Linking Practice Guidelines & Abstraction of Electronic Medical Records to Measure
advertisement

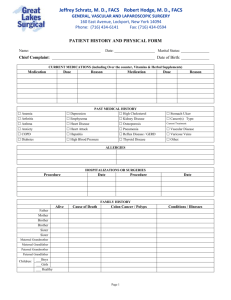
Linking Practice Guidelines & Abstraction of Electronic Medical Records to Measure the Quality of Cancer Care AcademyHealth Annual Research Meeting June 30, 2009 Chicago, IL George L. Jackson, Ph.D., MHA Assistant Professor Durham Veterans Affairs Medical Center Duke University Medical Center Cancer Treatment in the VA • Largest provider of cancer treatment in the United States – 3% of cancer cases in the United States – >43,000 incident cases in 2005 – >4,600 new colorectal cancer cases Source: VA Central Cancer Registry Colorectal Cancer (CRC) • In the United States – 3rd leading cause of cancer cases • Estimated 147,000 new cases in 2009 – 2nd leading cause of cancer deaths • Estimated 50,000 deaths in 2009 Source: Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin 2009. Colorectal Cancer Performance Screening, Diagnosis, and Treatment: Simplified Process View Screening Diagnosis Work-up, (possibly Neo-adjuvant Therapy), and Surgery Adjuvant Threrapy Developed for the VA Colorectal Cancer Care Collaborative (C4) Surveillance EPRP CRC Special Study Specific Aims • VA External Peer Review Program (EPRP) special study explored CRC in order to: – Determine VA-system gaps in established standards of care – Identify opportunities to improve CRC care – Test the process of obtaining information on the quality of cancer care VA External Peer Review Program (EPRP) • VA’s official quality measurement program – Abstracts data from the VA electronic health record [Computerized Patient Record System] – Conducted by Office of Quality and Performance – Contractor – West Virginia Medical Institute • Data abstracted as part of a one-time special study of EPRP • CRC data were abstracted remotely between June-September 2007 • CRC quality indicators not official VA performance measures Special Study Methods • Obtaining the CRC patient sample – Based on data from the VA Decision Support System (DSS) – Goal to define a sample of patients treated within the VA and diagnosed between October 1, 2003 and March 31, 2006 – Not intended to capture all VA CRC patients diagnosed during the time period DSS Search Inclusion • Between July 1, 2003-June 30, 2006, had CRC diagnosis code plus one of the following: – Medical oncology plus radiation oncology, surgery, hospice, pathology, gastroenterology, or colonoscopy – Radiation oncology only – Surgery plus medical oncology, radiation oncology, hospice, pathology, gastroenterology, or colonoscopy – Hospice only – Pathology plus medical oncology, radiation oncology, surgery, or hospice Final Study Eligibility • Eligibility for final dataset – Diagnosed October 1, 2003 to March 31, 2006 – Stage I-IV colon/rectal cancer – Incident case (initial diagnosis of colon/rectal cancer during the study period) – Received definitive surgery for colon/rectal cancer (e.g. partial colectomy) Defining Quality Indicators • 7 measures of guideline concordance – Based on 2003 version of the National Comprehensive Cancer Network (NCCN) clinical practice guidelines for colon and rectal cancer • 3 timeliness of care measures – Not guideline based – Timeliness of care is focus of VA performance improvement Quality Indicators 1. Documented preoperative CT scan of the abdomen and pelvis (or patient refusal of CT scan) for stage II and III colon/rectal cancer patients undergoing curative-intent surgical resection. -More recent guidelines also recommend a chest CT 2. Preoperative CEA [carcinoembryonic antigen] determination for stage II and III colon/rectal cancer patients undergoing curative-intent surgical resection. Quality Indicators 3. 13 or more lymph nodes resected for stage II and III colon/rectal cancer patients undergoing curative-intent surgical resection. -Guidelines say ≥12 lymph nodes -Error in data collection instrument caused use of ≥13 4. Documented clear margins for stage II and III colon/rectal cancer patients undergoing curative-intent surgical resection. Quality Indicators 5. Documented referral to a medical oncologist for patients with stage II and III colon/rectal cancer (or documented reason why not). 6. Adjuvant (post-operative) 5-FU or capecitabine following curative-intent resection of stage III colon/rectal cancer (or documented reason why not). Quality Indicators 7. Surveillance colonoscopy within 7 to 18 months after curative-intent resection for stage I, II and III colon/rectal cancer patients with no preoperative obstructing lesion documented. Timeliness Indicators 8. Days from diagnosis to initiation of treatment for stage II and III colon/rectal cancer patients who had curative-intent surgical resection, chemotherapy, and/or radiation therapy. 9. Days from curative-intent surgical resection to start of adjuvant (postoperative) chemotherapy treatment for stage II and III colon/rectal cancer patients. Timeliness Indicators 10. Days from curative-intent surgical resection to surveillance colonoscopy in stage I, II, and III colon/rectal cancer patients with no preoperative obstructing lesion documented. Patient Characteristics (n = 2,777) VA Medical Centers 128 of 153 total represented Stage Stage I Stage II Stage III Stage IV 27.5% 33.2% 29.1% 10.3% Age Mean age (SD) 65.0 (10.6) Gender Male 97.9% Race African American White Other Unknown 13.0% 71.9% 13.6% 14.2% Year of Diagnosis 2003 2004 2005 2006 10.0% 45.2% 38.9% 6.0% Results # Indicator Description n % 1 Documented preoperative CT scan 1,729 72.1% 2 Preoperative CEA 1,729 82.8% 3 13 or more lymph nodes resected 1,729 42.7% 4 Documented clear margins 1,729 81.1% 5 Documented medical oncology referral 1,729 77.5% 6 Adjuvant 5-FU based chemotherapy 808 73.5% 7 Surveillance colonoscopy = 7-18 months 1,259 43.5% mean (SD) median 8 Time from diagnosis to treatment (days) 1,729 26.6( 38.2) 29.0 9 Time from surgery to adjuvant chemo (days) 767 64.9 (54.9) 50 10 Time from surgery to surveillance colonoscopy (days) 644 444.1 (182.1) 393.0 Limitations • Data were remotely abstracted, potentially impacting what an abstractor could view. • Information on care from outside the VA would have only been captured if information was in the VA record. • Search algorithm assumes appropriate DSS specialty mapping at the facility level when patients were seen. Conclusions • VA appears to perform well in meeting established guidelines for CRC work-up and treatment. • Greatest opportunity for improvement in the area of surveillance. • With the possible exception of surveillance, VA compares well with private sector.1 • Lack of access to all parts of the medical record, even in the presence of an electronic health record, may result in an underestimate of quality. 1 – Malin JL, Schneider EC, Epstein AM, Adams J, Emanuel EJ, Kahn KL. Results of the National Initiative for Cancer Care Quality: How can we improve the quality of cancer care in the United States? J Clin Oncol. Feb 1 2006;24(4):626-634. Project Team VA Center for Health Services Research in Primary Care • • • • • • • • • • • George L. Jackson, Ph.D., MHA1,2 L. Douglas Melton, MPH1,3 David H. Abbott, MS, MPH1 Leah L. Zullig, MPH1 Diana L. (Dede) Ordin, MD, MPH4 Steven C. Grambow, Ph.D.1,2 Natia S. Hamilton, MA1 S. Yousuf Zafar, MD, MHS1,2 Ziad F. Gellad, MD, MPH1,2 Michael J. Kelley, MD1,2,5 Dawn T. Provenzale, MD, MS1,2 1 – Durham Veterans Affairs Medical Center; 2 – Duke University Medical Center; 3 – University of North Carolina at Chapel Hill; 4 – VA Office of Quality and Performance; 5 – VA Office of Patient Care Services Funding • VA Office of Quality and Performance • Career Development and Postdoctoral Awards – VA Health Services Research & Development (HSR&D) Merit Review Entry Program [MRP 05-312] – Agency for Healthcare Research and Quality (AHRQ) [T32 HS000079] – National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) [T32 DK007568-17 and 5 K24 DK002926] Contact Information George L. Jackson, Ph.D., MHA Center for Health Services Research in Primary Care (HSR&D), Durham VA Medical Center Email: george.jackson3@va.gov Phone: (919) 286-0411 x 7091 The views expressed in this presentation are those of the presenter and do not necessarily reflect the position or policy of the Department of Veterans Affairs or the United States government.