrated in Fig. 2, the MTV&T measurement. phosphorescence emission of the tagged molecular tracers is interrogated at two successive times after the same laser excitation pulse. The collected phosphorescence intensity in each of the acquired phosphorescence images can be expressed according to Eq. 1. The intensity ratio of the second image interrogation ( S P 2 ) to the first image interrogation ( S P 1 ) will be − δ t / τ (T ) R 1 S 1− e = P1 = ⋅ e − Δ t /τ / ( ) − δ t τ T S P2 2 1− e = R (τ (T ), δ t 1 , δ t 2 , Δ t ) (2) From Eq. 2, it is evident that the intensity ratio of the two successive phosphorescence images is only the function of phosphorescence lifetime ( τ ) and the parameters for image acquisition ( δ t1 , δ t 2 and Δt). The effects of the incident laser intensity (Ii) and the non-uniformity of the phosphorescent dye concentration (e.g. due to photobleaching) are eliminated by taking the intensity ratio of the two phosphorescence images. For a fixed set of δ t1 , δ t 2 and Δt values, Eq. 2 defines a unique relation between phosphorescence intensity ratio R and fluid temperature T, which can be used for thermometry. For the examples shown in the present study, the gate periods ( δ t1 and δ t 2 ) for acquiring the first and second phosphorescence images were set to be the same, i. e. δ t1 = δ t 2 . Therefore, Eq. 2 can be further simplified, and the phosphorescence lifetime can be calculated according to: τ = Δt Δt = , ln R ln(S p1 / S p 2 ) 4 American Institute of Aeronautics and Astronautics (3) 44th AIAA Aerospace Sciences Meeting and Exhibit AIAA-2006-0041 Jan 9 – 12, 2006, Reno, Nevada 5.0 exponential fit o T = 50 C o T = 40 C o T = 30 C o T = 25 C Normalized intensity 1.0 4.5 0.8 0.6 3.5 set set set set 4 3 2 1 3.0 2.5 2.0 1.5 0.4 1.0 0.2 0 0.5 data data data data 4.0 lifetime (ms) 1.2 0.5 1.0 1.5 2.0 2.5 Time delay to after laser pulse (ms) Figure 3. Phosphorescence intensity decay curves at several temperature levels. 3.0 20 25 30 35 40 45 50 o temperature ( c) Figure 4. Phosphorescence lifetime of 1-BrNp⋅GβCD⋅ROH complex versus temperature. resulting in the distribution of the phosphorescence lifetime over a two-dimentional domain, and the temperature distribution in the flow as long as the temperature dependance of phosphorescence lifetime is known. A calibration procedure to obtain the quantitative relationship between the temperature and the phosphorescence lifetime of the phosphorescent triplex (1-BrNp⋅Gβ-CD⋅ROH) was conducted. Figure 3 depicts the measured phosphorescence intensity decay curves at several temperature levels. It can be seen that the phosphorescence intensity decay curves are very well approximated by single-exponential curves, as expected theoretically. Therefore, Eq. 3 can be used to calculate the phosphorescence lifetime of the phosphorescent triplex (1-BrNp⋅GβCD⋅ROH). Figure 4 shows the calibration curve of the phosphorescence lifetime of the phosphorescent triplex (1-BrNp⋅GβCD⋅ROH) changing with temperature. It can be seen that the phosphorescence lifetime of 1-BrNp•Mβ-CD•ROH varies significantly with temperature. The relative temperature sensitivity of the phosphorescence lifetime ranges between 5.0% per ºC at 20ºC to 20.0% per ºC degree at 50ºC. The temperature sensitivity is much higher than those of fluorescent dyes (such as Rhodamine B which is only about 2.0% per degree Celsius27 ). Furthermore, since the intensity ratio R is also a function of the controllable parameters ( δ t1 , δ t 2 and Δt) for the image acquisition according to Eq. 2, the sensitivity for temperature measurement can be further increased by adjusting these parameters. By adjusting the controllable parameters ( δ t1 , δ t 2 and Δt ) for the image acquisition, Hu et al.28 have recently reported a super-high temperature sensitivity (up to 18.2% per degree Celsius) that is nearly 10 times higher than most fluorescent dyes used for temperature measurements. In summary, the MTV&T technique achieve the simultaneous measurements of velocity and temperature distributions in fluid flows by using a pulsed laser to “tag” the specially-designed tracer molecules in the regions of interest. The movements of the tagged tracer molecules are interrogated at two successive times within the photoluminescence emission lifetime of the tagged molecules. The measured Lagrangian displacement of the tagged molecules provides the estimate of the fluid velocity vector. The simultaneous temperature measurement is achieved by taking advantage of the temperature dependence of phosphorescence lifetime, which is estimated from the phosphorescence intensity ratio of the tagged molecules in the two images. III. Application to the Wake of a Heated Cylinder In order to demonstrate the feasibility of the technique described above, MTV&T is applied to conduct simultaneous temperature and velocity measurements in the wake of a heated cylinder. A schematic of the experimental setup is shown in Figure 5. The test cylinder was installed horizontally in a gravity-driven vertical water channel. The dimensions of the test section were 50mm (width) × 30mm (height) × 200mm (length). Two sides of the test section contained quartz windows to allow the transmission of the excimer laser UV light. The 1BrNp•Mβ-CD•ROH phosphorescent triplex was premixed with water in a reservoir tank. A constant head tank was 5 American Institute of Aeronautics and Astronautics 44th AIAA Aerospace Sciences Meeting and Exhibit AIAA-2006-0041 Jan 9 – 12, 2006, Reno, Nevada used to maintain a steady inflow condition during the experiment. The overflow constant head tank honeycomb constant head tank was filled from the mesh structure reserve tank by using an electric pump. A convergent section with heated honeycomb and mesh structures was cylinder used upstream of the test section to constant power 12-bit intensified pulsed DC supply produce a uniform condition for the CCD camera laser grid flow approaching the test cylinder. Y DICAM-PRO The velocity of the flow in the water thermometer channel was adjustable by operating the valve at the downstream end of x To laser the water channel. A copper tube with outer Quartz Digital delay diameter of D = 4.76 mm and inner windows generator diameter of 4.00 mm was used as the test cylinder. The cylinder was host reserve tank valve computer heated using a 3.1 mm diameter rod cartridge heater (Watlow Firerod) that was placed inside the copper tube. pump High thermal conductivity paste (OMEGATHERM 201) was pressed in to fill the gap between the rod Figure 5. Experiment setup cartridge heater and the inner wall of the copper tube. The rod cartridge heater was powered by a DC power supply (Kepco, BOP-200-2M). Two J-type thermocouples were embedded in the gap at the mid-span of the cylinder at two angular locations to provide the estimate of the cylinder temperature. The thermocouples were connected to a two-channel thermometer (Omega HH23), which had a resolution of ±0.1°C. A grid of intersecting laser lines were used for molecular tagging. The 20ns, 150mJ/pulse rectangular beam from an Excimer UV laser (308nm wavelength) was manipulated by a set of cylindrical optics to increase its aspect ratio. The resulting laser sheet was split by a 50:50 beam splitter; each of the two resulting sheets passed through a beam blocker to generate the grid pattern. A 12-bit (1280 × 1024 pixels) gated intensified CCD camera (PCO DICAM-Pro) with a fast decay phosphor (P46) was used in the present study to conduct image recording. The camera was operated in the dual-frame mode, where two full-frame images of phosphorescence were acquired in quick succession from the same laser excitation pulse. For the results given in the present study, the first images were captured at the time delay of 1ms after the laser pulse with the interrogation gate period of 0.5ms. The second phosphorescence images were captured at the time delay of 6 ms after the laser excitation pulse with the same interrogation gate period. The laser and the camera were synchronized using a digital delay generator (SRS DDG535), which controlled the timing of the laser sheet illumination and the CCD camera data acquisition. The phosphorescence images captured by the CCD camera were subsequently transferred to a host computer for analysis. In the present study, the heated cylinder was placed horizontally in the middle of the water channel, and the forced flow is vertically downward to approach the heated cylinder. Such arrangement makes the heated cylinder operates in a contra flow, i.e. the direction of the approaching forced flow opposes the direction of the buoyancy force acting on the fluids around the heated cylinder. The velocity of the approaching forced flow was measured at about 10 diameters upstream of the test cylinder, which is 0.0255 m/s ( U ∞ =0.0255 m/s). The temperature of the forced flow in the head tank was 24.0 oC ( T∞ = 24.0 o C ), and the corresponding Reynolds number o Re D = ρ U ∞ D / μ ∞ = 130 . For the results shown in Fig. 6, the temperature of the test cylinder was 42.0 C. The corresponding Grashof number GrD = gβ (Tw - T∞ )D 3 /ν ∞ 2 was 5400, and the corresponding Richardson number ( Ri D = GrD / Re D ) is 0.31. A typical pair of phosphorescence images acquired after the same excitation laser pulse for the experimental conditions described above were given in Figure 6(a) and 6(b). The dark bands on the top left of the images are shadows caused by the cylinder blocking the laser beams. The “dark regions” in the phosphorescence images downstream of the cylinder correspond to the warm fluid shedding periodically from the hot boundary layer around 6 American Institute of Aeronautics and Astronautics 44th AIAA Aerospace Sciences Meeting and Exhibit AIAA-2006-0041 Jan 9 – 12, 2006, Reno, Nevada the heated cylinder. From the comparison of the two images it can be seen that the dark regions become more pronounced as the time delay between the laser pulse and phosphorescence acquisition increases. This is due to the fact that the warmer fluid has a shorter phosphorescence lifetime, resulting in a larger decay in emission intensity than that in the cooler ambient fluid. The instantaneous velocity distribution can be derived from the image pair by measuring the displacements of the tagged regions using a spatial correlation approach21. A source (or interrogation) window size of 32 × 32 pixel (corresponding to a region 1.12mm × 1.12mm in physical space) was used in the present study, along with 50% overlap between consecutive windows. Figure 6(c) shows the instantaneous velocity distribution determined from the image pair. Note that velocity data are not available within the shadow regions caused by the cylinder blocking the laser light. The instantaneous velocity field shows a long re-circulation region downstream of the heated cylinder, extending to an X/D of about 3 in this realization, and unsteady shedding of vortex structures. These general features are similar to the case of unheated, isothermal, cylinder. By contrast, however, the time series of the measured instantaneous velocity fields indicates that the unsteady vortex structures shed periodically at a frequency of f ≈0.83 Hz, corresponding to a Strouhal number St ≡ fD / U ≈ 0.155 for the present experimental condition. This value is noticeably smaller than the Strouhal number of about 0.176 found in the literature (also confirmed in our experiments, results not shown) for an unheated cylinder at the Reynolds number of 130 in this experiment. This is believed to be a buoyancy-induced effect; a systematic study of the influence of increasing Richardson number on vortex shedding is currently under way. The image pair in Figure 6 (a) and 6(b) also allows the determination of the temperature distribution simultaneous with the velocity field already described. Consistent with the correlation method used for the measurement of the displacement of tagged regions, the same interrogation regions of 32 × 32 pixel in size were chosen in the first phosphorescence image to provide the average phosphorescence intensity S P1 within those regions. The molecules tagged within each region convect to a new region in the second phosphorescence image according to their Lagrangian displacement by the flow over the time delay between the two images. This displacement field is, of course, the basis of measuring the velocity field with MTV and is already available from Figure 6(c). The mass diffusion of tagged molecules out of interrogation windows is negligibly small (the mass diffusion length in this experiment is about 1/500 of the interrogation window size). Therefore, for each interrogation window in the first phosphorescence image, the position of the corresponding “displaced” window in the second phosphorescence image was determined based on the already measured velocity field, and this provided the corresponding average phosphorescence intensity S P 2 within each region. Note that the procedure here is a first order method that uses a linear displacement model consistent with small Lagrangian displacements (i.e. small time delay between images) and small distortion of the tagged regions due to velocity gradients. Once the average phosphorescence intensities, S P1 and S P 2 , were determined for the corresponding regions in the two phosphorescence images, the phosphorescence lifetime was calculated based on Eq. 3, resulting in the measurement of temperature according to the lifetime-versus-temperature calibration curve in Figure 4. The simultaneous temperature field derived from the phosphorescence image pair, which is shown in Fig. 6(d), illustrates the instantaneous temperature distribution in the wake of the heated cylinder. The alternate shedding of “warm blobs” associated with the Karman vortices is clearly seen. The mean velocity and temperature fields were calculated from 360 instantaneous measurements, and their overall distributions are shown in Figure 6(e). As expected, a recirculation zone at the downstream of the heated cylinder is revealed clearly in the ensemble-averaged velocity field. The ensemble-averaged temperature field reveals that two high-temperature regions exist at the two sides of the wake region, which are corrsponding to the shedding paths of the “Karman” vortices and “warm blobs” revealed in the instantaneous measurement results. Since the velocity and temperature fields were measured simultaneously, the correlation between the velocity and temperature fluctuations can be calculated to generate the distribution of the mean turbulent heat flux u ' j T ' , as shown in Figure 6(f). The distribution of the mean turbulent heat flux vectors indicate how the heat flux was propagated in the wake of the heated cylinder. As shown in the figure, the structure of u j ' T ' in the near wake is dominated by two large counter-rotating vortex-like structures, which is almost symmetrical about the wake centerline. The counter-rotating vortex-like structures were found to be closely related to the appearances of the “Karman”vortex structures and “warm blobs” revealed in the above velocity and temperature measurement results. Therefore, the structures are believed to be the results of the large entrainment of the cold free stream fluid by the shedding of “Karman” vortices in the wake behind the heated cylinder. Based on the MTV&T measurements, a systematic study of the buoyancy effect on the wake instability behind a heated cylinder is currently under way. 7 American Institute of Aeronautics and Astronautics 44th AIAA Aerospace Sciences Meeting and Exhibit AIAA-2006-0041 Jan 9 – 12, 2006, Reno, Nevada (a). phosphorescence image of the tagged molecules acquired at 1.0 ms after the laser pulse (b). phosphorescence image of the same tagged molecules acquired 5ms later -1 -1 0.026 m/s 0 0 1 2 5 6 7 8 9 3 4 X/D 4 5 6 7 8 9 10 10 11 Temperature o ( C) 26.50 26.30 26.10 25.90 25.70 25.50 25.30 25.10 24.90 24.70 24.50 2 0.026 0.024 0.022 0.020 0.018 0.016 0.014 0.012 0.010 0.008 0.006 0.004 0.002 0.000 -0.002 -0.004 -0.006 -0.008 3 X/D 1 axial velocity (m/s) -5 -4 -3 -2 -1 0 1 2 3 4 5 6 11 7 -5 -4 -3 -2 -1 0 (c). instantaneous velocity field derived from the image pair. 0.03 m/s 0 3 4 5 6 7 (d). the derived simultaneous temperature field. 0 0.15 1 1 2 Temperature O ( C) 2 3 26.00 25.85 25.70 25.55 25.40 25.25 25.10 24.95 24.80 24.65 24.50 3 5 6 7 6 7 8 9 9 10 10 -4 -3 -2 -1 0 1 2 3 4 5 6 0.200 0.180 0.160 0.140 0.120 0.100 0.080 0.060 0.040 0.020 5 8 -5 normalized tubulent thermal flux 4 X/D 4 X/D 2 -1 -1 11 1 Y/D Y/D 7 11 -5 -4 -3 -2 -1 Y/D 0 1 2 3 4 (e). ensemble-averaged velocity and temperature distributions. (f). normalized turbulent thermal fluxes Figure 6. The MVT&T measurement results in the wake of a heated cylinder. 8 American Institute of Aeronautics and Astronautics 5 6 7 Y/D u ' j T ' /[U inlet (Tc − Tinlet )] 44th AIAA Aerospace Sciences Meeting and Exhibit AIAA-2006-0041 Jan 9 – 12, 2006, Reno, Nevada IV. Application to an Electroosmotic Flow Fluid transport through microchannels plays a significant role in a great number of emerging technologies such as micro-power generation, chemical separation, cell analysis, and biomedical diagnostics. A considerable amount of pressure difference may be required to drive fluid through a channel of tens of micron meters in size by using conventional pressure-driven technology. An alternative and efficient way of moving fluid within microchannels is through electroosmosis. Electroosmosis is the bulk movement of liquid relative to a stationary charged surface due to externally applied electrical field, which was first observed and reported about two centuries ago29. Most solid substance will acquire a relative electric charge (negative charge for fused-silica material such as glass) when in contact with an aqueous electrolytic solution, which, in turn, influences the charge distribution in the solution. Ions of opposite charge (counter-ions) to that of surface are attracted to the surface, and ions of the same charge (co-ions) are repelled from the surface as illustrated in Figure 7. The net effect is the formation of a region close to the charged surface called electrical double layer (EDL) in which there is the an excess of counter-ions distributed in a diffusion manner. The charge distribution in the solution, therefore, falls from its maximum near the wall (which is usually termed as zeta potential) to a zero charge in the fluid core. The thickness of the EDL is characterized by Debye length, which is the wall-normal distance over which the net charge has decreased to 1/e (37%) of the surface charge30. The thickness of EDL (i.e. Debye length) can range from angstroms to nanometers depending on the electrolyte solution. When an electric field is applied parallel to the charged surface, the positively charged cations and solvent molecules strongly absorbed at the wall will remain stationary. However, the mobile cations in the EDL near the surface wall will migrate toward the cathode due to the excess charge in the layers. Through the action of viscous forces, the core fluid will be pulled towards the cathode as well. The resulting electroosmotic flow velocity, ueof, is given by the well-known Smoluchowski equation30 ueof =εrεoζV/(µL), where μ is the liquid viscosity, εo the permittivity of vacuum, εr the relative permittivity, ζ the zeta potential, V the applied voltage, and L the length over which the voltage is applied. (a). Ion distribution (b). electric charge distribution (c). electroosmotic flow Figure 7. Schematic of electroosmotic flow Joule heating is the inherent by-product of the electric work in electroosmotic flows. The heat is generated by ohmic resistance of the electrolyte solution due to the passing electrical current. From the microscopic viewpoint, the frequent collision of migrated ions and solvent molecules convert some of the kinetic energy done by the electric field into the heat. This scenario is similar to the electrons moving through metal atoms. This internal heat source not only elevates the absolute fluid temperature but also generates temperature gradients in the microchannels31-32, the flow behavior is therefore strongly affected. The effects of Joule heating can compromise the performances of microfluidics or “lab-on-a-chip” devices by increasing dispersion in electrokinetic separation and inducing temperature sensitive chemical reactions31-32. Joule heating can also cause local liquid boiling in microfluidics, sometimes even to the point of destroying microchips33. As a consequence, Joule heating and micro-scale heat transfer in electrokinetically-driven microfluidics has attracted much attention in recent years. The inherent nature of the coupling of Joule heating to electroosmotic flows requires simultaneous information on electroosmotic velocity and fluid temperature in order to elucidate the underlying physics and thus further our understanding about Joule heating and micro-scale heat transfer process in microfluidics. Although several advanced flow diagnostic techniques, which include μ-PIV34, microscopic caged-dye imaging35, photobleach imaging36, and µ-LIF37, have been developed for in-situ measurements in microflows, none of these techniques is capable of simultaneously measuring “in-channel” flow velocity and fluid temperature in microflows. By using the MTV&T technique described above, Lum et al.38 have recently achieved simultaneous measurements of “in-channel” electroosmotic velocity and fluid temperature in an electroosmotic flow inside a microchannel. Figure 8 shows 9 American Institute of Aeronautics and Astronautics 44th AIAA Aerospace Sciences Meeting and Exhibit AIAA-2006-0041 Jan 9 – 12, 2006, Reno, Nevada example results of the MTV&T measurements in an transient electroosmotic flow, which include the initial position of the tracer molecules in the electroosmotic flow tagged with a focused laser beam and their subsequent position after a time delay of 5 ms. The simultaneous electroosmotic velocity and fluid temperature profiles across the 300 µm channel derived from the image pair, as well as the dynamic responses of the “in-channel” electroosmotic velocity and fluid temperature before and after “switch-on” of the electric field (260V/cm), are also shown in the figure. Further details about the MTV&T measurements in the electroosmotic flow to study the effects of Joule heating are available at Ref. 39. Temperature o ( C) Wall 40 150 3.0 2.5 Electroosmotic Velocity 100 50 Velocity 0 Temperature -50 Velocity (mm/s) Wall 35 300µm Spatial Location (μm) (a). 0.5 ms after laser pulse 2.0 1.5 30 1.0 Temperature 0.5 25 0 -100 -0.5 Turn on electric field -150 (b). 5 ms later 0 Temperature 30 o ( C) 1 2 3 4 5 32 34 36 38 40 20 -1.0 0 5 10 15 20 25 30 35 Time (seconds) Velocity(mm/s) (c). Instantaneous velocity and temperature profiles (d). dynamic response of “in-channel” electroosmotic velocity and fluid temperature Figure 8. MTV&T measurements in a transient electroosmotic flow (Lum et al. 38) V. Conclusion A completely molecule-based method, named Molecular Tagging Velocimetry and Thermometry (MTV&T) technique, was presented for the simultaneous whole-field mapping of velocity and temperature fields in fluid flows. The method uses a molecular tagging approach that combines molecular tagging velocimetry (MTV) with molecular tagging thermometry (MTT), and because of its molecular nature it eliminates issues such as the tracking of the flow by seed particles. The water-soluble phosphorescent triplex, 1-BrNp•Mβ-CD•ROH, was used as a tracer for both velocity and temperature measurements. A pulsed laser was used to “tag” the molecules in the regions of interest. The long-lived photoluminescence of the tagged molecules were imaged at two successive times after the same laser excitation pulse. The displacement of the tagged molecules between the two images provides the estimation of flow velocity. The phosphorescence intensity decay rate (i.e. phosphorescence lifetime) of the tagged tracer molecules is used to determine the fluid temperature through the temperature dependence of phosphorescence lifetime. The implementation of the MTV&T technique is demonstrated by its applications to study the wake behavior behind a heated cylinder and the transient behavior of an electroosmotic flow inside a microchannel. The measurements demonstrate that MTV&T can be a viable tool for simultaneous mapping of velocity and temperature fields in fluids flows. Acknowledgments This work was supported by the CRC Program of the National Science Foundation, Grant Number CHE0209898, and made use of shared facilities of the MRSEC Program of the National Science Foundation, Award Number DMR-9809688. 10 American Institute of Aeronautics and Astronautics 44th AIAA Aerospace Sciences Meeting and Exhibit AIAA-2006-0041 Jan 9 – 12, 2006, Reno, Nevada References 1. Antonia, R. A., Prubhu A. and Stephenson S. E. (1975) “Conditionally sampled measurements in a heated turbulent jet, ” J. Fluid Mech., Vol. 72, 455-480. 2. Chevray, R. and Tutu, N. K. (1978) “Intermittency and preferential transport of heat in a round jet,” J. Fluid Mech., Vol. 88, 133-160. 3. Dibble, R. W., Kollmann, W., and Schefer, R. W. (1984) “Conserved scalar fluxes measurement in a turbulent non-premixed flame by combined Laser Doppler Velocimetry and laser Raman Scattering,” Combustion and Flame, Vol. 55, 307-321. 4. Lemoine, L., Antonie, Y., Wolff, M. and Lebouche, M. (1999) “Simultaneous temperature and 2D velocity measurements in a turbulent heated jet using combined laser-induced fluorescence and LDA,” Exp. Fluids, Vol. 26, 315-323. 5. Sakakibara, J., Hishida, K. and Maeda, M. (1997) “Vortex structure and heat transfer in the stagnation region of an impinging plane jet,” Int. J. Heat and Mass Transfer, Vol. 40, 3163-3176. 6. Hishida, K. and Sakakibara, J. (2000) “Combined Planar laser-induced fluorescence – particle image velocimetry technique for velocity and temperature fields,” Exp. Fluids, Vol. 29, s129-s140. 7. Grissino, A. S., Hart, D. P., and Lai, W. T. (1999) “Combined dual emission LIF and PIV to resolve temperature and velocity,” Proceedings of the 3rd International Workshop on Particle Image Velocimetry, Sept. 16-18, 1999, Santa Barbara, CA, USA. 8. Dabiri, D. and Gharib, M. (1991) “Digital Particle Image Thermometry: the method and implementation,” Exp. Fluids, Vol. 11, 77-86. 9. Park, H. G., Dabiri, D. and Gharib, M. (2001) “Digital particle image velocimetry/thermometry and application to the wake of a heated circular cylinder,” Exp. Fluids, Vol. 30, 327-338. 10. Raffel, M., Willert, C. and Kompenhans, J. (1998), Particle Image Velocimetry. A Practical Guide. Springer 11. Gendrich, C. P., Koochesfahani, M. M. and Nocera, D. G. (1997) “Molecular Tagging Velocimetry and other novel application of a new phosphorescent supramolecule,” Exp. Fluids, Vol. 23, 361-372. 12. Koochesfahani, M. M. (1999) "Molecular Tagging Velocimetry (MTV): Progress and Applications," AIAA Paper No. AIAA-99-3786. 13. Hartmann, W. K., Gray, M. H. B., Ponce, A. and Nocera, D. G. (1996) “Substrate induced phosphorescence from cyclodextrin • lumophore host-guest complex,” Inorg. Chim. Acta., Vol. 243, 239-248. 14. Ponce, A., Wong, P. A., Way, J. J. and Nocera, D. G. (1993) “Intense phosphorescence trigged by alcohol upon formation of a cyclodextrin ternary complex,” J. Phys. Chem., Vol. 97, 11137-11142. 15. Bohl, D, Koochesfahani, M. and Olson, B. (2001) "Development of stereoscopic Molecular Tagging Velocimetry," Exp. Fluids, Vol. 30, No. 2, 302-308. 16. Falco, R. E. and Nocera, D. G. (1993) "Quantitative multipoint measurements and visualization of dense solid-liquid flows using laser induced photochemical anemometry (LIPA),” in Particulate Two-Phase Flow, Ed. M. C. Rocco; ButterworthHeinemann, 59-126. 17. Koochesfahani, M. M., Cohn, R. K., Gendrich, C. P., and Nocera, D. G. (1996) Molecular tagging diagnostics for the study of kinematics and mixing in liquid phase flows, Proceedings of the Eighth International Symposium on Applications of Laser Techniques to Fluids Mechanics, July 8 - 11, 1996, Lisbon, Portugal, vol. I, 1.2.1-1.2.12; Also in Developments in Laser Techniques and Fluid Mechanics, Chapter 2, section 1, p. 125, Eds. Adrian, Durao, Durst, Maeda, and Whitelaw; SpringerVerlag, Berlin, 1997. 18. Koochesfahani, M. M., Cohn, R. K. and Mackinnon, C. G. (2000) "Simultaneous whole-field measurements of velocity and concentration fields using combined MTV and LIF," Meas. Sci. Technol., Vol. 11, 1289-1300 19. Lempert, W. R. and Harris, S. R. (2000) Molecular Tagging Velocimetry, Flow Visualization – Techniques and Examples, ed. A. J. Smits and T. T. Lim (London: Imperial College Press, London) 73-92. 20. Koochesfahani, M. M. (Ed) (2000) Special Feature: Molecular Tagging Velocimetry, Meas. Sci. Technol., Vol. 11, No. 9, 1235-1300. 21. Gendrich, C. P. and Koochesfahani, M. M. (1996) "A spatial correlation technique for estimating velocity fields using Molecular Tagging Velocimetry (MTV)," Exp. Fluids, Vol. 22, No. 1, 67-77. 22. Pringsheim, P. (1949) Fluorescence and Phosphorescence, Interscience publishers, Inc., New York 23. Hu, H., and Koochesfahani, M. M. (2003) “A novel technique for quantitative temperature mapping in liquid by measuring the lifetime of laser induced phosphorescence,” Journal of Visualization, Vol. 6, No. 2, 143-153. 24. Hu, H. and Koochesfahani, M. M. “Molecular tagging velocimetry and thermometry (MTV&T) technique and its application to the wake of a heated circular cylinder”, Measurement Science and Technology, 2005 (accepted, in press). 25. Ferraudi, G. J. (1988) Elements of Inorganic Photochemistry, Wiley-Interscience, New York. 26. Thompson, S. L. and Maynes, D. (2001) “Spatially resolved temperature measurement in a liquid using laser induced phosphorescence,” J. Fluid Engr., Vol. 123, 293-302. 27. Coppeta, J. and Rogers, C. (1998) "Dual emission laser induced fluorescence for direct planar scalar behavior measurements," Exp. Fluids, Vol. 25, No. 1, 1-15. 28. Hu, H., Lum, C., and Koochesfahani, M. M. (2005) “Molecular tagging thermometry with adjustable temperature sensitivity,” submitted to Exp. Fluids; in review. 29. Reuss, F. F. (1809). Charge-induced flow. In Proceedings of the Imperial Society of Naturalists of Moscow 3:327–44. 30. Probstein, R. (1994). Physicochemical Hydrodynamics: An Introduction. Wiley Interscience. 11 American Institute of Aeronautics and Astronautics 44th AIAA Aerospace Sciences Meeting and Exhibit AIAA-2006-0041 Jan 9 – 12, 2006, Reno, Nevada 31. Guijt, R. M., A. Dodge, G. W. K. van Dedem, N. F. de Rooij, and E. Verpoorte.(2003), “Chemical and physical processes for integrated temperature control in microfluidic devices”. Lab Chip 3(1):1–4. 32. Erickson, D., D. Sinton, and D. Li. (2003). Joule heating and heat transfer in poly(dimethylsiloxane) microfluidic systems. Lab Chip 3:141–9. 33. Dawoud, A., T. Kawaguchi, Y. Markushin, M. D. Porter, and R. Jankowiak. (2005). Separation of catecholamines and dopamine-derived DNA adduct using a microfluidic device with electrochemical detection. Electrophoresis, (In review). 34. Santiago, J. G., S. T. Wereley, C. D. Meinhart, D. J. Beebe, and R. J. Adrian. (1998). A particle image velocimetry system for microfluidics. Exp. Fluids 25:316–9. 35. Pual, P. H., M. G. Garguilo, and D. J. Rekesraw. (1998). “Imaging of pressure and electrokinatically driven flows through open capillaries”. Anal. Chem. 70:2459–67. 36. Molho, J. I., A. E. Herr, B. P. Mosier, J. G. Santiago, T. W. Kenny, R. A. Brennen, G. B. Gordon, and B. Mohammadi. (2001). “Optimization of turn geometries for microchip electrophoresis”. Anal. Chem. 73:1350–60. 37. Ross, D., M. Gaitan, and L. E. Locascio. (2001). “Temperature measurement in microfluidic systems using a temperaturedependent fluorescent dye”. Anal. Chem. 73:4117–23. 38. Lum, C., H. Hu, and M. M. Koochesfahani. (2004) “Simultaneous velocity and temperature measurements in electroosmotically driven flow in a microchannel using molecular tagging velocimetry and thermometry”. In Proceedings of the 57th Annual Meeting of APS Division of Fluid Dynamics. 21–23 November, Seattle, Washington. 39. Lum, C. (2005), “An experimental study of pressure- and electroosmotically-driven flows in microchannels with surface modifications”. Ph.D. thesis, Department of Mechanical Engineering, Michigan State University. 12 American Institute of Aeronautics and Astronautics